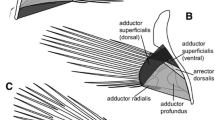
Summary
A histochemical study was carried out on muscle fibre types in the myotomes of post-larval and adult stages of seven species of notothenioid fish. There was little interspecific variation in the distribution of muscle fibre types in post-larvae. Slow fibres (diameter range 15–60 μm) which stained darkly for succinic dehydrogenase activity (SDHase) formed a superficial layer 1–2 fibres thick around the entire lateral surface of the trunk. In all species a narrow band of very small diameter fibres (diameter range 5–62 μm), with only weak staining activity, occurred between the skin and slow fibre layer. These have the characteristics of tonic fibres found in other teleosts. The remainder of the myotome was composed of fast muscle fibres (diameter range 9–75 μm), which stain weakly for SDHase, α-glycerophosphate dehydrogenase, glycogen and lipid. Slow muscle fibres were only a minor component of the trunk muscles of adult stages of the pelagic species Champsocephalus gunnari and Pseudochaenichthys georgianus, consistent with a reliance on pectoral fin swimming during sustained activity. Of the other species examined only Psilodraco breviceps and Notothenia gibberifrons had more than a few percent of slow muscle in the trunk (20%–30% in posterior myotomes), suggesting a greater involvement of sub-carangiform swimming at cruising speeds. The ultrastructure of slow fibres from the pectoral fin adductor and myotomal muscles of a haemoglobinless (P. georgianus) and red-blooded species (P. breviceps), both active swimmers, were compared. Fibres contained loosely packed, and regularly shaped myofibrils numerous mitochondria, glycogen granules and occasional lipid droplets. Mitochondria occupied >50% of fibre volume in the haemoglobinless species P. georgianus, each myofibril was surrounded by one or more mitochondria with densely packed cristae. No significant differences, however, were found in mean diameter between fibres from red-blooded and haemoglobinless species. The activities of key enzymes of energy metabolism were determined in the slow (pectoral) and fast (myotomal) muscles of N. gibberifrons. In contrast to other demersal Antarctic fish examined, much higher glycolytic activities were found in fast muscle fibres, probably reflecting greater endurance during burst swimming.
Similar content being viewed by others
References
Andriashev AP (1964) On the composition and origin of the Antarctic fish fauna. In: Carrick R, Holdgate M, Provost J (eds) Biologie Antarctique (Prem Symp SCAR). Hermann, Paris, pp 271–272
Bennet AF (1978) Activity metabolism of the lower vertebrates. Annu Rev Physiol 40:447–469
Batty RS (1984) Development of swimming movements and musculature of larval herring (Clupea harengus). J Exp Biol 110:217–222
Davison W, Macdonald JA (1985) A histochemical study of the swimming musculature of Antarctic fish. NZ J Zool 12:473–483
Davison W, Foster ME, Franklin CE, Taylor HH (1988) Recovery from exhausting exercise in an Antarctic fish, Pagothenia borchgrevinki. Polar Biol 8:167–171
DeWitt HH (1971) Coastal and deepwater benthic fishes of the Antarctic. In: Bushnell VC (ed) Antarctic map folio, vol 15. American Geographical Society, New York, pp 1–10
Dunn JF, Johnston IA (1986) Metabolic constraints on burst-swimming in the Antarctic teleost Notothenia neglecta. Mar Biol 91:433–440
Eastman JT, DeVries AL (1981) Buoyancy adaptations in a swim-bladderless Antarctic fish. J Morphol 167:91–102
Eastman JT, DeVries AL (1982) Buoyancy studies on Notothenioid fishes in McMurdo Sound, Antarctica. Copeia 1982:385–393
Eastman JT, DeVries AL (1985) Adaptations for cryopelagic life in the Antarctic Notothenioid fish Pagothenia borchgrevinki. Polar Biol 4:45–52
Egginton S, Sidell BD (1986) Changes in mitochondrial spacing in fish muscle induced by environmental temperature. J Physiol (London) 373:78pp
El-Fiky N, Hinterleitner S, Wieser W (1987) Differentiation of swimming muscles and gills, and development of anaerobic power in the larvae of cyprinid fish (Pisces, Teleostei). Zoomorphology 107:126–132
Fitch NA, Johnston IA, Wood RE (1984) Skeletal muscle capillary supply in a fish that lacks respiratory pigments. Resp Physiol 57:201–211
Forster ME, Franklin CE, Taylor HH, Davison W (1987) The aerobic scope of an Antarctic fish Pagothenia borchgrevinki and its significance for metabolic cold adaptation. Polar Biol 8:155–159
Harrison P, Nicol CJM, Johnston IA (1987) Gross morphology, fibre composition and mechanical properties of pectoral fin muscles in the Antarctic teleost Notothenia neglecta Nybelin. In: Kullander SO, Fernholm B (eds) Proc Vth Congr Eur Ichthyol, Stockholm 1985, pp 459–465
Hoppeler H, Kayar SR, Claasen H, Uhlmann E, Karas RH (1987) (Switzerland and U.S.A.): Adaptive variation in the mammalian respiratory system in relation to energetic demand: III. Skeletal muscles: setting the demand for oxygen (RSP 01300). Resp Physiol 69:27–46
Johnston IA (1987) Respiratory characteristics of muscle fibres in a fish (Chaenocephalus aceratus) that lacks haem pigments. J Exp Biol 133:415–428
Johnston IA, Patterson S, Ward S, Goldspink G (1974) The histochemical demonstration of myofibrillar adenosine triphosphatase activity in fish muscle. Can J Zool 52:871–877
Johnston IA, Harrison P (1985) Contractile and metabolic characteristics of muscle fibres from Antarctic fish. J Exp Biol 116:223–236
Johnston IA, Maitland B (1980) Temperature acclimation in crucian carp Carassius carassius L. morphometric analyses of muscle fibre ultrastructure. J Fish Biol 17:113–125
Johnston IA, Camm J-P (1987) Muscle structure and differentiation in pelagic and demersal stages of the Antarctic teleost Notothenia neglecta. Mar Biol 94:183–190
Johnston IA, Camm J-P, White M (1988) Specialisation of swimming muscles of a pelagic Antarctic fish, Pleuragramma antarcticum. Mar Biol 100:3–12
Kilarski W, Kozlowska M (1987) Comparison of ultrastructure and morphometrical analysis of tonic, white and red muscle fibres in the myotome of teleost fish (Neomacheilus barbatulus L.). Z Mikroski-Anat Forsch 4:S636–648
Kunzmann A (1987) Gill morphometrics of an Antarctic fish, Pleuragramma antarcticum. Proc Vth Congr Eur Ichtyol, Stockholm 1985, pp 467–468
Montgomery JC, Macdonald JA (1984) Performance of motor systems in Antarctic fishes. J Comp Physiol 154A:241–248
Nachlas MM, Tsou KC, De Sonza E, Cheng CS, Seligman AM (1957) Cytochemical demonstration of succinic dehydrogenase by the use of a new p-nitrophenyl substituted ditetrazole. J Histochem Cytochem 5:420–436
Nybelin O (1947) Antarctic fishes. Sci Res Norw Antarct Exped 1927–1928, 26:1–76
Pennyquick CJ, Rezende MA (1984) The specific power output of aerobic muscle, related to the power density of mitochondria. J Exp Biol 108:377–392
Permitin YY (1970) The consumption of krill by Antarctic fishes. In: Holdgate MW (ed) Antarctic ecology, vol XV. Academic Press, London, pp 177–182
permitin YY, Tarverdieva MI (1972) Feeding of some species of Antarctic fishes in South Georgia area. J Ichtyol 12:104–114
Proctor C, Mosse PRL, Hudson RCL (1980) A histochemical and ultrastructural study of the development of the propulsive musculature of the brown trout Salmo trutta L, in relation to its swimming behaviour. J Fish Biol 16:309–329
Raamsdonk W van, Veet L van't, Veeken K, Heyting C, Pool CW (1982) Differentiation of muscle fibre types in the teleost Brachydanio rerio, the Zebra fish. Posthatching development. Anat Embryol 164:51–62
Smialowska E, Kilarski W (1981) Histological analysis of fibres in myotomes of antarctic fish (Admiralty Bay, King George Island, South Shetland Islands). I. Comparative analysis of muscle fibre size. Pol Polar Res 2:109–129
Targett TE (1981) Trophic ecology and structure of coastal Antarctic fish communities. Mar Ecol Prog Ser 4:243–263
Tyler S, Sidell BD (1984) Changes in mitochondrial distribution and diffusion distances in muscle of goldfish upon acclimation to warm and cold temperatures. J Exp Zool 232:1–9
Walesby NJ, Johnston IA (1980) Fibre types in the locomotory muscles of an Antarctic teleost, Notothenia rossii. A histochemical, ultrastructural and biochemical study. Cell Tissue Res 208:143–164
Wattenberg LW, Leong JL (1960) Effects of Coenzyme Q10 and Menadione on Succinic Dehydrogenase activity as measured by Tetrazolium salt reduction. J Histochem Cytochem 8:296–303
Weihs D (1980) Energetic significance of changes in swimming modes during growth of larval anchovy, Engraulis mordax. Fish Bull 77:597–604
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Dunn, J.F., Archer, S.D. & Johnston, I.A. Muscle fibre types and metabolism in post-larval and adult stages of notothenioid fish. Polar Biol 9, 213–223 (1989). https://doi.org/10.1007/BF00263769
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00263769