Summary
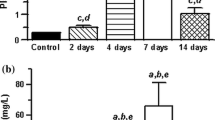
The appearance of low-molecular-weight metabolites of cisplatin in the cytosol of cells from the cortex and outer medulla, of the rat kidney has been examined using HPLC up to 24 h following cisplatin administration. Comparison was made between these metabolites and those present in plasma, urine and liver. The effect of sodium chloride (NaCl) pretreatment, which is known to reduce cisplatin-induced nephrotoxicity, on these metabolites was also investigated. Platinum levels in the kidney cortex and medulla and the cytosol reached maximal levels within 1 h of i.p. injection of 5 mg/kg cisplatin. At least six platinum species, including cisplatin, were present 1 h post-dosing, with the principal species being the parent drug; all of these species were either neutral or negatively charged. Although the concentration of most of the platinum species fell with time, that of one species eluting before cisplatin rose, and by 24 h it was the major metabolite. Cisplatin and two other major cytosolic platinum species were also present in urine and plasma, both of which also contained a number of charged species that were absent from the cytosol. The liver cytosol contained at least five metabolites 1 h post-dosing, but, in contrast to the kidney cytosol at the same time, the predominant species was that eluting before cisplatin and not cisplatin itself. One of the metabolites in the cytosol and urine had the same retention time as an adduct of cisplatin with glutathione and with cysteine. Urinary samples also contained a metabolite coeluting with aquated cisplatin. Pretreatment of animals with NaCl significantly reduced the platinum concentration in the kidney, with a corresponding decrease in the cytosolic metabolites; this may have contributed significantly to the reduction in cisplatin-induced nephrotoxicity after NaCl pretreatment. NaCl also significantly reduced a possible aquated species present in the urine, which may also have contributed to the reduction in nephrotoxicity. The data suggest that cisplatin itself may be the nephrotoxic species, since it is the intracellular platinum compound present in highest concentration during the early critical period after its administration.
Similar content being viewed by others
References
Borch RF, Pleasants ME (1979) Inhibition of cisplatinum nephrotoxicity by diethyldithiocarbamate rescue in a rat model. Proc Natl Acad Sci USA 76: 6611
Choie DD, DelCampo AA, Guarino AM (1980) Subcellular localisation of cis-dichlorodiammineplatinum (II) in rat kidney and liver. Toxicol Appl Pharmacol 55: 245
Daley-Yates PT, McBrien DCH (1983) Cisplatin metabolites: a method for their separation and for measurement of their renal clearance in vivo. Biochem Pharmacol 32: 181
Daley-Yates PT, McBrien DCH (1984) Cisplatin metabolites in plasma, a study of their pharmacokinetics and importance in the nephrotoxic and antitumour activity of cisplatin. Biochem Pharmacol 33: 3063
Daley-Yates PT, McBrien DCH (1985) The renal fractional clearance of platinum antitumour compounds in relation to nephrotoxicity. Biochem Pharmacol 34: 1423
Daley-Yates PT, McBrien DCH (1985) A study of the protective effect of chloride salts on cisplatin nephrotoxicity. Biochem Pharmacol 34: 2363
Jacobs C, Kalman SM, Tretton M, Weiner MW (1980) Renal handling of cis-diammine dichloro-platinum (II). Cancer Treat Rep 64: 1223
Lim MC, Martin RB (1976) The nature of cis amine Po (II) and antitumour cis ammine Pt (II) complexes in aqueous solutions. J Inorg Nucl Chem 38: 1911
Litterst CL (1981) Alterations in the toxicity of cis-dichlorodiammine platinum II and in tissue localisation of platinum as a function of NaCl concentration in the vehicle of administration. Toxicol Appl Pharmacol 61: 99
Litterst CL, Torres IJ, Guarino AM (1976) Plasma levels and organ distribution of platinum in the rat, dog, and dog-fish shark following single intravenous administration of cis-dichlorodiammineplatinum II. J Clin Hematol Oncol 7: 169
Mason RW, Hogg ST, Edwards IR (1986) Time course of the binding of platinum to subfractions of the kidney cytosol in the cisplatin-treated rat. Res Commun Chem Pathol Pharmacol 52: 51
Mason RW, Hogg ST, Edwards IR (1986) Distribution of Pt in the urine and kidney of the cisplatin treated rat. Toxicology 38: 219
Mauldin SK, Fichard FA, Plescia M, Wynck SD, Saucer A, Chaney SG (1986) High-performance liquid chromatographic separation of platinum complexes containing the cis-1,2-diaminocyclohexane carrier ligand. Anal Biochem 157: 129
McBrien DCH, Daley-Yates PT, Mistry P (1986) Modification of the renal toxicity of cisplatin by salts. In: McBrien DCH, Slater TF (eds) Biochemical mechanisms of platinum antitumour drugs. IRL Press, Oxford, p 255
Safirstein R, Miller P, Guttenplan JB (1984) Uptake and metabolism of cisplatin by rat kidney. Kidney Int 25: 753
Siddik ZH, Boxall FE, Harrap KR (1987) Flameless atomic absorption spectrophotometric determination of platinum in tissues solubilised in hyamine hydroxide. Anal Biochem 163: 21
Siddik ZH, Newell DR, Boxall FE, Harrap KR (1987) The comparative pharmacokinetics of carboplatin and cisplatin in mice and rats. Biochem Pharmacol 36: 1925
Slater TF, Ahmed M, Ibrahim SA (1977) Studies on the nephrotoxicity of cis-dichlorodiammine platinum (II) and related substances. J Clin Hematol Oncol 7: 534
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Mistry, P., Lee, C. & McBrien, D.C.H. Intracellular metabolites of cisplatin in the rat kidney. Cancer Chemother. Pharmacol. 24, 73–79 (1989). https://doi.org/10.1007/BF00263124
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00263124