Summary
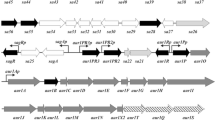
The cluster of streptomycin (SM) production genes in Streptomyces griseus was further analysed by determining the nucleotide sequence of genes strFGHIK. The products of the strF and/or strG genes may be involved in the formation of N-methyl-l-glucosamine, and that of the strH gene in the first glycosylation step condensing streptidine-6-phosphate and dihydrostreptose. The putative Strl protein showed strong similarity to the amino-terminal NAD(P)-binding sites of many dehydrogenases, especially of the glyceraldehyde-3-phosphate dehydrogenases. The product of the strK gene strongly resembles the alkaline phosphatase of Escherichia coli. It was shown that S. griseus excretes an enzyme that specifically cleaves both SM-6-phosphate and — more slowly — SM-3′-phosphate during the production phase for SM. The identity of this enzyme with the StrK protein was demonstrated by expression of the strK gene in Streptomyces lividans 66. Further evidence for an involvement of these genes in SM biosynthesis came from the fact that genes homologous to them were found in the equivalent gene cluster of the hydroxy-SM producer Streptomyces glaucescens; these, however, were in part differently organized. The ca. 5 kb DNA segment downstream of strI in S. griseus which contains the strK gene was found to be located in inverse orientation between the homologues of the aphD and strR genes in S. glaucescens.
Similar content being viewed by others
References
Alefounder PR, Perham RN (1989) Identification, molecular cloning and sequence analysis of a gene cluster encoding the Class II fructose 1,6-bisphosphate aldolase, 3-phosphoglycerate kinase and a putative second glyceraldehyde 3-phosphate dehydrogenase of Escherichia coli. Mol Microbiol 3:723–732
Ambartsumyan NS, Mazo AM (1980) Elimination of the secondary structure effect in gel sequencing. FEBS Lett 114:265–272
Arber W, Enquist L, Hohn B, Murray NE, Murray K (1983) Experimental methods for use with lambda. In: Hendrix RW, Roberts JW, Stahl FW, Weisberg RA (eds) Lambda II. Cold Spring Harbor Laboratory, New York, pp 433–466
Bibb MJ, Findlay PR, Johnson MW (1984) The relationship between base composition and codon usage in bacterial genes and its use in simple and reliable identification of protein coding sequences. Gene 30:157–166
Birch A, Häusler A, Vögtli M, Krek W, Hütter R (1989) Extremely large chromosomal deletions are intimately involved in genetic instability and genomic rearrangements in Streptomyces glaucescens. Mol Gen Genet 217:447–458
Branlant G, Branlant C (1985) Nucleotide sequence of the Escherichia coli gap gene: different evolutionary behaviour of the NAD+-binding domain and of the catalytic domain of D-glyceraldehyde-3-phosphate dehydrogenase. Eur J Biochem 150:61–66
Burnett WV, Henner J, Eckhardt T (1987) The nucleotide sequence of the gene coding for XP55, a major secreted protein from Streptomyces lividans. Nucleic Acids Res 15:3926
Chang PC, Kuo T-C, Tsugita A, Lee Y-HW (1990) Extracellular metalloprotease gene of Streptomyces cacaoi: structure, nucleotide sequence and characterization of the cloned gene product. Gene 88:87–95
Cundliffe E (1989) How antibiotic-producing organisms avoid suicide. Annu Rev Microbiol 43:207–233
Davies J (1990) What are antibiotics? Archaic functions for modern activities. Mol Microbiol 4:1227–1232
Distler J, Piepersberg W (1985) Cloning and characterisation of a gene from Streptomyces griseus coding for a streptomycin-phosphorylating activity. FEMS Microbiol Lett 28:113–117
Distler J, Mansouri K, Piepersberg W (1985) Streptomycin biosynthesis in Streptomyces griseus II: Adjacent genomic location of biosynthetic genes and one of two streptomycin resistance genes. FEMS Microbiol Lett 30:151–154
Distler J, Braun C, Ebert A, Piepersberg W (1987 a) Gene cluster for streptomycin biosynthesis in Streptomyces griseus: Analysis of a central region including the major resistance gene. Mol Gen Genet 208:204–210
Distler J, Ebert A, Mansouri K, Pissowotzki K, Stockmann M, Piepersberg W (1987 b) Gene cluster for streptomycin biosynthesis in Streptomyces griseus: Nucleotide sequence of three genes and analysis of transcriptional activity. Nucleic Acids Res 15:8041–8056
Distler J, Mansouri K, Mayer G, Piepersberg W (1990) Regulation of biosynthesis of streptomycin. In: The proceedings of the sixth international symposium on genetics of industrial microorganisms, Strasbourg
Donadio S, Tuan JS, Staver MJ, Weber M, Paulus TJ, Maine GT, Leung JO, Dewitt JP, Vara JA, Wang Y-G, Hutchinson CR, Katz L (1989) Genetic studies on erythromycin biosynthesis in Saccharopolyspora erythrea. In: Hershberger CL, Queener SW, Hegeman G (eds) Genetics and molecular biology of industrial microorganisms. American Society for Microbiology, Washington DC, pp 53–59
Doran JL, Leskiw BK, Aippersbach S, Jensen SE (1990) Isolation and characterization of a β-lactamase-inhibitor protein from Streptomyces clavuligerus and cloning and analysis of the corresponding gene. J Bacteriol 172:4909–4918
Duffaud G, Inouye M (1988) Signal peptidases recognize a structural feature at the cleavage site of secretory proteins. J Biol Chem 263:10224–10228
Forsman M, Hägström B, Lindgren L, Jaurin B (1990) Molecular analysis of β-lactamases from four species of Streptomyces: comparison of amino acid sequences with those of other β-lactamases. J Gen Microbiol 136:589–598
Frischauf AM, Lehrach H, Poustka A, Murray N (1983) Lambda replacement vectors carrying polylinker sequences. J Mol Biol 170:827–842
Froman BE, Tait RC, Gottlieb LD (1989) Isolation and characterization of the phosphoglucose isomerase gene from Escherichia coli. Mol Gen Genet 217:126–131
Gierasch LM (1989) Signal sequences. Biochemistry 28:923–930
Gram H, Rüger W (1985) Genes 55, gt, 47 and 46 of bacteriophage T4: the genomic organization as deduced by sequence analysis. EMBO J 4:257–264
Griesebach H (1978) Biosynthesis of sugar components of antibiotic substances. Adv Carbohydr Chem Biochem 35:81–126
Guijarro J, Santamaria R, Schauer A, Losick R (1988) Promoter determining the timing and spatial localization of transcription of a cloned Streptomyces coelicolor gene encoding a spore-associated polypeptide. J Bacteriol 170:1895–1901
Gurney ME, Heinrich SP, Lee MR, Yin H (1986) Molecular cloning and expression of neuroleukin, a neurotrophic factor for spinal and sensory neurons. Science 234:566–574
Haas MJ, Dowding JE (1975) Aminoglycoside-modifying enzymes. Methods Enzymol 43:611–633
von Heijne G (1986) A new method for predicting signal sequence cleavage sites. Nucleic Acids Res 14:4683–4690
Heinzel P, Werbitzky O, Distler J, Piepersberg W (1988) A second streptomycin resistance gene from Streptomyces griseus codes for streptomycin 3″-phosphotransferase. Relationship between antibiotic and protein kinases. Arch Microbiol 150:184–192
Hohn B, Murray K (1977) Packaging recombinant DNA molecules into bacteriophage particles in vitro. Proc Natl Acad Sci USA 74:3259–3263
Hopwood DA, Bibb MJ, Chater KF, Kieser T, Bruton CJ, Kieser HM, Lydiate DJ, Smith CP, Ward JM, Schrempf H (1985) Genetic manipulation of Streptomyces. A laboratory manual. The John Innes Foundation, Norwich
Hopwood DA, Malpartida F, Chater KF (1986) Gene cloning to analyse the organization and expression of antibiotic biosynthesis genes in Streptomyces. In: Kleinkauf H, von Döhren H, Dornauer H, Nesemann G (eds) Regulation of secondary metabolite formation. VCH Verlagsgesellschaft, Weinheim, pp 23–33
Huber M, Hutter R, Lerch K (1987) The promoter of the Streptomyces glaucescens mel operon. Nucleic Acids Res 15:8106
Katz E, Thompson CJ, Hopwood DA (1983) Cloning and expression of the tyrosinase gene from Streptomyces antibioticus in Streptomyces lividans. J Gen Microbiol 129:2703–2714
Kumada Y, Horinouchi S, Uozumi T, Beppu T (1986) Cloning of a streptomycin-production gene directing synthesis of N-methyl-L-glucosamine. Gene 42:221–224
Maniatis T, Fritsch EF, Sambrook J (1982) Molecular cloning. A laboratory manual. Cold Spring Harbor Laboratory Press, Cold Spring Harbor, New York
Mansouri K, Pissowotzki K, Distler J, Mayer G, Braun C, Ebert A, Piepersberg W (1989) Genetics of streptomycin production. In: Hershberger CL, Queener SW, Hegeman G (eds) Genetics and molecular biology of industrial microorganisms. American Society for Microbiology, Washington DC, pp 61–67
Marquardt R, Bräu B, Wöhner G, Schlingmann M, Präve P, Birr E, Wohlleben W, Aufderheide K, Schneider T, Pühler A (1989) Lysozym aus Streptomyceten, Verfahren zu seiner Gewinnung und seine Verwendung. Deutsche Patentschrift
Martin JF, Liras P (1989) Organization and expression of genes involved in the biosynthesis of antibiotics and other secondary metabolites. Annu Rev Microbiol 43:173–206
Mayer G, Vögtli M, Pissowotzki K, Hütter R, Piepersberg W (1988) Colinearity of streptomycin production genes in two species of Streptomyces. Evidence for occurrence of a second amidinotransferase gene. Mol Genet (Life Sci Adv) 7:83–87
Miller JH (1972) Experiments in molecular genetics. Cold Spring Harbor Laboratory, New York
Nakai R, Horinouchi S, Beppu T (1988) Cloning and nucleotide sequence of a cellulase gene, casA, from an alkalophilic Streptomyces strain. Gene 65:229–238
Nimi O, Kiyohara H, Mizoguchi T, Ohata Y, Nomi R (1970) Biosynthesis of streptomycin. Part VII. A specific enzyme responsible for dephosphorylation of phosphorylated streptomycin. Agr Biol Chem 34:1150–1156
Norrander J, Kempe T, Messing J (1983) Construction of improved M13 vectors using oligonucleotide-directed mutagenesis. Gene 26:101–106
Obata S, Taguchi S, Kumagai I, Miura K-I (1989) Molecular cloning and nucleotide sequence determination of gene encoding Streptomyces subtilisin inhibitor (SSI). J Biochem105:367–371
Ohnuki T, Imanaka T, Aiba S (1985) Self-cloning in Streptomyces griseus of an str gene cluster for streptomycin biosynthesis and streptomycin resistance. J Bacteriol 164:85–94
Pearson WR, Lipman DJ (1988) Improved tools for biological sequence analysis. Proc Natl Acad Sci USA 85:2444–2448
Piepersberg W, Distler J, Heinzel P, Perez-Gonzalez J-A (1988) Antibiotic resistance by modification: Many resistance genes could be derived from cellular control genes in actinomycetes — A hypothesis. Actinomycetology 2:83–98
Robbins PW, Albright C, Benfield B (1988) Cloning and expression of a Streptomyces plicatus chitinase (chitinase-63) in Escherichia coli. J Biol Chem 263:443–447
Sanger F, Nicklen S, Coulson AR (1977) DNA sequencing with chain-terminating inhibitors. Proc Natl Acad Sci USA 74:5463–5467
Seno ET, Baltz RH (1989) Structural organization and regulation of antibiotic biosynthesis and resistance genes in actinomycetes. In: Shapiro S (ed) Regulation of secondary metabolism in actinomycetes. CRC Press, London
Soltyk A, Krygsman P, Henderson G, Walczyk E, Escote-Carlson L, Plawinska E, Soostmeyer G, Malek L (1990) Isolation and characterization of two amylase genes from Streptomyces griseus. J Cell Biochem [Suppl] 14A:130
Sowadski JM, Handschumacher MD, Murthy HMK, Foster BA, Wyckoff HW (1985) Refined structure of alkaline phosphatase from Escherichia coli at 2.8 A resolution. J Mol Biol 186:417–433
Thompson CJ, Movva NR, Tizard R, Crameri R, Davies JE, Lauwereys M, Botterman J (1987) Characterization of the herbicide-resistance gene bar from Streptomyces hygroscopicus. EMBO J 6:2519–2523
Vögtli M (1989) Structure and expression of the sph gene of Streptomyces glaucescens and its genomic environment. Dissertation no. 8366, ETH Zurich
Vögtli M, Hatter R (1987) Characterization of the hydroxystreptomycin phosphotransferase gene (sph) of Streptomyces glaucescens: nucleotide sequencing and promoter analysis. Mol Gen Genet 208:195–203
Wohlleben W, Arnold W, Broer I, Hillemann D, Strauch E, Pühler A (1988) Nucleotide sequence of the phosphinothricin N-acetyltransferase gene from Streptomyces viridochromogenes Tü494 and its expression in Nicotiana tabaccum. Gene 70:25–37
Yanisch-Perron C, Vieira J, Messing J (1985) Improved M13 phage cloning vectors and host strains: Nucleotide sequences of the M13mp18 and pUC19 vectors. Gene 33:103–119
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Mansouri, K., Piepersberg, W. Genetics of streptomycin production in Streptomyces griseus: nucleotide sequence of five genes, strFGHIK, including a phosphatase gene. Molec. Gen. Genet. 228, 459–469 (1991). https://doi.org/10.1007/BF00260640
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00260640