Summary
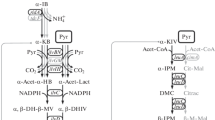
The phosphoenolpyruvate carboxylase gene (ppc) of Escherichia coli K-12 was cloned on the multi-copy plasmid pLG339. Plasmid pST101, which carried a 4.3-kb SalI fragment, was introduced into Serratia marcescens T-1165, which carried the seven regulatory mutations for three aspartokinases and two homoserine dehydrogenases. Strain T-1165[pST101] produced phosphoenolpyruvate carboxylase at a rate 26 times higher than the control strain T-1165[pLG339]. While T-1165[pST101] produced 63 mg/ml l-threonine in a medium containing sucrose and urea, whereas T-1165 only produced 52 mg/ml.
Similar content being viewed by others
References
Bachmann BJ (1972) Pedigrees of some mutant strains of Escherichia coli K-12. Bacteriol Rev 36:525–557
Backman K, Ptashne M, Gilbert W (1976) Construction of plasmids carrying the CI gene of bacteriophage λ. Proc Natl Acad Sci USA 73:4174–4178
Davis BD, Mingioli ES (1950) Mutants of Escherichia coli requiring methionine or vitamin B12. J Bacteriol 60:17–28
Komatsubara S (1987) Construction of amino acid-hyperproducing strains of Serratia marcescens by transductional and recombinant DNA techniques. In: Neijssel OM, van der Meer RR, Luyben KCAM (eds), Proceedings 4th European Congress on Biotechnology 1987, vol 4. Elsevier, Amsterdam, pp 757–765
Komatsubara S, Murata K, Kisumi M, Chibata I (1978) Threonine degradation by Serratia marcescens. J Bacteriol 135:318–323
Komatsubara S, Kisumi M, Chibata I (1983) Transductional construction of a threonine-hyperproducing strain of Serratia marcescens: lack of feedback controls of three aspartokinases and two homoserine dehydrogenases. Appl Environ Microbiol 45:1445–1452
Laemmli UK (1970) Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 227:680–685
Maniatis T, Fritsch EF, Sambrook J (1982) Molecular cloning. A laboratory manual. Cold Spring Harbor Laboratory, Cold Spring Harbor, NY
Matsumoto H, Hosogaya H, Suzuki K, Tazaki T (1975) Arginine gene cluster of Serratia marcescens. Jpn J Microbiol 19:35–44
Mori M, Shiio I (1985) Purification and some properties of phosphoenolpyruvate carboxylase from Brevibacterium flavum and its aspartate-overproducing mutant. J Biochem 97:1119–1128
Sabe H, Miwa T, Kodai T, Izui K, Hiraga S, Katsuki H (1984) Molecular cloning of the phosphoenolpyruvate carboxylase gene, ppc, of Escherichia coli. Gene 31:279–283
Stoker NG, Fairweather NF, Spratt BG (1982) Versatile low-copy-number plasmid vectors for cloning in Escherichia coli. Gene 18:335–341
Sugita T, Komatsubara S, Kisumi M (1987) Cloning and characterization of the mutated threonine operon (thrA 1 5A 2 5BC) of Serratia marcescens Gene 57:151–158
Takagi T, Kisumi M (1985) Isolation of a versatile Serratia marcescens mutant as a host and molecular cloning of the aspartase gene. J Bacteriol 161:1–6
Theodore TS, Englesberg E (1964) Mutant of Salmonella typhimurium deficient in the carbon dioxide-fixing enzyme phosphoenolpyruvic carboxylase. J Bacteriol 88:946–955
Thèze J, Saint-Girons I (1974) Threonine locus of Escherichia coli K-12: genetic structure and evidence for an operon. J Bacteriol 118:990–998
Umbarger HE (1978) Amino acid biosynthesis and its regulation. Annu Rev Biochem 47:533–606
Yoshinaga T, Izui K, Katsuki H (1970) Purification and molecular properties of allosteric phosphoenolpyruvate carboxylase from Escherichia coli. J Biochem 68:747–750
Yoshinaga T, Teraoka H, Izui K, Katsuki H (1974) Molecular properties of phosphoenolpyruvate carboxylase of Escherichia coli W. J Biochem 75:913–924
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Sugita, T., Komatsubara, S. Construction of a threonine-hyperproducing strain of Serratia marcescens by amplifying the phosphoenolpyruvate carboxylase gene. Appl Microbiol Biotechnol 30, 290–293 (1989). https://doi.org/10.1007/BF00256220
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00256220