Abstract
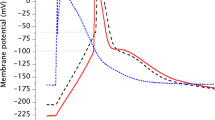
A method was developed to determine electrical potential differences across the plasma membrane of the microalga Dunaliella by means of potential-sensitive microelectrodes. Special emphasis was put on the measurement of the membrane potential in the acidophilic Dunaliella acidophila (optimal growth at pH 1.0), but neutrophilic, halotolerant Dunaliella species were used as reference systems. For Dunaliella acidophila positive membrane potentials (cytoplasma relative to the medium), ranging from +30 to +65mV were measured. Illumination caused a decrease of the positive potential by about 10 mV. The ATPase inhibitor omeprazole caused an increase of the positive membrane potential ranging from +60 to +100 mV, whereas the ionophore gramicidin caused a decrease of the MP to +10 to +30 mV. The salt tolerant, neutrophilic Dunaliella parva and Dunaliella bardawil exhibited negative membrane potentials in the order of -40 to -60mV, and light caused a hyperpolarization of about 10 mV. A negative membrane potential was measured also in D. acidophila cells transferred to pH 7.0. The physiological significance of a positive membrane potential for acidophilic algae is discussed.
Similar content being viewed by others
Abbreviations
- E m :
-
membrane potential
- PM:
-
plasma membrane
- TPB− :
-
tetraphenylborone anion
- TPP+ :
-
tetraphenyl-phosphonium cation
- SCN− :
-
isothiocyanate
References
Bakker E (1990) The role alkali-cation transport in energy coupling of neutrophilic and acidophilic bacteria: an assessment of methods and concepts. FEMS Microbiol Rev 75: 319–334
Blatt MR (1987) Electrical characteristics of stomatal guard cells. The ionic basis of the membrane potential and the consequence of potassium chloride leakage from micro-electrodes. Planta 170: 272–287
Blatt MR, Beilby MJ, Tester M (1990) Voltage dependence of the Chara proton pump revealed by current — voltage measurements during rapid metabolism blockade with cyanide. J Membrane Biol 114: 205–227
Brüggemann M, Weiger C, Gimmler H (1978) Synchronized culture of the halotolerant unicellular green alga Dunaliella parva. Biochem Physiol Pflanzen 172: 487–505
Bulychev AA, Andrianov VK, Kurella GA, Litvin FF (1972) Microelectrode measurements of the transmembrane potential of chloroplasts and its photoinduced changes. Nature 236: 175–177
Carandang JS, Pick U, Sekler I, Gimmler H (1992) K+ content and K+ fluxes in Dunaliella acidophila, an alga with positive electrical potentials. I. Low affinity uptake system. J Plant Physiol 139: 413–421
Gilmour DJ, Kaaden R, Gimmler H (1985) Vanadate inhibition of ATPase of Dunaliella parva in vitro and in vivo. J Plant Physiol 118: 111–126
Gimmler H, Kugel H, Leibfritz D, Mayer A (1988) Cytoplasmic pH of Dunaliella parva and Dunaliella acidophila as monitored by (31P) NMR spectroscopy and the DMO technique. Physiol Plant 74: 521–530
Gimmler H, Weis U, Kugel H, Treffney B (1989) Dunaliella acidophila (Kalina) Masyuk-an alga with a positive membrane potential. N Phytol 113: 175–184
Gimmler H, Schieder M, Kowalski M, Zimmermann U, Pick U (1991) Dunaliella acidophilia-an alga with a positive zeta potential. Plant Cell Environ 14: 261–269
Gimmler H, Weis U (1992) Dunaliella acidophila-life at pH 1.0. In: Avron M, Ben-Amotz A. (eds) Dunaliella-Physiology, biochemistry and biotechnology. CRS Press, Boca Raton, Fla., USA, pp 99–133
Gläser HU, Sekler I, Pick U (1990) Indications for a K+, H+ cotransport systems in plasma membranes from to acidophilic microorganism. Biochim Biophys Acta 1019: 293–299
Hirsch R, Carandang J, Gimmler H (1992) Cl- fluxes and Cl- content of Dunaliella acidophila-an alga with a positive membrane potential. J Exp Bot, 43: 887–896
Köhler K, Geisweid HJ, Simonis W, Urbach W (1963) Changes in the membrane potential and resistance caused by transient increase of potassium conductance in the unicellular green alga Eremosphaera viridis. Planta 159: 165–171
Lindberg P, Brandstrom A, Wallmark B, Mattson H, Rikner L, Hoffmann KJ (1990) Omeprazole-the 1st proton pump inhibitor. Med Res Rev 10: 1–54
Mitchell P (1961) Coupling of phosphorylation to electron and hydrogen transfer by a chemi-osmotic type of mechanism. Nature 191: 144–148
Mitchell P (1966) Chemiosmotic coupling in oxidative and photosynthetic phosphorylation. Biol Rev 41: 445–502
Raven JA (1976) Transport in algal cells. In: Pirson A, Zimmermann MH (eds) Encyclopedia of plant physiology (New Series). Springer, Berlin Heidelberg New York, vol. 2A, pp 129–188
Remis D, Bulychev AA, Kurella GA (1986) The electrical and chemical components of the proton-motive force in chloroplasts as measured with capillary and pH-sensitive micro-electrodes. Biochim Biophys Acta 852: 68–72
Schroeder JI (1988) K+ transport properties of K+ channels in the plasma membrane of Vicia faba guard cells. J Gen Physiol 92: 667–683
Sekler I, Gläser HU, Pick U (1991) Characterization of a plasma membrane H+ ATPase from the extremely acidophilic alga Dunaliella acidophila. J Membr Biol 121: 51–57
Serrano R (1985) Plasma membrane ATPase of plants and fungi. CRS Press, Boca Raton, Fla., USA
Serrano R (1988) Structure and function of proton translocating ATPase in plasma membranes of plants and fungi. Biochim Biophys Acta 947: 1–28
Serrano R (1989) Structure and function of plasma membrane ATPase. Ann Rev Plant Physiol Plant Mol Biol 40: 61–94
Serrano R (1990) Plasma membrane ATPase. In: Larson C, Möller IM (eds) The plant plasma membrane. Structure, function and molecular biology. Springer, Berlin Heidelberg New York, pp 127–153
Slayman Cl (1987) The plasma membrane ATPase of Neurospora: a proton pumping electroenzyme. J Bioenerg Biomembr 19: 1–20
Spanswick RM (1974) Evidence for an electrogenic ion pump in Nitella translucents. II Control of the light-stimulated component of the membrane potential. Biochim Biophys Acta 332: 387–398
Walker NA, Smith FA (1977) The H+ ATPase of the Chara cell membrane: its role in determing membrane P. D. and cytoplasmic pH. In: Thellier M, Monnier A, Demarty M, Dainty J (eds) Echanges ioniques transmembranes chez les végétaux. Publications de l'université de Rouen, pp 255–261
Zeldin MH (1977) Light-induced electrophysiological changes in Euglena. In: Thellier M, Monnier A, Demarty M, Dainty J (eds) Echanges ioniques transmembranes chez les végétaux. Publications de l'université de Rouen, 591–597
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Remis, D., Simonis, W. & Gimmler, H. Measurement of the transmembrane electrical potential of Dunaliella acidophila by microelectrodes. Arch. Microbiol. 158, 350–355 (1992). https://doi.org/10.1007/BF00245364
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00245364