Abstract
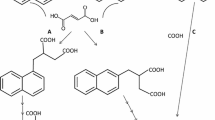
Aerobic organisms degrade hydroaromatic compounds via the hydroaromatic pathway yielding protocatechuic acid which is further metabolized by oxygenase-mediated ring fission in the 3-oxoadipate pathway. No information exists on anaerobic degradation of hydroaromatics so far. We enriched and isolated from various sources of anoxic sediments several strains of rapidly growing gram-negative bacteria fermenting quinic (1,3,4,5-tetrahydroxy-cyclohexane-1-carboxylic acid) and shikimic acid (3,4,5-trihydroxy-1-cyclohexene-1-carboxylic acid) in the absence of external electron acceptors. Quinic and shikimic acid were the only ones utilized of more than 30 substrates tested. The marine isolates formed acetate, butyrate, and H2, whereas all freshwater strains formed acetate and propionate as typical fermentation products. Aromatic intermediates were not involved in this degradation. Characterization of the isolates, fermentation balances for both hydroaromatic compounds, and enzyme activities involved in one degradation pathway are presented.
Similar content being viewed by others
Abbreviations
- BV:
-
benzyl viologen (1,1′-dibenzyl-4,4′-bipyridinium dichloride)
- CoA:
-
coenzyme A
- CTAB:
-
cetyltrimethylammonium bronide
- DCPIP:
-
2,4-dichlorophenolindophenol
- DTT:
-
1,4-dithiotheriol
- MV:
-
methyl viologen (1,1′-dimethyl-4,4′-bipyridinium dichloride)
- Tricine:
-
N-[tris-(hydroxymethyl)-methyl]-glycine
- Tris:
-
tris-(hydroxymethyl)-aminomethane
References
Bartholomew (1962) Variables influencing results, and the precise definition of steps in gram staining as a means of standardizing the results obtained. Stain Technol 37: 139–165.
Beri RK, Grant S, Roberts CF, Smith M, Hawkins AR (1990) Selective overexpression of the QUTE gene encoding catabolic 3-dehydroquinase in multicopy transformants of Aspergillus nidulans. Biochem J 265: 337–342
Blenden DC, Goldberg HS (1965) Silver impregnation stain for Leptospira and flagella. J Bacteriol 89: 899–900
Boudet A (1973) Quinic and shikimic acids in woody angiosperms. Phytochemistry 12: 363–370
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72: 248–254
Brewster D, Jones R, Parke D (1978) The metabolism of shikimate in the rat. Biochem J 170: 257–264
Bruce NC, Cain RB (1990) Hydroaromatic metabolism in Rhodococcus rhodochrous: purification and characterisation of its NAD-dependent quinate dehydrogenase. Arch Microbiol 154: 179–186.
Brune A, Schink B (1990a) Pryrogallol-to-phloroglucinol conversion and other hydroxyl-transfer reactions catalyzed by cell extracts of Pelobacter acidigallici. J Bacteriol 172: 1070–1076
Brune A, Schink B (1990b) A complete citric acid cycle in assimilatory metabolism of Pelobacter acidigallici, a strictly anaerobic, fermenting bacterium. Arch Microbiol 154: 394–399
Brune A, Schink B (1991) Phloroglucinol pathway in the striclty anaerobic Pelobacter acidigallici: fermentation of trihydroxybenzenes to acetate via triacetic acid. Arch Microbiol (in press)
Buchanan BB (1969) Role of ferredoxin in the synthesis of α-ketobutyrate from propionyl coenzyme A and carbon dioxide by enzymes from photosynthetic and nonphotosynthetic bacteria. J Biol Chem 244: 4218–4223
Cánovas J, Wheelis ML, Stanier RY (1968) Regulation of the enzymes of the β-ketoadipate pathway in Moraxella calcoacetica. 2. The role of protocatechuate as inducer. Eur J Biochem 3: 293–304
Cashion P, Holder-Franklin MA, McCully J and Franklin M (1977) A rapid method for base ratio determination of bacterial DNA. Anal Biochem 81: 461–466
Cord-Ruwisch R (1985) A quick method for the determination of dissolved and precipitated sulfides in cultures of sulfate-reducing bacteria. J Microbiol Methods 4: 33–36
Diekert GB, Thauer RK (1978) Carbon monooxide oxidation by Clostridium thermoaceticum and Clostridium formicoaceticum. J Bacteriol 136: 597–606
Dörner C, Schink B (1990) Clostridium homopropionicum sp. nov., a new strict anaerobe growing with 2-, 3-, or 4-hydroxybutyrate. Arch Microbiol 154: 342–348
Elstner EF, Youngman RJ, Oßwald W (1985) Superoxide dismutase. In: Bergmeyer HU (ed) Methods in enzymatic analysis, 3rd edn., vol III. Verlag Chemie, Weinheim, pp 293–302
Geever RF, Huiet L, Baum JA, Tyler BM, Patel VB, Rutledge BJ, Case ME, Giles NH (1989) DNA sequence, organization and regulation of the qa gene cluster of Neurospora crassa. J Mol Biol 207: 15–34
Gregersen T (1978) Rapid method for distinction of gram-negative from gram-positive bacteria. Eur J Appl Microbiol Biotechnol 5: 123–127
Grund E, Knorr C, Eichenlaub R (1990) Catabolism of benzoate and monohydroxylated benzoates by Amycolatopsis and Streptomyces spp. Appl Environ Microbiol 56: 1459–1464
Hayaishi O, Katagiri M, Rothberg S (1957) Studies on oxygenases: pyrocatechase. J Biol Chem 229: 905–920
Herbert RB (1981) The biosynthesis of secondary metabolites. Chapman & Hall, London, New York
Ingledew WM, Tai CC (1972) Quinate metabolism in Pseudomonas aeruginosa. Can J Microbiol 18: 1817–1824
Ingledew WM, Tresguerres MEF, Cánovas JL (1971) Regulation of the enzymes of the hydroaromatic pathway in Acinetobacter calcoaceticus. J Gen Microbiol 68: 273–282
Krieg NR, Holt JG (eds) (1984) Bergey's manual of systematic bacteriology, vol 1 Williams & Wilkins, Baltiore, Md., USA
Martin AK (1982) The origin of urinary aromatic compounds excreted by ruminants. 1. Metabolism of quinic acid, cyclohexanecarboxylic acid, and non-phenolic aromatic acids to benzoic acid. Br J Nutr 47: 139–154
Matthies C, Mayer F, Schnk B (1989) Fermentative degradation of purescine by new strictly anaerobic bacteria. Arch Microbiol 151: 498–505
Mesbah M, Premachandran U and Whitman W (1989) Precise measurement of the G+C content of deoxyribonucleic acid by high performance liquid chromatography. Int J Syst Bacteriol 39: 159–167
Minamikawa T (1976) A comparative study on the metabolism of quinic acid and shikimic acid in plants. Bot Mag Tokyo 89: 141–144
Möller B, Hermann K, (1983) Quinic acid esters of hydroxycinnamic acids in stone and pome fruit. Phytochemistry 22: 477–481
Nishimura H; Nonaka G-I, Nishioka I (1984) Seven quinic acid gallates from Quercus stenophylla. Phytochemistry 23: 2621–2623
Parke D, Ornston LN (1984) Nutritional divesity of Rhizobiaceae revealed by auxanography. J Gen Microbiol 130: 1743–1750
Pfennig N, Wagener S (1986) An improved method of preparing wet mounts for photomicrographs of microogranisms. J Microbiol Methods 4: 303–306
Platen H, Schink B (1987) Methanogenic degradation of acetone by an enrichment culture. Arch Microbiol 149: 136–141
Platen H, Schink B (1989) Anaerobic degradation of acetone and higher ketones via carboxylation by newly isolated denitrifying bacteria. J Gen Microbiol 135: 883–891
Platen H, Temmes A, Schink B (1990) Anaerobic degradation of acetone by Desulfococcus biacutus spec. nov.. Arch Microbiol 154: 355–361
Procházková L (1959) Bestimmung der Nitrate im Wasser. Z Anal Chem 167: 254–260
Schnell S, Brune A, Schink B (1991) Degradation of hydroxyhydroquinone by the strictly anaerobic fermenting bacterium Pelobactermassiliensis s sp. nov.. Arch Microbiol 155: 511–516
Smibert AM, Krieg NR (1981) General characterization. In: Gerhardt P (ed) Manual of methods for general bacteriology. American Society for Microbiology, Washington DC, pp 409–443
Stanier RY, Ingraham JL (1954) Protocatechuic acid oxidase. J Biol Chem 210: 799–808
Stoeckigt J, Zenk MH (1974) Enzymic synthesis of chlorogenic acid from caffeoyl coenzyme A and quinic acid. FEBS Lett 42: 131–134
Stouthamer AH (1979) The search for correlation between theoretical and experimental growth yields. In: Quayle JR (ed) Microbial biochemistry, vol 21. University Park Press, Baltimore, pp 1–47
Tamoaka J and Komagate K (1984) Determination of DNA base composition by reversed-phase high-performance liquid chromatography. FEMS Microbiol Lett 25: 125–128
van Kleef MAG, Duine JA (1988) Bacterial NAD(P)-independent quinate dehydrogenase is a quinoprotein. Arch Microbiol 150: 32–36
Wallhöfer P, Baldwin RL (1967) Pathway of propionate fermentation in Bacteroides ruminicola. J Bacteriol 93: 504–505
Wheeler LA, Halula M, DeMeo M, Sutter VL, Finegold SM (1979) Metabolism of shikimic, quinic, cyclohexanecarboxylic acids in germfree, conventional, and gnotobiotic rats. Curr Microbiol 2: 85–90
Whiting GC, Coggins RA (1971) The role of quinate and shikimate in the metabolism of lactobacilli. Antonie van Leuwenhoek J Microbiol Serol 37: 33–49
Whiting GC, Coggins RA (1974) A new nicotinamide-adenine dinucleotide-dependent hydroaromatic dehydrogenase of Lactobacillus plantarum and its role in formation of (-)t-3, t-4-dihydroxycycloheane-c-1-carboxylate. Biochem J 141: 35–42
Widdel F, Pfenning N (1981) Studies on dissimilatory sulfate-reducing bacteria that decompose fatty acids I. Isolation of a new sulfatereducer enriched with acetate from saline environments. Description of Desulfobacter postgatei gen. nov. sp. nov. Arch Microbiol 129: 395–400
Widdel F and Pfennig N (1984) Dissimilatory sulfate- or sulfurreducing bacteria. In: Krieg NR, Holt JG (eds) Bergey's manual of systematic bacteriology, vol 1, Williams & Wilkins, Baltimore, Md, pp 663–679
Yoshida S, Tazaki K, Minamikawa T (1975) Occurrence of shikimic and quinic acids in angiosperms. Phytochemistry 14: 195–197
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Brune, A., Schink, B. Anaerobic degradation of hydroaromatic compounds by newly isolated fermenting bacteria. Arch. Microbiol. 158, 320–327 (1992). https://doi.org/10.1007/BF00245360
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00245360