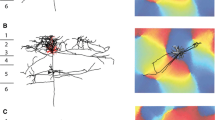
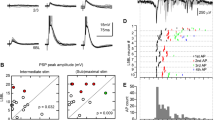
Summary
Inhibitory interactions between interneurones of the lateral geniculate nucleus (LGN) of the cat were studied with an indirect method based on intracellular recordings of synaptic responses in principal cells. Recurrent inhibitory postsynaptic potentials (IPSPs), evoked by antidromic activation of principal cell axons in the visual cortex, were depresse by a preceding stimulation of the optic tract or the visual cortex. Disynaptic feed-forward IPSPs, evoked by optic tract stimulation, were likewise depressed after cortex stimulation. The duration of the depression was in both cases about 100 ms. The effect was not due to conductance changes in the recorded principal cells or to activation of corticogeniculate fibres. The observations indicate that perigeniculate neurones, the recurrent inhibitory interneurones of the LGN, have mutual inhibitory connexions and that they also project to intrageniculate interneurones, the inhibitory cells in the feed-forward pathway to principal cells. These conclusions were supported by intracellular recordings from a few interneurones. No evidence was found for interaction between feed-forward interneurones activated from separate eyes or for a projection from intrageniculate interneurones to perigeniculate cells. The results point to an unexpected similarity in the organization of the recurrent inhibitory system of principal cells in the LGN and of spinal motoneurones. It is suggested that the recurrent system of the LGN serves as a variable gain regulator in analogy with a recently proposed model for the spinal system.
Similar content being viewed by others
References
Ahlśen G (1984) Brain stem neurones with differential projection to functional subregions of the dorsal lateral geniculate complex in the cat. Neuroscience 12: 817–838
Ahlsén G, Grant K, Lindström S (1982a) Monosynaptic excitation of principal cells in the lateral geniculate nucleus by corticofugal fibers. Brain Res 234: 454–458
Ahlsén G, Lindström S (1978) Axonal branching of functionally identified neurones in the lateral geniculate body of the cat. Neurosci Lett Suppl 1: 156
Ahlsén G, Lindström S (1982a) Excitation of perigeniculate neurones via axon collaterals of principal cells. Brain Res 236: 477–481
Ahlsén G, Lindström S (1982b) Mutual inhibition between perigeniculate neurones. Brain Res 236: 482–486
Ahlsén G, Lindström S (1983) Corticofugal projection to perigeniculate neurones in the cat. Acta Physiol Scand 118: 181–184
Ahlsén G, Lindström S, Lo F-S (1982b) Interactions between inhibitory interneurones in lateral geniculate nucleus of the cat. J Physiol (Lond) 328: 38–39P
Ahlsén G, Lindström S, Lo F-S (1982c) Functional distinction of perigeniculate and thalamic reticular neurones in the cat. Exp Brain Res 46: 118–126
Ahlsén G, Lindström S, Lo F-S (1983) Excitation of perigeniculate neurones from X and Y principal cells in the lateral geniculate nucleus of the cat. Acta Physiol Scand 118: 445–448
Ahlsén G, Lindström S, Lo F-S (1984) Inhibition from the brain stem of inhibitory interneurones of the cat's dorsal lateral geniculate nucleus. J Physiol (Lond) 347: 593–609
Ahlsén G, Lindström S, Sybirska E (1978) Subcortical axon collaterals of principal cells in the lateral geniculate body of the cat. Brain Res 156: 106–109
Ahlsén G, Lo F-S (1982) Projection of brain stem neurones to the perigeniculate nucleus and the lateral geniculate nucleus in the cat. Brain Res 238: 433–438
Andersen P, Andersson SA (1968) Physiological basis of the alpha rhythm. Appleton-Century-Crofts, New York
Burke W, Cole AM (1978) Extraretinal influences on the lateral geniculate nucleus. Rev Physiol Biochem Pharmacol 80: 105–166
Burke W, Sefton AJ (1966) Recovery of responsiveness of cells of lateral geniculate nucleus of rat. J Physiol (Lond) 187: 213–229
Cleland BG, Dubin MW, Levick WR (1971) Sustained and transient neurones in the cat's retina and lateral geniculate nucleus. J Physiol (Lond) 217: 473–496
Dubin MW, Cleland BG (1977) Organization of visual inputs to interneurons of lateral geniculate nucleus of the cat. J Neurophysiol 40: 410–427
Eccles JC, Fatt P, Koketsu K (1954) Cholinergic and inhibitory synapses in a pathway from motor-axon collaterals to motoneurones. J Physiol (Lond) 126: 524–562
Famiglietti EV, Peters A (1972) The synaptic glomerulus and the intrinsic neuron in the dorsal lateral geniculate nucleus of the cat. J Comp Neurol 144: 285–334
Ferster D, Lindström S (1983) An intracellular analysis of geniculo-cortical connectivity in area 17 of the cat. J Physiol (Lond) 342: 181–215
Friedlander MJ, Lin C-S, Stanford LR, Sherman SM (1981) Morphology of functionally identified neurons in lateral geniculate nucleus of the cat. J Comp Neurol 46: 80–128
Gilbert CD, Kelly JP (1975) The projection of cells in different layers of the cat's visual cortex. J Comp Neurol 163: 81–106
Guillery RW (1969) The organization of synaptic interconnections in the laminae of the dorsal lateral geniculate nucleus of the cat. Z Zellforsch Mikrosk Anat 96: 1–38
Hernandez-Peon R (1966) In frontiers in physiological psychology. Ed. Russell, Academic Press, New York
Hoffmann K-P, Stone J, Sherman SM (1972) Relay of receptive field properties in dorsal lateral geniculate nucleus of the cat. J Neurophysiol 35: 518–531
Hubel DH, Wiesel T (1961) Integrative action in the cat's lateral geniculate body. J Physiol (Lond) 155: 385–398
Hultborn H, Jankowska E, Lindström S (1971) Recurrent inhibition from motor axon collaterals of transmission in the Ia inhibitory pathway to motoneurones. J Physiol (Lond) 215: 591–612
Hultborn H, Lindström S, Wigström H (1979) On the function of recurrent inhibition in the spinal cord. Exp Brain Res 37: 399–403
Lee BB, Virsu V, Creutzfeldt OD (1983) Linear signal transmission from prepotentials to cells in the macaque lateral geniculate nucleus. Exp Brain Res 52: 50–56
Léger L, Sakai K, Salvert D, Touret M, Jouvet M (1975) Delineation of dorsal lateral geniculate afferents from the cat brain stem as visualized by the horseradish peroxidase technique. Brain Res 93: 490–496
Levick WR, Cleland BG, Dubin MW (1972) Lateral geniculate neurons of cat: retinal inputs and physiology. Invest Ophthal 11: 302–311
Lindström S (1975) Postsynaptic inhibition of the principal cell of the lateral geniculate body of the cat. Abstract ARVO Spring Meeting Sarasota, Florida
Lindström S (1982) Synaptic organization of inhibitory pathways to principal cells in the lateral geniculate nucleus of the cat. Brain Res 234: 447–453
Lindström S (1983) Interneurones in the lateral geniculate nucleus with monosynaptic excitation from retinal ganglion cells. Acta Physiol Scand 119: 101–103
Lindström S, Wróbel A (1984) Separate inhibitory systems for X and Y principal cells in the lateral geniculate nucleus of the cat. Acta Physiol Scand 121: 37A
Montero VM, Singer W (1984) Ultrastructure and synaptic relations of neural elements containing glutamic acid decarboxylase (GAD) in the perigeniculate nucleus of the cat: a light and electron microscopic immunocytochemical study. Exp Brain Res 56: 115–125
Moruzzi G, Magoun HW (1949) Brain stem reticular formation and activation of the EEG. Electroenceph Clin Neurophysiol 1: 455–473
Ryall RW (1970) Renshaw cell mediated inhibition of Renshaw cells: patterns of excitation and inhibition from impulses in motor axon collaterals. J Neurophysiol 33: 257–270
Schmielau F, Singer W (1977) The role of visual cortex for binocular interactions in the cat lateral geniculate nucleus. Brain Res 120: 354–361
Sillito AM, Kemp JA (1983) The influence of GABAergic inhibitory processes on the receptive field structure of X and Y cells in cat dorsal lateral geniculate nucleus (dLGN). Brain Res 277: 63–77
Sillito AM, Kemp JA, Berardi N (1983) The cholinergic influence on the function of the cat dorsal lateral geniculate nucleus (dLGN). Brain Res 280: 299–307
Singer W (1973) The effect of mesencephalic reticular stimulation on intracellular potentials of cat lateral geniculate neurones. Brain Res 61: 35–54
Singer W (1977) Control of thalamic transmission by cortifugal and ascending reticular pathways in the visual system. Physiol Reviews 57: 386–420
Singer W, Bedworth N (1973) Inhibitory interaction between X and Y units in the cat lateral geniculate nucleus. Brain Res 49: 291–307
Tsumoto T, Creutzfeldt OD, Legendy CR (1978) Functional organization of the corticofugal system from visual cortex to lateral geniculate nucleus in the cat (with an appendix on geniculo-cortical mono-synaptic connections). Exp Brain Res 32: 345–364
Wróbel A (1982) Inhibitory mechanisms within the receptive fields of the lateral geniculate body of the cat. Acta Neurobiol Exp 42: 93–106
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Ahlsén, G., Lindström, S. & Lo, FS. Interaction between inhibitory pathways to principal cells in the lateral geniculate nucleus of the cat. Exp Brain Res 58, 134–143 (1985). https://doi.org/10.1007/BF00238961
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00238961