Summary
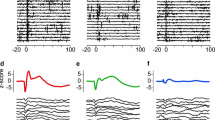
The temporo-parietal association cortex around the caudal end of the Sylvian fissure was studied with the single cell recording technique in three awake behaving Macaca speciosa-monkeys. Of the 197 cells isolated, 5% were active only during the monkey's own movements, mostly during head rotation, and 95% were responsive to sensory stimulation: 54% to auditory stimuli, 24% to somatosensory stimuli, 13% to both of these and 4% to visual stimuli. Some cells, classified as responsive to somatosensory stimuli, were activated only by passive rotation of the head on the cervical axis; it is possible that they were driven by vestibular stimuli. Half of the cells were activated by stimuli on both sides of the monkey, and almost all the rest, only by stimuli on the side contralateral to the hemisphere recorded.
Of the acoustically drivable cells, 95% responded to natural sounds, such as, rubbing hands together, rustle of clothes, clicks or jingles (sounds with noise spectrum and rapid intensity transitions). Most of these neurons were also examined with pure tones of 0.2–20 kHz: various inhibitory or excitatory responses were elicited in half of them, usually over a wide range of frequencies. The responses of most acoustically drivable cells (62%) depended on the location of the sound source with reference to the monkey's head so that the maximal response was elicited by sounds with a certain angle of incidence, usually on the contralateral side.
The present results suggest that the area studied participates in the analysis of the temporal pattern of a sound, the location of the sound source and in spatial control of head movements.
Similar content being viewed by others
References
Beaton R, Miller JM (1975) Single cell activity in the auditory cortex of the unanesthetized, behaving monkey: correlation with stimulus controlled behavior. Brain Res 100: 543–562
Bechterew W von (1911) Die Funktionen der Nervencentra. G. Fischer, Jena (Band 3, p 1861)
Benevento LA, Fallon J, Davis BJ, Rezak M (1977) Auditory-visual interaction in single cells in the cortex of the superior temporal sulcus and the orbital frontal cortex of the macaque monkey. Exp Neurol 57: 849–872
Bertrand M (1969) The behavioral repertoire of the stumptail macaque. Karger, Basel, pp 232–254
Bond HW, Ho P (1970) Solid miniature silver-silver chloride electrodes for chronic implantation. Electroencephalogr Clin Neurophysiol 28: 206–208
Brugge JF, Merzenich MM (1973) Responses of neurons in auditory cortex of the macaque monkey to monaural and binaural stimulation. J Neurophysiol 36: 1138–1158
Burton H, Jones EG (1976) The posterior thalamic region and its cortical projection in new world and old world monkeys. J Comp Neurol 168: 249–302
Chavis DA, Pandya DN (1976) Further observations on corticofrontal connections in the rhesus monkey. Brain Res 117: 369–386
Evarts EV (1966) Methods for recording activity of individual neurons in moving animals. In: Rushmer RF (ed) Methods in medical research. Year Book Medical Publisher, Chicago, pp 241–250
Galaburda AM, Sanides F, Geschwind N (1978) Human brain. Cytoarchitectonic left-right asymmetries in the temporal speech region. Arch Neurol 35: 812–817
Heilman KM, Pandya DN, Karol EA, Geschwind N (1971) Auditory inattention. Arch Neurol 24: 323–325
Heilman KM, Valenstein E (1972) Auditory neglect in man. Arch Neurol 26: 32–35
Hocherman S, Benson DA, Goldstein MH Jr, Heffner HE, Hienz RD (1976) Evoked unit activity in auditory cortex of monkeys performing a selective attention task. Brain Res 117: 51–68
Hyvärinen J, Poranen A (1974) Function of the parietal associative area 7 as revealed from cellular discharges in alert monkeys. Brain 97: 673–692
Hyvärinen J (1977) Function of parietal associative area 7 (abstract). Proceedings of XXVIIth International Congress of Physiological Sciences (Paris) 12: 600
Imig TJ, Ruggero MA, Kitzes LM, Javel E, Brugge JF (1977) Organization of auditory cortex in the owl monkey (Aortus trivigatus). J Comp Neurol 171: 111–128
Jones EG, Powell TPS (1970) Connexions of the somatic sensory cortex of the rhesus monkey. III. Thalamic connexions. Brain 93: 37–56
Le Gros Clark WE (1936) The thalamic connections of the temporal lobe of the brain in the monkey. Anatomy 70: 447–464
Leinonen L, Hyvärinen J (1978) Functional differentiation within the parietal association cortex of the monkey (abstract). Neurosci Lett [Suppl] 1: 391
Leinonen L, Hyvärinen J, Nyman G, Linnankoski I (1979) Functional properties of neurons in lateral part of associative area 7 in awake monkeys. Exp Brain Res 34: 299–320
Leinonen L, Nyman G (1979) Functional properties of cells in anterolateral part of area 7, associative face area of awake monkeys. Exp Brain Res 34: 321–333
Leinonen L, Hyvärinen J (1980) (in press) Parietal association cortex of the monkey as revealed by cellular recordings. In: Stelmach GE (ed) Tutorials in motor behaviour. North Holland, Amsterdam
Leinonen L (1980) (in press) Functional properties of neurones in the posterior part of area 7 in awake monkey. Acta Physiol Scand
Lilly JC (1958) Correlations between neurophysiological activity in the cortex and short-term behavior in the monkey. In: Harlow HF, Woolsey CN (eds) Biological and biochemical bases of behavior. The University of Wisconsin Press, Madison, pp 83–100
Luria AR (1976) Basic problems of neurolinguistics. Mouton, The Hague, pp 106–114
Merzenich MM, Brugge JF (1973) Representation of the cochlear partition on the superior temporal plane of the macaque monkey. Brain Res 50: 275–296
Miller JM (1971) Single unit discharges in behaving monkeys. In: Sachs MB (ed) Physiology of the auditory system. National Educational Consultants, Baltimore, pp 317–326
Mountcastle VB, Lynch JC, Georgopoulos A, Sakata H, Acuna C (1975) Posterior parietal association cortex of the monkey: command functions for operations within extrapersonal space. J Neurophysiol 38: 871–908
Newman JD, Lindsley DF (1976) Single unit analysis of auditory processing in squirrel monkey frontal cortex. Exp Brain Res 25: 169–181
Nyysönen J, Sovijärvi RA (1975) A method for simulating sound source movement suitable for neurophysiological studies. In: Uusitalo A, Saranummi N (eds) Proceedings of III nordic meeting on medical and biological engineering. Lyriiri Oy, Helsinki, pp 69.1–69.3
Pandya DN, Hallett M, Mukherjee SK (1969) Intra- and interhemispheric connections of the neocortical auditory system in the rhesus monkey. Brain Res 14: 49–65
Pandya DN, Karol EA, Heilbronn D (1971) The topographical distribution of interhemispheric projections in the corpus callosum of the rhesus monkey. Brain Res 32: 31–43
Pandya DN, Sanides F (1973) Architectonic parcellation of the temporal operculum in rhesus monkey and its projection pattern. Z Anat Entwickl Gesch 139: 127–161
Robinson DL, Goldberg ME (1978) Sensory and behavioral properties of neurons in posterior parietal cortex of the awake, trained monkey. Fed Proc 37: 2258–2261
Robinson DL, Goldberg ME, Stanton GB (1978) Parietal association cortex in the primate: sensory mechanisms and behavioral modulations. J Neurophysiol 41: 910–932
Sanchez Longo LP, Forster FM, Auth TL (1957) A clinical test for sound localization and its applications. Neurology 7: 655–663
Seltzer B, Pandya DN (1978) Afferent cortical connections and architectonics of the superior temporal sulcus and surrounding cortex in the rhesus monkey. Brain Res 149: 1–24
Sovijärvi ARA, Hyvärinen J (1974) Auditory cortical neurons in the cat sensitive to the direction of sound source movement. Brain Res 73: 455–471
Vogt C, Vogt O (1919) Allgemeinere Ergebnisse unserer Hirnforschung. J Psychol Neurol (Lpz) 25: 279–462
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Leinonen, L., Hyvärinen, J. & Sovijärvi, A.R.A. Functional properties of neurons in the temporo-parietal association cortex of awake monkey. Exp Brain Res 39, 203–215 (1980). https://doi.org/10.1007/BF00237551
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00237551