Summary
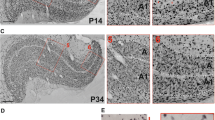
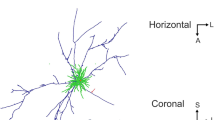
Intracortical injections of horseradish peroxidase conjugated with wheat-germ agglutinin (WGA-HRP) reveal a characteristic patchy staining pattern within the superficial layers of cat striate cortex. The patches consist of a dense accumulation of labeled neurons and axonal arborizations. We have investigated the tangential organization and the development of these intrinsic cortical connections by using a flat-mount preparation of area 17. The diameter of the patches varied from 200 to 400 μm, the center-to-center distance ranged from 400 to 800 μm, and the spread of patches extended further in the anterior-posterior than in the medial-lateral direction. The expression of these horizontal patchy connections is age- and experience-dependent. From ten days to six weeks of age patches are exuberant and on occasion fuse to beaded bands extending radially from the injection site. From 6 weeks onwards the number and the tangential spread of the patches decreases to one or two rows of isolated clusters. Long-term binocular deprivation disrupts this pattern of intrinsic connections nearly completely. We infer from these results that there is an inborn pattern of discrete horizontal connections in striate cortex which is shaped by visual experience and requires contour vision for its maintenance.
Similar content being viewed by others
References
Albus K, Wolf W (1984) Early post-natal development of neuronal function in the kitten's visual cortex: a laminar analysis. J Physiol 348: 153–185
Blakemore C, van Sluyters RC (1975) Innate and environmental factors in the development of the kitten's visual cortex. J Physiol 248: 663–716
Fawcett JW, O'Leary DDM (1985) The role of electrical activity in the formation of topographic maps in the nervous system. TINS 5: 201–206
Gilbert CD, Wiesel TN (1979) Morphology and intracortical projections of functionally characterized neurons in the cat visual cortex. Nature 280: 120–125
Hubel DH, Wiesel TN (1962) Receptive fields, binocular interaction and functional architecture in the cat's visual cortex. J Physiol 160: 106–154
Hubel DH, Wiesel TN (1968) Receptive fields and functional architecture of monkey striate cortex. J Physiol 195: 215–243
Hubel DH, Wiesel TN (1970) The period of susceptibility to the physiological effects of unilateral eye closure in kittens. J Physiol 206: 419–436
Innocenti GM, Frost DO (1980) The postnatal development of visual callosal connections in the absence of visual experience or of the eyes. Exp Brain Res 39: 365–375
Innocenti GM, Frost DO, Illes J (1985) Maturation of visual callosal connections in visually deprived kittens: a challenging critical period. J Neurosci 5: 255–267
Kaas JH, Cusick CG (1984) Instrinsic and extrinsic cortical connections of area 17 of the prosimian primate Galago. Soc Neurosci Abstr 10: 729
Katz LC, Burkhalter A, Dreyer WJ (1984) Fluorescent latex microspheres as a retrograde neuronal marker for in vivo and in vitro studies of visual cortex. Nature 310: 498–500
LeVay S, Stryker MP (1979) The development of ocular dominance columns in the cat. Soc Neurosci Symp 4: 83–98
LeVay S, Stryker MP, Shatz CJ (1978) Ocular dominance columns and their development in layer IV of the cat's visual cortex: a quantitative study. J Comp Neurol 179: 223–244
Livingstone MS, Hubel DH (1984) Specificity of intrinsic connections in primate primary visual cortex. J Neurosci 4: 2830–2835
Luhmann HJ, Martínez Millán L, Singer W (1985) Organization of intrinsic connections in cat striate cortex. Neurosci Lett Suppl 22: 442
Martin KAC, Whitteridge D (1984) Form, function and intracortical projections of spiny neurones in the striate visual cortex of the cat. J Physiol 353: 463–504
Matsubara J, Cynader M, Swindale NV, Stryker MP (1985) Intrinsic projections within visual cortex: evidence for orientation-specific local connections. Proc Natl Acad Sci 82: 935–939
Mesulam MM (1978) Tetramethyl benzidine for horseradish peroxidase neurohistochemistry: a non-carcinogenic blue reaction-product with superior sensitivity for visualizing neural afferents and efferents. J Histochem Cytochem 26: 106–117
Mitchison G, Crick F (1982) Long axons within the striate cortex: their distribution, orientation, and patterns of connection. Proc Natl Acad Sci 79: 3661–3665
Mountcastle VB (1957) Modality and topographic properties of single neurons of cat's somatic sensory cortex. J Neurophysiol 20: 408–434
Price DJ (1986) The postnatal development of clustered intrinsic connections in area 18 of the visual cortex in kittens. Dev Brain Res 24: 31–38
Price DJ, Blakemore C (1985) The postnatal development of the association projection from visual cortical area 17 to area 18 in the cat. J Neurosci 5: 2443–2452
Rockland KS (1985a) A reticular pattern of intrinsic connections in primate area V2 (area 18). J Comp Neurol 235: 467–478
Rockland KS (1985b) Anatomical organization of primary visual cortex (area 17) in the Ferret. J Comp Neurol 241: 225–236
Rockland KS, Lund JS (1982) Widespread periodic intrinsic connections in the tree shrew visual cortex. Science 215: 1532–1534
Rockland KS, Lund JS (1983) Intrinsic laminar lattice connections in primate visual cortex. J Comp Neurol 216: 303–318
Rockland KS, Lund JS, Humphrey AL (1982) Anatomical banding of intrinsic connections in striate cortex of tree shrews (Tupaia glis). J Comp Neurol 209: 41–58
Schmidt JT (1985) Formation of retinotopic connections: selective stabilization by an activity-dependent mechanism. Cell Molec Neurobiol 5: 65–84
Swindale NV (1982) The development of columnar systems in the mammalian visual cortex. The role of innate and environmental factors. TINS 5: 235–240
Szentágothai J (1975) The “module-concept” in cerebral cortex architecture. Brain Res 95: 475–496
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Luhmann, H.J., Millán, L.M. & Singer, W. Development of horizontal intrinsic connections in cat striate cortex. Exp Brain Res 63, 443–448 (1986). https://doi.org/10.1007/BF00236865
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00236865