Summary
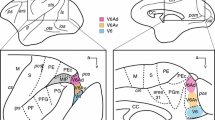
The anterograde transport of lectin-conjugated horseradish peroxidase (WGA-HRP) was here employed in order to visualize crossed cortico-thalamic efferents of the motor cortex in rats and cats. After WGA-HRP cortical injections in the rat retrogradely labeled cells were observed in the ipsilateral thalamus, and heavy anterograde labeling was observed both in the ipsi-and contralateral thalamus. The contralateral anterograde labeling was less intense than the ipsilateral one and it was distributed in the anterior intralaminar structures, in the parafascicular nucleus, in the ventromedial, ventrolateral and ventrobasal nuclei and in the posterior complex, symmetrically to the labeling observed on the ipsilateral side. Further experiments were made in the rat in order to ascertain that the bilateral anterograde labeling in the thalamus derived unilaterally from the cortex. To this purpose, kainic acid was injected unilaterally either into the frontal cortex or into the thalamus, and WGA-HRP was later injected on the same side in the frontal cortex. Moreover, WGA-HRP was injected into the frontal cortex after splitting of the corpus callosum. The results obtained in these experiments confirmed that cortical neurons projected bilaterally upon the thalamus. Further, these experiments indicated that at least the majority of the contralateral fronto-thalamic fibers crossed the midline in the thalamic massa intermedia. WGA-HRP injections into the pericruciate cortex in the cat confirmed the presence of anterogradely labeled terminals in the contralateral anterior and posterior intralaminar, ventral anterior, ventromedial and ventrolateral nuclei. The labeling was in all cases heavier in the intralaminar nuclei than in the other structures, but it was less intense than that observed in the rat. The labeling in the contralateral principal nuclei was heavier in one case in which the injection area diffused into the proreal cortex than in the other cats. Thus, the present results indicate that frontal efferents terminate more densely in the rat's than in the cat's contralateral thalamus. In the latter species frontal fibers are mainly distributed upon the contralateral intralaminar nuclei, whereas in the rat frontal efferents are also heavily distributed upon the ventral nuclear complex.
Similar content being viewed by others
References
Beckstead RM (1979) An autoradiographic examination of cortico-cortical and subcortical projections of the mediodorsal-projection (prefrontal) cortex in the rat. J Comp Neurol 184: 43–62
Berman N, Payne BR (1982) Contralateral corticofugal projection from the lateral, suprasylvian and ectosylvian gyri in the cat. Exp Brain Res 47: 234–238
Cajal S Ramón Y (1911) Histologie du système nerveux de l'homme et des vertébrés. Maloine, Paris
Carreras M, Culzoni V, Eager P, Gueli P (1969) Cortico-cortical and cortico-thalamic connexions of somatic sensory area III in the cat. Anat Rec 163: 298
De Vito JL (1969) Projections from the cerebral cortex to intralaminar nuclei in monkey. J Comp Neurol 136: 193–202
Gerfen CR, O'Leary DDM, Cowan WM (1982) A note on the transneuronal transport of wheat germ agglutinin-conjugated horseradish peroxidase in the avian and rodent visual systems. Exp Brain Res 48: 443–448
Goldman PS (1978) Neuronal plasticity in primate telencephalon: anomalous projections induced by prenatal removal of frontal cortex. Science 202: 768–770
Goldman PS (1979) Contralateral projections to the dorsal thalamus from frontal association cortex in the rhesus monkey. Brain Res 166: 166–171
Itaya SK, Van Hoesen GW (1982) WGA-HRP as a transneuronal marker in the visual pathways of monkey and rat. Brain Res 236: 199–204
Jones EG (1981) Functional subdivision and synaptic organization of the mammalian thalamus. Int Rev Physiol 25: 173–245
Jones EG (1983) The thalamus. In: Emson PC (ed) Clinical neuroanatomy. Raven Press, New York, pp 257–293
Jones EG (1984) Laminar distribution of cortical efferents cells. In: Peters A, Jones EG (eds) Cellular components of the cerebral cortex. I. Plenum Press, Amsterdam, pp 521–553
Kaitz SS, Robertson RT (1981) Thalamic connections with limbic cortex. II. Cortico-thalamic projections. J Comp Neurol 195: 527–545
Kawana E, Kusama T (1964) Projection of the sensory motor cortex to the thalamus, dorsal column nucleus, the trigeminal nucleus and the spinal cord in the cat. Folia Psychiat Neurol Jpn 18: 337–380
Künzle H (1976) Thalamic projections from the precentral motor cortex in Macaca fascicularis. Brain Res 105: 253–267
Künzle H (1978) An autoradiographic analysis of the efferent connections from premotor and adjacent prefrontal regions (areas 6 and 9) in Macaca fascicularis. Brain Behav Evol 15: 185–234
Leonard CM (1969) The prefrontal cortex of the rat. I. Cortical projection of mediodorsal nucleus. II. Efferent connections. Brain Res 12: 321–343
Macchi G, Rinvik E (1977) Thalamo-telencephalic circuits: A neuroanatomical survey. In: Remond A, Creutzfeldt O (eds) Handbook of electroencephalography and clinical neurophysiology, Vol 2. Elsevier, Amsterdam, pp 96–113
Mesulam MM (1978) Tetramethyl benzidine for horseradish peroxidase neurohistochemistry: A non-carcinogenic blue reaction-product with superior sensitivity for visualizing neural afferents and efferents. J Histochem Cytochem 26: 106–117
Neuman S, Pritzel M, Huston JP (1982) Plasticity of corticothalamic projections and functional recovery in the unilateral detelencephalized infant rat. Behav Brain Res 4: 377–388
Niimi K, Kishi S, Miki M, Fujita S (1963) An experimental study of the course and termination of the projection fibers from cortical areas 4 and 6 in the cat. Folia Psychiat Neurol Jpn 17: 167–213
Payne BR, Berman N (1984) Contralateral corticofugal projections to cat visual thalamus. Exp Brain Res 53: 462–466
Pritzel M, Huston JP (1981) Unilateral ablation of telencephalon induces appearance of contralateral cortical subcortical projections to thalamic nuclei. Behav Brain Res 3: 43–54
Reep RL, Winans SS (1982) Efferent connections of dorsal and ventral agranular insular cortex in the hamster Mesocricetus Auratus. Neuroscience 7: 2609–2635
Rinvik E (1968a) The corticothalamic projection from the gyrus proreus and the medial wall of the rostral hemisphere in the cat. An experimental study with silver impregnation methods. Exp Brain Res 5: 129–152
Rinvik E (1968b) The corticothalamic projection from the pericruciate and coronal gyri in the cat. An experimental study with silver-impregnation methods. Brain Res 10: 79–119
Rinvik E (1984) Thalamic commissural connections in the cat. Neurosci Lett 44: 311–316
Schnyder H, Künzle H (1983) Differential labeling in neuronal tracing with wheat germ agglutinin. Neurosci Lett 35: 115–120
Sotnichencko TS (1976) Convergence of descending pathways of motor, visual and limbic cortex in the cat di- and mesencephalon. Brain Res 116: 401–415
Trojanowski JQ (1983) Time of arrival of wheat germ agglutinin-HRP conjugates in superior colliculus after intraocular injections in the rat. Brain Res 267: 365–370
Trojanowski JQ, Gonatas JO, Gonatas NK (1981) Conjugates of horseradish peroxidase (HRP) with cholera toxin and wheat germ agglutinin are superior to free HRP as orthogradely transported markers. Brain Res 223: 381–385
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Molinari, M., Minciacchi, D., Bentivoglio, M. et al. Efferent fibers from the motor cortex terminate bilaterally in the thalamus of rats and cats. Exp Brain Res 57, 305–312 (1985). https://doi.org/10.1007/BF00236536
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00236536