Summary
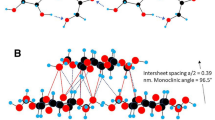
Mineralized pieces of tendons from the tibio-tarsus of turkeys were (i) shock-frozen, freeze-dried, embedded and cut without staining, or (ii) fixed, embedded and stained after sectioning. Micrographs were taken with an electron microscope on longitudinally cut sections. The center-to-center distances of neighboring apatitic needles within collagen fibrils were measured. For shock-frozen and freeze-dried specimens, the average of these distances is 4.7 nm and the most frequent value 4.2 nm; for fixed and stained specimens, 3.8 nm and 3.6 nm, respectively. Laser diffraction of the electron micrographs showed a dumbbell-like intensity pattern (two diffuse maxima of intensity on the equator, one on each side of the central spot), giving an average distance of about 6 nm. This value represents the upper range of the direct measurements. The measurements demonstrate that the arrangement of the collagen microfibrils is mainly preserved during mineralization. However, using laser diffraction, distances of 9–11 nm were also observed. Such large distances can also be demonstrated by X-ray diffraction on collagen fibrils stained under special conditions. This may indicate that special conditions of apatitic mineralization or staining may alter the arrangement of the microfibrils.
Similar content being viewed by others
References
Althoff, J., Quint, P., Höhling, H.J.: Activation and specificity of alkaline phosphatase of a mineralizing collagen rich system. Experientia 34, 692–693 (1978)
Bond, P.J., Hosemann, R., Newesely, H.: Collagen mineralization. J. Dent. Res. 58(B), 991 (1979)
Bouteille, M., Pease, D.C.: The three dimensional structure of native collageneous fibrils, their proteinaceous filaments. J. Ultrastruct. Res. 35, 314–338 (1971)
Boyde, A.: Transmission electron microscopy of ion beam thinned dentine. Cell Tissue Res. 152, 543–550 (1974)
Brodsky, B., Hukins, W.L., Hulmes, D.J.S., Miller, A., White, S., Woodhead-Galloway, J.: Low angle X-ray diffraction studies on stained rat tail tendon. Biochim. Biophys. Acta 539, 25–32 (1978)
Eanes, E.D., Lundy, D.R., Martin, G.N.: X-ray diffraction study of the mineralization of turkey leg tendon. Calcif. Tiss. Res. 6, 239–248 (1970)
Höhling, H.J., Dahmen, G.: Sublichtmikroskopische Untersuchungen an gesunden und degenerierten Sehnen. Z. Orthop. 97, 339–353 (1963)
Höhling, H.J., Kreilos, R., Neubauer, G., Boyde, A.: Electron microscopy and electron microscopical measurements of collagen mineralization in hard tissues. Z. Zellforsch. 122, 36–52 (1971)
Höhling, H.J., Ashton, B.A., Köster, H.D.: Quantitative electron microscopic investigations of mineral nucleation in collagen. Cell Tissue Res. 148, 11–26 (1974)
Höhling, H.J., Steffens, H., Stamm, G., Mays, U.: Transmission microscopy of freeze dried, unstained epiphyseal cartilage of the guinea pig. Cell Tissue Res. 167, 243–263 (1976)
Katz, E.P., Li, S.T.: Structure and function of bone collagen fibrils. J. Mol. Biol. 80, 1–15 (1974)
Krefting, E.R., Barckhaus, R., Höhling, H.J., Bond, P.J., Hosemann, R.: Structural similarities between naturally mineralized collagen in turkey tibia tendon and heavy-metal-stained rat tail collagen. Calcif. Tiss. Res. Suppl. to Vol. 24, R13 (1977)
Kushida, H.: A styrene-methacrylate resin embedding method for ultrathin sectioning. J. Electronmicroscopy 10, 16–19 (1961)
Miller, A., Parry, D.A.D.: The structure and packing of microfibrils in collagen. J. Mol. Biol. 75, 441–447 (1973)
Miller, A., Wray, J.S.: Molecular packing in collagen. Nature (London) 230, 437–439 (1971)
Nemetschek, Th., Hosemann, R.: A kink model of native collagen. Kolloid. Z. u. Z. Polymere 251, 1044–1056 (1973)
Olsen, B.R.: Electron microscope studies on collagen. I. Native collagen fibrils. Z. Zellforsch. 59, 199–213 (1963)
Quint, P., Althoff, J., Höhling, H.J.: Topochemical analysis of a mineralizing collagen-rich system. Naturwiss. 64, 389 (1977)
Reimer, L., Badde, H.-G., Drewes, E., Gilde, H., Kappert, H., Höhling, H.J., v. Bassewitz, D.B., Rössner, A.: Laserbeugung an elektronenmikroskopischen Aufnahmen — Forschungsbericht des Landes NRW, Nr. 2314, Opladen: Westdeutscher Verlag 1973
Spurr, A.R.: A low viscosity epoxy resin embedding medium for electron microscopy. J. Ultrastruct. Res. 26, 31–43 (1969)
Tromans, W.J., Horne, R.W., Gresham, G.A., Baily, A.J.: electron microscope studies on the structure of collagen fibres by “negative staining”. Z. Zellforsch. 58, 798–802 (1963)
Umrath, W.: Cooling bath for rapid freezing in electron microscopy. J. Micr. 101, 103–105 (1974)
Woodhead-Galloway, J., Hukins, D.W.L., Wray, J.S.: Closest packing of two-stranded coiled-coils as a model for the collagen fibril. Biochem. Biophys. Res. Commun. 64, 1237–1244 (1975)
Author information
Authors and Affiliations
Additional information
The authors thank the Deutsche Forschungsgemeinschaft for financial support
Rights and permissions
About this article
Cite this article
Krefting, E.R., Barckhaus, R.H., Höhling, H.J. et al. Analysis of the crystal arrangement in collagen fibrils of mineralizing turkey tibia tendon. Cell Tissue Res. 205, 485–492 (1980). https://doi.org/10.1007/BF00232288
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00232288