Summary
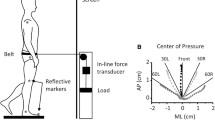
This study examined whether the torques and EMG activity that precede and accompany bilateral arm pulls made by standing humans demonstrate a pulse height form of organization. Nine adults made abrupt bilateral pulls in the sagittal plane against a handle, to force targets equal to 5, 10, 20, 40, 60, 80 and 95% of their maximal pulling force (%MPF). The force applied at the handle, ground reaction forces, the center of pressure (CP), and EMG activity in gastrocnemius (GS), biceps femoris (BF), tibialis anterior (TA) and quadriceps (QD) muscles were recorded. Our analysis divided the action into a pre-pull phase (events prior to the increase of handle force) and a pulling phase (while handle force was greater than zero). We evaluated the effects of %MPF on the durations and peak amplitudes of the pre-pull and pulling angular impulses about the ankle joint and on pre-pull EMG patterns. The results showed that the angular impulse associated with the pulling torque (due to the reactive force on the body during the pull) had a pulse height organization: peak torque increased linearly with %MPF, and the durations of the pulling torque were relatively constant. In contrast, a pulse height organization did not characterize the pre-pull period for either the angular impulse associated with ankle torque (due to net ground reaction force) or EMG activity in the leg muscles. Rather, peak ankle torque typically increased up to some submaximal %MPF and then plateaued, perhaps due to a constraining effect of foot length on CP. The durations of pre-pull ankle torques increased over the whole range of %MPF, thereby compensating for the limit on ankle torque. Depending on the subject, the muscles were recruited in two different orders: GS-BF-TA-QD, or GS-TA-BF-QD. As the %MPF increased, the EMG onset times of all four muscles occurred earlier, and there was a greater likelihood that the BF, TA and QD muscles would be recruited on a given trial. The changes in the ankle torque and EMG patterns were gradual, suggesting that the pre-pull phase could have one underlying form of organization, with parameters that are tuned to task goals and anatomical constraints.
Similar content being viewed by others
References
Brown JE, Frank JS (1987) Influence of event anticipation on postural actions accompanying voluntary. Exp Brain Res 67:645–650
Cordo PJ, Nashner LM (1982) Properties of postural adjustments associated with rapid arm movement. J Neurophysiol 47:187–302
Crenna P, Frigo C, Massion J, Pedotti A (1987) Forward and backward axial synergies in man. Exp Brain Res 65:538–548
Dempster WT (1958) Analysis of two-handed pulls using free body diagrams. J Appl Physiol 13:469–480
Freund HJ, Budingen HJ (1978) The relationship between speed and amplitude of the fastest voluntary contractions of human arm muscles. Exp Brain Res 31:1–12
Friedli WG, Hallett M, Simon SR (1984) Postural adjustments associated with rapid voluntary arm movements. 1. Electromyographic data. J Neurol Neurosurg Psychiat 47:611–622
Gaughran GR, Dempster WT (1956) Force analyses of horizontal two-handed pushes and pulls in the sagittal plane. Hum Biol 28:67–92
Ghez C, Gordon J (1987) Trajectory control in targeted force impulses. I. Role of opposing muscles. Exp Brain Res 67:225–240
Gordon J, Ghez C (1987) Trajectory control in targeted force impulses. II. Pulse height control. Exp Brain Res 67:241–252
Gottlieb GL, Corcos DM, Agarwal GC (1989) Strategies for control of single degree-of-freedom voluntary movements. Behav Brain Sci 12:189–250
Hardcastle WJ (1981) Experimental studies in lingual coarticulation. In: Archer RE, Henderson EJA (eds) Towards a history of phonetics. Edinburgh University Press, Edinburgh, Scotland, pp 50–66
Hening W, Vicario D, Ghez C (1988) Trajectory control in targeted force impulses. IV. Influence of choice, prior experience and urgency. Exp Brain Res 71:103–115
Horak FB, Nashner LM (1986) Central programming of postural movements: adaptation to altered support-surface configurations. J Neurophysiol 55:1369–1381
Hubbard AW, Seng CN (1954) Visual movements of batters. Res Quar 25:42–57
Jeannerod M (1984) The timing of natural prehension movements. J Motor Behav 26:235–254
Lee WA (1984) Neuromotor synergies as a basis for coordinated intentional action. J Motor Behav 16:135–170
Lee WA, Deming L (1988) Correlations between age and the size of the normalized support base during quiet standing. Phys Ther 68:259
Lee WA, Michaels CM, Pai YC (1989) Changes in joint posture and center of mass location during abrupt pulls of different forces made by standing humans. Neuroscience Abstr 15:1202
Lee WA, Rogers MW (1987) Mechanical and cognitive constraints on standing influence postural adjustments and maximal forces during pulling. Soc Neuroscience Abstr 13:347
McCollum G, Horak F, Nashner L (1985) Parsimony in neural calculations of postural movements. In: Bloedel J, Dichgans J, Precht W (eds) Cerebellar functions. Springer, Berlin Heidelberg New York Tokyo, pp 52–66
Nashner LM, Cordo PJ (1981) Relation of automatic postural responses and reaction-time voluntary movements of human leg muscles. Exp Brain Res 43:395–405
Woollacott MH, Bonnet M, Yabe K (1984) Preparatory process for anticipatory postural adjustments: modulation of leg muscles reflex pathways during preparation for arm movements in standing man. Exp Brain Res 55:263–271
Zajac FE, Gordon ME (1989) Determining muscle's force and action in multiarticular movement. In: Pandolf K (ed) Exercise and sport science reviews, 17. Williams & Wilkins, Baltimore, pp 187–230
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Lee, W.A., Michaels, C.F. & Pai, Y.C. The organization of torque and EMG activity during bilateral handle pulls by standing humans. Exp Brain Res 82, 304–314 (1990). https://doi.org/10.1007/BF00231250
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00231250