Summary
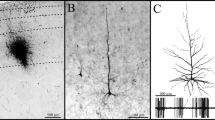
During the course of an in vivo intracellular labeling study, a chandelier (axo-axonic) cell was completely filled with biocytin in the CA1 region of the hippocampus. Chandelier cells are known to provide GABAergic terminals exclusively to the axon initial segment of pyramidal cells. The lateral extent and laminar distribution of the dendritic arborization of the chandelier cell was very similar to that of pyramidal cells; the numerous basal and apical dendrites reached the ventricular surface and the hippocampal fissure, respectively. The dendrites, however, had very few spines. The neuron had an asymmetric axonal arbor occupying an elliptical area of 600 by 850 μm in the pyramidal cell layer and stratum oriens, with over three-quarters of the axon projecting to the fimbrial side of the neuron. Counting all clusters of terminals, representing individually innervated axon initial segments, the chandelier cell was estimated to contact 1214 pyramidal cells, a number that exceeds previous estimations, based on Golgi studies, by several-fold. The findings support the view that chandelier cells may control the threshold and/or synchronize large populations of principal cells.
Similar content being viewed by others
References
Andersen P (1975) Organization of hippocampal neurons and their interconnections. In: Isaacson RL, Pribram KH (eds) The hippocampus, Vol 1. Plenum Press, New York, pp 155–175
Boss BD, Turlejski K, Stanfield BB, Cowan WM (1987) On the numbers of neurons in fields CA1 and CA3 of the hippocampus of Sprague-Dawley and Wistar rats. Brain Res 406: 280–287
Buzsáki G (1984) Feed-forward inhibition in the hippocampal formation. Prog Neurobiol 22: 131–153
Cassell MD (1980) The number of cells in the stratum pyramidale of the rat and human hippocampal formation. PhD Thesis, University of Bristol, UK
Christian EP, Dudek FE (1988) Electrophysiological evidence from glutamate microapplications for local excitatory circuits in the CA1 area of rat hippocampal slices. J Neurophysiol 59: 110–123
Doller HJ, Weight FF (1982) Perforant pathway activation of hippocampal CA1 stratum pyramidale neurons: electrophysiological evidence for a direct pathway. Brain Res 237: 1–13
Doller HJ, Weight FF (1985) Perforant pathway-evoked long-term potentiation of CA1 neurons in the hippocampal slice preparation. Brain Res 333: 305–310
Douglas RJ, Martin KAC (1990) Control of neuronal output by inhibition at the axon initial segment. Neural Comp 2: 283–292
Freund TF, Martin KAC, Smith AD, Somogyi P (1983) Glutamate decarboxylase-immunoreactive terminals of Golgi-impregnated axo-axonic cells and of presumed basket cells in synaptic contact with pyramidal neurons of the cat's visual cortex. J Comp Neurol 221: 263–278
Freund TF, Antal M (1988) GABA-containing neurons in the septum control inhibitory interneurons in the hippocampus. Nature 336: 170–173
Horikawa K, Armstrong WE (1988) A versatile means of intracellular labeling: injection of biocytin and its detection with avidin conjugates. J Neurosci Meth 25: 1–11
Kawaguchi Y, Hama K (1987) Two subtypes of non-pyramidal cells in rat hippocampal formation identified by intracellular recording and HRP injection. Brain Res 411: 190–195
Kawaguchi Y, Wilson CJ, Emson PC (1989) Intracellular recording of identified neostriatal patch and matrix spiny cells in a slice preparation preserving cortical inputs. J Neurophysiol 62: 1052–1068
Knowles WD, Schwartzkroin PA (1981) Axonal ramifications of hippocampal CA1 pyramidal cells. J Neurosci 1: 1236–1241
Kosaka T (1980) The axon initial segment as a synaptic site: ultrastructure and synaptology of the initial segment of the pyramidal cell in the rat hippocampus (CA3 region). J Neurocytol 9: 861–882
Kosaka T (1983) Axon initial segments of the granule cells in the rat dentate gyrus: synaptic contacts on bundles of axon initial segments. Brain Res 274: 129–134
Lorente de Nó R (1934) Studies on the structure of the cerebral cortex: II. Continuation of the study of the ammonic system. J Psychol Neurol 46: 113–177
Lytton WW, Sejnowski TJ (1991) Simulations of cortical pyramidal neurons synchronized by inhibitory interneurons. J Neurophysiol 66: 1059–1079
Ramón y Cajal S (1911) Histologie du systéme nerveux de l'homme et des vertebrés. Maloine, Paris.
Ribak CE, Vaughn JE, Barber RP (1981) Immunocytochemical localization of GABAergic neurones at the electron microscopical level. J Histochem 13: 555–582
Somogyi P (1977) A specific ‘axo-axonal’ interneuron in the visual cortex of the rat. Brain Res 136: 345–350
Somogyi P, Freund TF, Cowey A (1982) The axo-axonic interneuron in the cerebral cortex of the rat, cat and monkey. Neuroscience 7: 2577–2607
Somogyi P, Nunzi MG, Gorio A, Smith AD (1983) A new type of specific interneuron in the monkey hippocampus forming synapses exclusively with the axon initial segments of pyramidal cells. Brain Res 259: 137–142
Somogyi P, Freund TF, Hodgson AJ, Somogyi J, Beroukas D, Chubb IW (1985) Identified axo-axonic cells are immunoreactive for GABA in the hippocampus and visual cortex of the cat. Brain Res 332: 143–149
Soriano E, Frotscher M (1989) A GABAergic axo-axonic cell in the fascia dentata controls the main excitatory hippocampal pathway. Brain Res 503: 170–174
Soriano E, Nitsch R, Frotscher M (1990) Axo-axonic chandelier cells in the rat fascia dentata-Golgi-electron microscopy and immunocytochemical studies. J Comp Neurol 293: 1–25
Steward O (1976) Topographic organization of the projections from the entorhinal area to the hippocampal formation of the rat. J Comp Neurol 167: 285–314
Szentágothai J, Arbib MA (1974) Conceptual models of neural organization. Neurosci Res Prog Bull 12: 307–510
Yeckel MF, Berger TW (1990) Feedforward excitation of the hippocampus by afferents from the entorhinal cortex: redefinition of the role of the trisynaptic pathway. Proc Natl Acad Sci (USA) 87: 5832–5836
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Li, X.G., Somogyi, P., Tepper, J.M. et al. Axonal and dendritic arborization of an intracellularly labeled chandelier cell in the CA1 region of rat hippocampus. Exp Brain Res 90, 519–525 (1992). https://doi.org/10.1007/BF00230934
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00230934