Summary
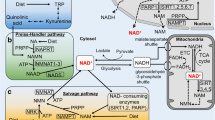
Tissue levels of NAD+ appear to be regulated primarily by the concentration of extracellular nicotinamide, which in turn is controlled by the liver in a hormone-sensitive manner. Hepatic regulation involves the conversion of excess serum nicotinamide to ‘Storage NAD+’ and inactive excretory products, and the replenishment of serum nicotinamide by the hydrolysis of ‘Storage NAD+.’ Tryptophan and nicotinic acid contribute to ‘Storage NAD+,’ and thus are additional sources of nicotinamide. In response to administered nicotinamide, there is a preferential utilization of ATP and PRPP (5-phosphorylribose-1-pyrophosphate) for the biosynthesis of NAD+. This biosynthetic priority, whose purpose appears to be the conservation of intracellular nicotinamide, may explain why nicotinamide inhibits RNA and DNA synthesis in regenerating tissues and why elevated nicotinamide levels are toxic to growing animals and to mammalian cells in culture.
Similar content being viewed by others
References
Seifert, R., Kittler, M., and Hilz, H., 1966, in Kaplan, N.O., and Kennedy, E. P., ed. Current Aspects of Biochemical Energetics, Academic Press, New York and London, pp. 413–431.
Chaykin, S., 1967. Ann. Rev. Biochem., 36, 149–170.
Gholson, R. K., Proc. Int. Symp. Vit. Nutr., Kyoto, 1967, J. Vitaminol., 14, 114–122 1968.
Hayaishi, O., Ijichi, H., and Ichiyama, A., 1967. Adv. Enz. Reg., 5, 9–22.
Dietrich, L. S., 1971. Am. J. Clin. Nutr., 24, 800–804.
Grunicke, H., Keller, H. J., Liersch, M., and Benaguid, A., 1974. Adv. Enz. Reg., 12, 397–418.
Sung, S.-C., and Williams, J. N., Jr., 1952. J. Biol. Chem., 197, 175–179.
Waravdekar, V. S., and Griffin, C. C., 1964. Exp. Cell Res., 33, 450–458.
McKee, R. W., Dickey, A., and Parks, M. E., 1968. Arch. Biochem. Biophys., 126, 760–763.
Banay-Schwartz, M., Benziman, M., and Strecker, H. J., 1969. Comp. Biochem. Physiol., 28, 177–184.
Bock, K. W., Siekevitz, P., and Palade, G. E., 1971. J. Biol. Chem., 246, 188–195.
Ueda, K., Fukushima, M., Okayama, H., and Hayaishi, O., 1975. J. Biol. Chem., 250, 7541–7546.
Chaykin, S., Dagani, M., Johnson, L., and Samli, M., 1965. J. Biol. Chem., 240, 932–938.
Huff, J. W., and Perlzweig, W. A., 1943. J. Biol. Chem., 150, 395–400.
Cantoni, G. L., 1953. J. Biol. Chem., 204, 403–416.
Mrochek, J. E., Jolley, R. L., Young, D. S., and Turner, W. J., 1976. Clin. Chem., 22, 1821–1827.
Bernofsky, C., 1979. Anal. Biochem., 96, 189–200.
Preiss, J. and Handler, P., 1958. J. Biol. Chem., 233, 493–500.
Nishizuka, Y. and Hayaishi, O., 1963. J. Biol. Chem., 238, 3369–3377.
Shuster, L., Langan, T. A., Kaplan, N. O., and Goldin, A., 1958 Nature, 182, 512–515.
Stollar, V., and Kaplan, N. O., 1961. J. Biol. Chem., 236, 1863–1866.
Rechsteiner, M., Hillyard, D., and Olivera, B. M., 1976. Nature, 259, 695–696.
Rechsteiner, M., Hillyard, D., and Olivera, B. M., 1976. J. Cell. Physiol., 88, 207–218.
Manlapaz-Fernandez, P., and Olivera, B. M., 1973. J. Biol. Chem., 248, 5150–5155.
Honjo, T., and Hayaishi, O., 1973. Current Top. Cell. Reg., 7, 87–127.
Sugimura, T., 1973. Prog. Nuc. Acid Res. Molec. Biol., 13, 127–151.
Nishizuka, Y., Ueda, K., Nakazawa, K., and Hayaishi, O., 1967. J. Biol. Chem., 242, 3164–3171.
Gardner, D. A., Sato, G. H., and Kaplan, N. O., 1972. Develop. Biol., 28, 84–93.
Eagle, H., 1955. J. Exptl. Med., 102, 595–600.
Eagle, H., 1956. Proc. Soc. Exp. Biol. Med., 91, 358–361.
Blomquist, C. H., Larson, K. E., and Taddeini, L., 1976. Exp. Cell Res., 100, 447–450.
Chang, S. C.-S-. and Bernofsky, C., 1975. Biochem. Biophys. Res. Commun., 64, 539–545.
Clark, B. R., Halpern, R. M., and Smith, R. A., 1975. Anal. Biochem., 68, 54–61.
Frank, O., Baker, H., and Sobotka, H., 1963. Nature, 197, 490–491.
Jacobson, E. L., Jacobson, M. K., and Bernofsky, C., 1974. FEBS Letters, 47, 23–25.
Jacobson E. L., Lange, R. A., and Jacobson, M. K., 1979. J. Cell. Physiol., 99, 417–426.
Schwartz, J. P., Passonneau, J. V., Johnson, G. S., and Pastan, I., 1974. J. Biol. Chem., 249, 4138–4143.
Jacobson, E. L. and Jacobson, M. K., 1976. Arch. Biochem. Biophys., 175, 627–634.
Handler, P. and Dann, W. J., 1943. J. Biol. Chem., 146, 357–368.
Brazda, F. G. and Coulson, R. A., 1946. Proc. Soc. Exptl. Biol. Med., 62, 19–20.
Revel, M. and Mandel, P., 1962. Cancer Res., 22, 456–462.
Hogeboom, G. H., and Schneider, W. C., 1952. J. Biol. Chem., 197, 611–620.
Oide, H., 1958. Gann, 49, 49–56.
Ferris, G. M. and Clark, J. B., 1971. Biochem. J., 121, 655–662.
Streffer, C. and Scholz, G., 1972. Hoppe-Seyler's Z. Physiol. Chem., 353, 1855–1862.
Swiatek, K. R., Simon, L. N., and Chao, K.-L., 1973. Biochemistry, 12, 4670–4674.
Bonsignore, A., and Bevilacqua, L., 1947. Boll. Soc. It. Biol. Sper., 23, 1216–1218.
Bonsignore, A., and Ricci, C., 1958. Scientia Med. Ital., 6,655–670.
Feigelson, P., Williams, J. N., Jr., and Elvehjem, C. A., 1951. Proc. Soc. Exp. Biol. Med., 78, 34–36.
Kaplan, N. O., Goldin, A., Humphreys, S. R., Ciotti, M. M., and Stolzenbach, F. E., 1956. J. Biol. Chem., 219, 287–298.
Kaplan, N. O., Goldin, A., Humphreys, S. R., and Stolzenbach, F. E., 1957. J. Biol. Chem., 266, 365–371.
Shuster, L., Langan, T. A., Kaplan, N. O., and Goldin, A., 1958. Nature, 182, 512–515.
Langan, T. A., Jr., Kaplan, N. O., and Shuster, L., 1959. J. Biol. Chem., 234, 2161–2168.
Dachà, U., and Ricci, C., 1948. Acta Vitaminol., 2, 30–32.
Krebs, H. A., and Veech, R. L., in Papa, S., Tager, J. M., Quagliariello, E., and Slater, E. C., eds., The Energy Level and Metabolic Control in Mitochondria, Adriatica Editrice, Bari, 1969, pp. 329–382.
Krebs, H. A., and Veech, R. L., in Sund, H., ed., Pyridine Nucleotide-Dependent Dehydrogenases, Springer-Verlag, Berlin, 1970, pp. 413–434.
Gumaa, K. A., McLean, P., and Greenbaum, A. L., 1971. Essays in Biochemistry, 7, 39–86.
Veech, R. L., in Srere, P. A. and Estabrook, R. W., eds., Microenvironments and Metabolic Compartmentation, Academic Press, New York, 1978, pp. 17–64.
Greengard, P., Quinn, G. P., and Reid, M. B., 1964. J. Biol. Chem., 239, 1887–1892.
Bernofsky, C., and Pankow, M., 1973. Arch. Biochem. Biophys., 156, 143–153.
Greengard, P., Petrack, B. and Kalinsky, H., 1967. J. Biol. Chem., 242, 152–154.
Bosch, A. J. and Harper, A. E., 1959. J. Biol. Chem., 234, 929–931.
Greengard, P., Kalinsky, H., Manning, T. J. and Zak, S. B., 1968. J. Biol. Chem., 243, 4216–4221.
Shall, S., O'Farrell, M. K., Stone, P. R., and Whish, W. J. D., in Nakahara, W., Ono, T., Sugimura, T., and Sugano, H., Differentiation and Control of Malignancy of Tumor Cells, University of Tokyo Press, 1974, pp. 69–87.
Hilz, H. and Stone, P., 1976. Rev. Physiol. Biochem. Pharmacol., 76, 1–58.
Sugimura, T., Miwa, M., Kanai, Y., Oda, K., Segawa, K., Tanaka, M., and Sakura, H., in Criss, W. E., Ono, T., and Sabine, J. R., eds., Control Mechanisms in Cancer, Raven Press, New York, 1976, pp. 231–239.
Hayaishi, O., and Ueda, K., 1977. Ann. Rev. Biochem., 46, 95–116.
Hayaishi, O., Ueda, K., Okayama, H., Kawaichi, M., Ogata, N., Oka, J., Ikai, K., Ito, S., and Shizuta, Y., in Li, C. H., ed., Versatility of Proteins, Academic Press, New York and elsewhere, 1978, pp. 151–165.
Robinson, E. A., Henrikson, O., and Maxwell, E. S., 1974. J. Biol. Chem., 249, 5088–5093.
Yoshihara, K., Tanigawa, Y., Burzio, L., and Koide, S. S., 1975. Proc. Nat. Acad. Sci., 72, 289- 293.
Astrachan, L., Colowick, S. P., and Kaplan, N. O., 1957. Biochim. Biophys. Acta, 24, 141–154.
Bernofsky, C., and Pankow, M., 1971. Biochim. Biophys. Acta, 242, 437–440.
Bücher, T., in Sund, H., ed., Pyridine Nucleotide-Dependent Dehydrogenases, Springer-Verlag, Berlin and elsewhere, 1970, pp. 439–461.
Lehninger, A., 1955. The Harvey Lectures, 49, 176–215.
Lester, R. L., Ziegler, D. M., and Green, D. E., 1957. Biochim. Biophys. Acta, 24, 155–160.
Lester, R. L., Hatefi, Y., and Welch, E., 1958. Biochim. Biophys. Acta, 29, 103–112.
Hunter, F. E., Jr., Malison, R., Bridgers, W. F., Schutz, B., and Atchison, A., 1959. J. Biol. Chem., 234, 693–699.
Birt, L. M., and Bartley, W., 1960. Biochem. J., 75, 303–315.
Purvis, J. L., and Lowenstein, J. M., 1961. J. Biol. Chem., 236, 2794–2803.
Chance, B., 1963. Ann. N. Y. Acad. Sci., 108, 322–330.
Greenspan, M. D., and Purvis, J. L., 1968. J. Biol. Chem., 243, 2921–2927.
Ernster, L., and Kuylenstierna, B., in Racker, E., ed., Membranes of Mitochondria and Chloroplasts, Van Nostrand Reinhold, New York and elsewhere, 1970, pp. 172–212.
Wenner, C. E., and Weinhouse, S., 1953. Cancer Res., 13, 21–26.
Wenner, C. E., Spirtes, M. A., and Weinhouse, S., 1951. Proc. Soc. Exptl. Biol. Med., 78, 416–421.
Kielley, R. K., 1952. Cancer Res., 12, 124–128.
Hawtrey, A. O. and Silk, M. H., 1960. Biochem. J. 74, 21–26.
Klingenberg, M., and Pette, D., 1962. Biochem. Biophys. Res. Commun., 7, 430–432.
Cori, C. F., Velick, S. F., and Cori, G. T., 1950. Biochim. Biophys. Acta, 4, 160–169.
Popják, G., in Boyer, P. D., ed., The Enzymes, Vol. 2, 3rd ed., Academic Press, New York, 1970, pp. 115–215.
Warburg, O., 1956. Science, 123, 309–314.
Boxer, G. E., and Devlin, T. M., 1961. Science, 134, 1495–1501.
Aisenberg, A. C., The Glycolysis and Respiration of Tumors, Academic Press, New York, 1961, 224 pp.
Burk, D., Woods, M., and Hunter, J., 1967. J. Nat. Cane. Inst., 38, 839–863.
von Euler, H., Schlenk, F., Heiwinkel, H., and Högberg, B., 1938. Z. Physiol. Chem., 256, 208–228.
Bernheim, F., and von Felsovanyi, A., 1940. Science, 91, p. 76.
Kensler, C. J., Suguira, K., and Rhoads, C. P., 1940. Science, 91, p. 623.
Pollack, M. A., Taylor, A., and Williams, R. J., 1942. U. of Tex. Pub. No. 4237, 56–71.
Taylor, A., Pollack, M. A., Hofer, M. J., and Williams, R. J., 1942. Cancer Res., 2, 744–747.
Schlenk, F., 1946. Cancer Res., 6, 495–496.
Carruthers, C., and Suntzeff, V., 1953. Arch. Biochem. Biophys., 45, 140–148.
Carruthers, C., and Suntzeff, V., 1954. Cancer Res., 14, 29–33.
Strength, D. R., and Seibert, M. A., 1954. Proc. Am. Assoc. Cancer Res., 1, p. 47.
Jedeikin, L., and Weinhouse, S., 1955. Proc. Am. Assoc. Cancer Res., 2, p. 26.
Jedeikin, L. A., and Weinhouse, S., 1955. J. Biol. Chem., 213, 271–280.
Jedeikin, L., Thomas, A. J., and Weinhouse, S., 1956. Cancer Res., 16, 867–872.
Narurkar, M. V., Kumta, U. S., and Sahasrabudhe, M. B., 1957. Brit. J. Cancer, 11, 482–486.
Glock, G. E., and McLean, P., 1957. Biochem. J., 65, 413–416.
Briggs, M. H., 1960. Nature, 187, 249–250.
Wintzerith, M., Klein, N., Mandel, L., and Mandel, P., 1961. Nature, 191, 467–469.
Clark, J. B., Greenbaum, A. L., and McLean, P., 1966. Biochem. J., 98, 546–556.
Oppenheimer, N. J., and Kaplan, N. O., 1974. Biochemistry, 13, 4675–4685.
Oppenheimer, N. J., and Kaplan, N. O. 1974. Biochemistry, 13, 4685–4694.
Bernofsky, C., 1978. Physiol. Chem. Physics, 10, 193–199.
Ijichi, H., Ichiyama, A., and Hayaishi, O., 1966. J. Biol. Chem., 241, 3701–3707.
Lee, Y. C., McKenzie, R. M., Gholson, R. K., and Raica, N., 1972. Biochim. Biophys. Acta, 264, 59–64.
Henderson, L. M., and Gross, C. J., 1979. J. Nutrition, 109, 654–662.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Bernofsky, C. Physiologic aspects of pyridine nucleotide regulation in mammals. Mol Cell Biochem 33, 135–143 (1980). https://doi.org/10.1007/BF00225285
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00225285