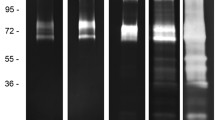
Summary
A polyclonal antibody was raised against trypsin purified from the midgut of blood-fed Aedes aegypti. Using this antibody and our modification of the peroxidase-antiperoxidase immunocytochemical reaction, strong activity was found in the lumen of the midgut at the light-microscopical level. The activity was localized mainly in the posterior part of the distensible, abdominal midgut, along the periphery of the blood bolus and within the peritrophic membrane. Immunoreactivity appeared 8 h after the blood meal and was most prominent around 24 h, coinciding with our previous spectrophotometric determinations of trypsin.
At the electron-microscopical level, secretory granules, immunocytochemically labelled with anti-trypsin antibody and protein A-colloidal gold, were first detected about 12 h after the blood meal. At 18 h, the secretory pathway could be followed immunocytochemically from the formation of granules in the Golgi complex until their release by exocytosis in the midgut lumen. By 24 h, there was a reduction in secretory granules, and large lysosomes appeared.
The process of secretion described for this mosquito is comparable to similar events in vertebrate secretory systems and the presence of an intracellular trypsinogen is suggested.
Similar content being viewed by others
References
Berner R, Rudin W, Hecker H (1983) Peritrophic membranes and protease activity in the midgut of the malaria mosquito, Anopheles Stephensi (Liston) (Insecta:Diptera) under normal and experimental conditions. J Ultrastruct Res 83:195–204
Bertram DS, Bird RG (1961) Studies on mosquito-borne viruses in their vectors. I. The normal fine structure of the midgut epithelium in the adult female Aedes aegypti L. and the functional significance of its modification following a blood meal. Trans R Soc Trop Med Hyg 55:404–423
Boorman JPT (1960) Observations on the feeding habits of the mosquito Aedes (Stegomyia) aegypti (Linnaeus): The loss of fluid after a blood meal and the amount of blood taken during feeding. Ann Trop Med Parasitol 54:8–14
Briegel H (1975) Excretion of proteolytic enzymes by Aedes aegypti after a blood meal. J Insect Physiol 21:1681–1684
Briegel H, Lea AO (1975) Relationship between protein and proteolytic activity in the midgut of mosquitoes. J Insect Physiol 21:1597–1604
Brown MR, Raikhel AS, Lea AO (1985) Ultrastructure of midgut endocrine cells in the adult mosquito Aedes aegypti (Diptera). Tissue and Cell 17:709–721
Espinoza-Fuentes FP, Ferreira C, Terra WR (1984) Spatial organization of digestion in the larval midgut and imaginal stages of the sciarid fly Trichosia pubescens. Insect Biochem 14:631–638
Farquhar MG, Palade GE (1981) The Golgi apparatus (complex) — (1954–1981) — from artifact to center stage. J Cell Biol 91:77–103
Freyvogel TA, Stäubli W (1965) The formation of the peritrophic membrane in Culicidae. Acta Trop (Basel) 22:118–147
Gooding RH (1966) Physiological aspects of digestion of the blood meal by Aedes aegypti (Linnaeus) and Culex fatigans (Wiedemann). J Med Entomol 3:53–60
Gooding RH (1973) The digestive processes of haematophagous insects. IV. Secretion of trypsin by Aedes aegypti (Diptera:Culicidae). Can Entomol 105:599–603
Gooding RH (1975) Digestive enzymes and their control in haematophagous arthropods. Acta Trop (Basel) 32:96–111
Graf R, Briegel H (1982) Comparison between aminopeptidase and trypsin activity in blood-fed females of Aedes aegypti. Rev Suisse Zool 89:845–850
Graf R, Briegel H (1985) Isolation of trypsin isozymes from the mosquito Aedes aegypti (L.) Insect Biochem 15:611–618
Hecker H (1977) Structure and function of midgut epithelial cells in Culicidae mosquitoes (Insecta, Diptera). Cell Tissue Res 184:321–341
Hecker H, Freyvogel TA, Briegel H, Steiger R (1971) Ultrastructural differentiation of the midgut epithelium in females Aedes aegypti (L.) (Insecta, Diptera) imagines. Acta Trop (Basel) 28:80–104
Houk EJ (1977) Midgut ultrastructure of Culex tarsalis (Diptera: Culicidae) before and after a blood meal. Tissue and Cell 9:103–118
Lämmli U (1970) Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 227:680–685
Lehane MJ (1976) Digestive enzyme secretion in Stomoxys calcitrans (Diptera:Muscidae). Cell Tissue Res 170:275–287
Ouchterlony O (1953) Antigen-antibody reactions in gels. Acta Pathol Microbiol Scand 32:231–240
Raikhel AS (1984) The accumulative pathway of vitellogenin in the mosquito oocyte: a high-resolution immuno-and cytochemical study. J Ultrastruct Res 87:285–302
Raikhel AS (1986) Lysosomes in the cessation of vitellogenin secretion by the mosquito fat body; selective degradation of Golgi complexes and secretory granules. Tissue and Cell (in press)
Richards AG, Richards PA (1971) Origin and composition of the peritrophic membrane of the mosquito, Aedes aegypti. J Insect Physiol 17:2253–2275
Rudin W, Hecker H (1979) Functional morphology of the midgut of Aedes aegypti L. (Insecta, Diptera) during blood digestion. Cell Tissue Res 200:193–203
Santos CD, Terra WR (1984) Plasma membrane-associated amylase and trypsin. Intracellular distribution of digestive enzymes in the midgut of the cassava hornworm, Erinnyis ello. Insect Biochem 14:587–594
Sharma BR, Martin MM, Shafer JA (1984) Alkaline proteases from the gut fluids of the detritus feeding larvae of the crane fly Tipula abdominalis (Say) (Diptera, Tipulidae). Insect Biochem 14:37–44
Shuckle RH, Murdock LL, Gallun RL (1985) Identification and partial characterization of a major gut proteinase from the larvae of the Hessian fly, Mayetiola destructor (Say) (Diptera: Cecidomyiidae). Insect Biochem 15:93–101
Stäubli W, Freyvogel TA, Suter J (1966) Structural modification of the endoplasmic reticulum of midgut epithelial cells of mosquitoes in relation to blood intake. J Microsc 5:189–204
Sternberger LA, Hardy PH, Cuculis JJ, Meyer HG (1970) The unlabelled antibody enzyme method of immunocytochemistry. Preparation and properties of soluble antigen-antibody complex (horseradish antihorseradish peroxidase) and its use in identification of spirochaetes. J Histochem Cytochem 18:315–333
Stohler H (1957) Analyse des Infektionsverlaufs von Plasmodium gallinaceum im Darme von Aedes aegypti. Acta Trop (Basel) 14:302–352
Terra WR, Ferreira C, De Bianchi AG (1979) Distribution of digestive enzymes among the endo- and ectoperitrophic spaces and midgut cells of Rhynchosciara and its physiological significance. J Insect Physiol 25:487–494
Towbin H, Staehelin T, Gordon J (1979) Electrophoretic transfer of proteins from polyacrylamide gels to nitrocellulose sheets: Procedure and some applications. Proc Natl Acad Sci USA 76:4350–4354
Williams JC, Hagedorn HH, Beyenbach KW (1983) Dynamic changes in flow rate and composition of urine during the postbloodmeal diuresis in Aedes aegypti (L.). J Comp Physiol 153:257–265
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Graf, R., Raikhel, A.S., Brown, M.R. et al. Mosquito trypsin: immunocytochemical localization in the midgut of blood-fed Aedes aegypti (L.). Cell Tissue Res. 245, 19–27 (1986). https://doi.org/10.1007/BF00218082
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00218082