Summary
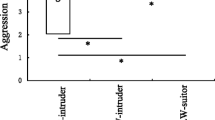
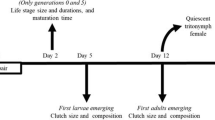
Two forms of males occur in the acarid mite, Caloglyphus berlesei. One of them uses its thickened third pair of legs to kill other males; the other, without modified legs, does not attack other males. Previous studies have shown that the form of the male is environmentally determined: “fighter” males develop only at low population densities and their development can be suppressed by substances emanating from dense colonies. In this study, the duration of development, longevity and virility of the two male morphs were measured. The only significant difference was that fighters had a shorter development time between the two last moults. In another experiment, two groups of colonies were maintained under the same conditions but had different numbers of individuals: small colonies contained 2 fighters and 2 non-fighters, whereas large colonies contained 30 males of each type. The relative reproductive success of both morphs was estimated from numbers of matings. In small colonies, the estimated reproductive success (ERS) was significantly higher for fighters than for non-fighters, mainly because in over 50% of these colonies a single fighter male managed to kill all the rival males and monopolize the females. The opposite was true in large colonies, in which non-fighters achieved significantly higher ERS, mainly because the fighters were killed in fights more often than the non-fighters. This implies that the ratio of costs to benefits obtained by adopting the fighter-male strategy increases with the number of rival males, which may explain conditional male development in C. berlesei.
Similar content being viewed by others
References
Alexander AJ, Staaden M van (1989) Alternative sexual tactics in male bladder grasshoppers (Orthoptera, Pneumoridae). In: Bruton MN (ed) Alternative life histories of animals Kluwer, Academic, Dordrecht, pp 261–277
Austad SN (1984) A classification of alternative reproductive behaviors and methods for field-testing ESS models. Am Zool 24:309–319
Chmiclewski W, Lipa JJ (1967) Biological and ecological studies on Caloglyphus mite (Acarina: Acaridae) associated with Scarabaeid. Acta Parasitol Pol 14:179–190
Crespi BJ (1988) Adaptation, compromise and constraint: the development, morphometrics and behavioral basis of a fighter-flier polymorphism in male Haplothrips karnyi (Insecta: Thysanoptera). Behav Ecol Sociobiol 23:93–104
Dawkins R (1980) Good strategy or evolutionary stable strategy? In: Barlow GW, Silverberg J (eds) Sociobiology: beyond nature/nurture? Westview, Boulder, pp 331–367
Dimock DV (1983) In defense of harem: intraspecific aggression by male water mites (Acari: Unionicolidae). Ann Entomol Soc Am 76:463–465
Dominey WJ (1980) Female mimicry in the bluegill sunfish — a genetic polymorphism? Nature 284:546–548
Dominey WJ (1984) Alternative mating tactics and evolutionary stable strategies. Am Zool 24:385–396
Eberhard WG (1982) Beetle horn dimorphism: Making the best of a bad lot? Am Nat 119:420–426
Emlen ST, Oring LW (1977) Ecology, sexual selection, and the evolution of mating systems. Science 197:215–223
Erkay SA (1987) Observations on the biology of two species of acarid mites. Folia Entomol Hung 48:21–27
Gross MR (1982) Sneakers, satellites and parentals: Polymorphic mating strategies in North American sunfishes. Z Tierpsychol 60:1–16
Hamilton WD (1979) Wingless and fighting males in fig wasps and other insects. In: Blum MS, Blum NA (eds) sexual selection and reproductive competition in insects. Academic Press, New York, pp 167–222
Houck MA, OConnor BM (1991) Ecological and evolutionary significance of phoresy in the Astigmata. Annu Rev Entomol 36:611–636
Hughes AM (1976) The mites of stored food and houses. Tech Bull Min Agric Fish Food 9
Hunter PE (1960) Plastic paint as a marker for mites. Ann Entomol Soc Am 53:698
Kranz GW (1978) A manual of acarology, 2nd edn. Oregon State University, Corvallis
Maynard Smith J (1982) Evolution and the theory of games. Cambridge University Press, Cambridge
Moran NA (1992) The evolutionary maintenance of alternative phenotypes. Am Nat 139:971–989
Radwan J (1991) Sperm competition in the mite Caloglyphus berlesei. Behav Ecol Sociobiol 29:291–296
Radwan J (1992) The influence of a crowded environment on the size of males of Caloglyphus berlesei. Int J Acarol 18:67–68
Radwan J (1993) Kin recognition in the acarid mite Caloglyphus berlesei: negative evidence. Anim Behav 45:200–202
Radwan J, Witaliński W (1991) Sperm competition. Nature 352:671–672
Samsinak K (1960) Über einige myrmekophile Milben aus der Familie Acaridae. Casopis Ceskoslovenske Spolecnosti Entomoloicke Acta. Soc Entomol Cechosloveniae 57:185–193
Shuster SM, Wade MJ (1991) Equal mating success among male reproductive strategies in a marine Isopod. Nature 350:608–610
Timms S, Ferro DN, Waller JB (1980) Suppression of production of pleomorphic males in Sancassania berlesei (Michael) (Acari: Acaridae). Int J Acarol 6:91–96
Timms S, Ferro DN, Emberson RM (1981) Andropolymorphism and its heritability in Sancassnia berlesei (Michael) (Acari: Acaridae). Acarologia 22:391–398
Woodring JP (1969a) Environmental regulation of andropolymorphism in Tyroglyphids (Acari). In: Evans GO (ed) Proceedings of the Second International Congress of Acarology. Academiai Kiado, Budapest, pp 433–440
Woodring JP (1969b) Observations on the biology of six species of acarid mites. Ann Entomol Soc Am 62:102–108
Zeh DW, Zeh JA (1992) Dispersal-generated sexual selection in a beetle-riding pseudoscorpion. Behav Ecol Sociobiol 30:135–142
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Radwan, J. The adaptive significance of male polymorphism in the acarid mite Caloglyphus berlesei . Behav Ecol Sociobiol 33, 201–208 (1993). https://doi.org/10.1007/BF00216601
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00216601