Summary
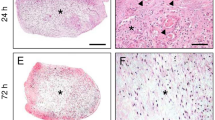
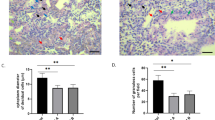
Sprague-Dawley rats were oöphorectomized and after a 2–3 week recovery period were given daily injections of progesterone (2.0 mg/0.1 ml) for six consecutive days. On the fourth day of progesterone treatment 0.2 mg of estradiol 17β was given in addition and the right uterine cornua were subjected to one of five experimental maneuvers. On the sixth day of progesterone treatment the uterine cornua were weighed and processed for light and electron microscopy. The weights of all left cornua (84.6 +- 3.7 mg) and the right cornua of PBS-injected (93.3 +- 11.5 mg) and sham operated uteri (83.6 +- 19.8 mg) were comparable. A significant increase (p<0.001) in weight was found in cornua that received PGF2α (144 +- 6.7 mg), PGF2α with mild local trauma (scratch) (146 +- 28.0 mg), and scratch alone (162 +- 12.7 mg). The majority of cornua treated by scratch alone, or by PGF2α with or without scratch, showed a decidual cell reaction by light microscopy and had a significantly higher mitotic index than those treated with saline, or by sham operation. When specimens were evaluated for the presence of the OCR, the highest rank was found in tissues treated by scratch alone or by PGF2α with or without scratch. Morphometric evaluation by light microscopy indicated that the extent of decidualization in PGF2α-treated tissue was comparable to that of scratchtreated tissue. Ultrastructural observation of PGF2α-treated tissue revealed that decidual cells closely resembled those treated with scratch. However, electron microscopic morphometry revealed that cells that responded to PGF2α had higher volume and surface densities of organelles associated with metabolic activity than did cells responding to scratch alone. These results demonstrate that locally administered PGF2α can initiate, in the hormonally prepared mature oöphorectomized rat, a DCR comparable to that induced by local trauma.
Similar content being viewed by others
References
Anteby SO, Bauminger S, Zor U, Under HR (1975) Prostaglandin synthesis in decidual tissue of the rat uterus. Prostaglandins 10(6):991–999
Castracane VD, Saksena SK, Shaikh AA (1974) Effect of IUDs prostaglandin and indomethacin on decidual cell reaction in the rat. Prostaglandins 6(5):397–404
DeFeo VJ (1963) Determination of the sensitive period for the induction of deciduomata in the rat by different inducing procedures. Endocrinol 73:488–497
DeFeo VJ (1967) Decidualization. In: Wynn RM (ed) Cellular biology of the uterus. Appleton-Century Crofts, New York, pp 192–290
Finn CA, Keen PM (1963) The induction of deciduomata in the rat. J Embryol Exp Morphol 11:673–682
Glassar S (1972) The uterine environment in implantation and decidualization. In: Baiin H, Glassar S (eds) Reproductive biology. Excerpta Medica, Amsterdam
Jollie WP, Bencosme SA (1965) Electron microscopic observations in primary decidua formation in the rat. Am J Anat 116:217–236
Kennedy TG (1979) Prostaglandins and increased endometrial vascular permeability resulting from the application of artificial stimulus to the uterus of the rat sensitized for the decidual cell reaction. Biol Reprod 20(3):560–566
Krehbiel RH (1937) Cytological studies of the decidual reaction in the rat during early pregnancy and the production of deciduomata. Physiol Zool 10:212–233
Loeb L (1908) The experimental production of the maternal part of the placenta in the rabbit. Proc Soc Exp Biol Med 5:102–105
Meyers KP (1970) Hormonal requirements for the maintenance of oestradiol-induced inhibition of uterine sensitivity in the ovariectomized rat. J Endocrinol 46:341–346
Miller M, O'Morchoe CCC (Submitted) Inhibition of artificially induced decidual cell reaction by indomethacin in the mature oophorectomized rat
Psychoyos A (1970) Hormonal requirements for egg implantation. In: Raspe G (ed) Schering symposium on mechanisms involved in conception (Advances in Biosciences 4). Pergamon Press New York
Psychoyos A (1973) Endocrine control of egg implantation. In: Greep RO, Astwood EB, Geiger SR (eds) APS, Washington DC, Vol 2: 187–215
Ramwell PW, Shaw JE (1970) Biological significance of the prostaglandins. Recent Prog Horm Res 26:139–173
Sananes N, Baulieu EE, LeGoscogne C (1976) Prostaglandins as inductive factor of decidualization in the rat uterus. Molec Cell Endocr 6:153–158
Sananes N, Weiller S, Baulieu EE, LeGoscogne C (1978) In vitro decidualization of rat endometrial cells. Endocrinol 103:86–95
Shelesnyak MC, Kraicer PF, Zeilmaker GH (1963) Studies on the mechanism of decidualization in the oestrogen surge of pseudopregnancy and progravidity and its role in the process of decidualization. Acta Endocrinol 42:225–232
Velardo JT, Dawson AB, Olsen AG, Hisaw FL (1953) Sequence of histological changes in the uterus during prolongation of pseudopregnancy associated with the presence of deciduoma. Am J Anat 93:273–305
Weems CW (1979) Prostaglandins F in uterine and ovarian compartments and in plasma from the uterine vein, ovarian artery and vein, and abdominal aorta of pseudopregnant rats with and without deciduomata. Prostaglandins 17(6):873–980
Weibel ER (1973) Stereological techniques for electron microscopic morphometry. In: Hayat MA (ed) Principles and techniques of electron microscopy. Van Nostrand Reinhold Company Princeton Vol 3:237–296
Yochim JM, DeFeo VJ (1963) Hormonal control of the onset, magnitude and duration of uterine sensitivity in the rat by steroid hormones of the ovary. Endocrinol 72:317–326
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Miller, M.M., O'Morchoe, C.C.C. Decidual cell reaction induced by prostaglandin F2α in the mature oöphorectomized rat. Cell Tissue Res. 225, 189–199 (1982). https://doi.org/10.1007/BF00216228
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00216228