Abstract
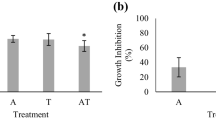
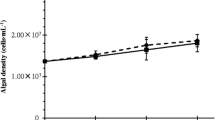
2,4-D was applied to a cove in Kentucky Lake which was highly infested with Myriophyllum spicatum (Eurasian watermilfoil). Effects of 2,4-D on nontarget algal communities were monitored concurrently in the field and in laboratory microcosms for eight days. Results indicated that indirect effects of water temperature and increased nutrient concentrations due to lysis in milfoil plants may be more important in the field community dominated by Chlorophyta, Pyrrhophyta, and Bacillariophyta. 2,4-D applied at the label-recommended rate of 2 mg/L or less stimulated total community growth in both laboratory and field indicating a possible hormonal effect of 2,4-D on algae. Reduced community growth and metabolism at high laboratory concentrations of 100 mg/L and 1000 mg/L may indicate an inhibitory effect on photosynthesis and/or respiration in algae. 2,4-D altered the laboratory community structure and function in all concentrations tested. Heterotrophic taxa such as Nitzschia, Euglena, Chlamydomonas, Mallomonas, Anabaena, and Oscillatoria appeared to be least affected by 2,4-D at high concentrations. Scenedesmus, Pediastrum, Characiosiphon, Navicula, Melosira, and Fragilaria appeared to be more sensitive, even in the lowest concentrations.
Similar content being viewed by others
References
American Public Health Association, American Water Works Association, and Water Environment Federation. Standard methods for the examination of water and wastewater, 18th ed. Washington, DC 1992
Ashton FM, Crafts AS (1973) Mode of action of herbicides. Wiley Interscience Publication, NY
Barnes LE (1984) The algal composition and population dynamics in selected embayment and channel areas of Kentucky Reservoir. Master's Thesis. Murray State University, Murray, KY
Bednarz T (1981) The effect of 2,4-D acid on green and blue-green algae in unialgal and mixed cultures. Acta Hydrobiol 23(2):173–182
Benson CE, Rushforth SR (1975) The algal flora of Huntington Canyon, Utah, U.S.A. Brigham Young University, UT
Bertagnolli BL, Nadakavokaren MJ (1974) Some physiological responses of Chlorella pyrenoidosa to 2,4-dichlorophenoxyacetic acid. J Exp Bot 25 (84):180–188
Boyle TP (1980) Effects of the aquatic herbicide 2,4-D DMA on the ecology of the experimental ponds. Environ Pollut 21(2):35–49
Bulding JG, Rass WR, Pirsch KM (1970) Effects of 2,4-D on species composition and chlorophyll contents in mixed cultures of algae. Weed Sci 18:750–753
Camburn KE (1982) The diatoms (Bacillariophyceae) of Kentucky: a checklist of previously reported taxa. Trans of Kentucky Acad Sci 43 (1–2):10–20
Chapman MA, Chapman DJ (1973) The algae. MacMillan, London
Couch RW, Nelson EN (1982) Effects of 2,4-D on non-target species in Kerr Reservoir. J Plant Manag 20:8–13
Das B, Singh PK (1977) The effect of 2,4-dichlorophenoxyacetic acid on growth and nitrogen-fixation of blue-green alga Anabaenopsis raciborskii. Arch Environ Contam Toxicol 5(4):437–445
Desikachary TV (1959) Cyanophyta. Indian Council of Agricultural Research, New Delhi
Dillard GE, Crider SB (1970) Kentucky algae, I. Trans Kentucky Acad Sci 31:66–72
—, Moore SP, Garrett LS (1976) Kentucky algae, II. Trans Kentucky Acad Sci 37:20–25
Dodd JJ (1987) Diatoms, the illustrated flora of Illinois. Southern Illinois University Press, IL
Dunnett CW (1955) A Multiple comparisons procedure for comparing several treatments with a control. J Amer Stat Assoc 50:1096–1121
Gangstad EO (1986) Freshwater vegetation management. Thomas Publications, Fresno, CA
International Organization for Standardization. Algal growth inhibition test. Draft Method. Delft, Netherlands, 1983
Kobraei ME (1994) Effects of the herbicide 2,4-D on algal communities in Kentucky Lake: Laboratory and field approaches. Ph.D. Dissertation. University of Louisville, Louisville, KY
McConnell WJ (1962) Productivity relations in carboy microcosms. Limnol Oceanogr 7:335–343
Pandey AK, Tiwari DN (1986) Action of 2,4-dichlorophenoxyacetic acid on Nostoc linckia: impact of glucose and tryptophan. Folia Microbiol 31(1):50–55
Poorman AE (1973) Effects of pesticides on Euglena gracilis. I. Growth studies. Bull Environ Contam Toxicol 10:25–28
Pratt JR, Bowers NJ (1990) A microcosm procedure for estimating ecological effects of chemicals and mixtures. Toxicol Assess 5:189–205
Prescott GW (1970) How to know the fresh-water algae. WM. C. Brown, IA
— (1982) Algae of the western Great Lakes area. Otto Koeltz Science Publ., Koenigstein, Germany
Prather KW (1977) Some limnological parameters of Vickers Bay, Kentucky Lake. Masters Thesis. Murray State University, Murray, KY
Round FE (1973) The biology of the algae. St. Martin's Press, NY
Ross PE and Munawar M (1981) Preference for nannoplankton size fractions in Lake Ontario zooplankton grazing. J Great Lakes Res 7(1):65–67
Silva H (1951) Algae of the Tennessee Valley region: a manual for identification. Part 1 and Part 2. Ph.D. Dissertation. Michigan State College, East Lansing, MI
Smith GM (1950) The fresh-water algae of the United States. McGraw-Hill, NY
Stevenson RJ, Singe R, Roberts DA, Boylen CW (1985) Patterns of epipelic algal abundance with depth, trophic status, and acidity in poorly buffered New Hampshire lakes. Can J Fish Aquat Sci 42:1501–1512
—, Lowe RL (1986) Sampling and interpretation of algal patterns for water quality assessments. In Isom BG (ed) Rationale for sampling and interpretation of ecological data in the assessment of freshwater ecosystems. Special Technical Publication 894. American Society for Testing and Materials, Philadelphia, p 118
Taub FB (1984) Synthetic microcosms as biological models of algal communities. In Shubert LE (ed) Algae as ecological indicators. Academic Press, NY, p 363
—, Conquest LL (1989) Repeatability and reproducibility of the standardized aquatic microcosm: statistical properties. Aquat Toxicol Hazard Assess 2:159–177
-- (1991) Standard practice for standardized aquatic microcosm: fresh water. In Annual Book of American Society for Testing and Materials Standards, Philadelphia, p 1017
Tennessee Valley Authority (1974) Quality of water in Kentucky Reservoir. Division of Environmental Planning, E-WQ-74-3
-- (1993) Supplemental environmental impact statement. Aquatic plant management program. Volume I. TVA/WM-93/8. EA Project No. 11955.02
Tiffany LH, Britton ME (1952) The algae of Illinois. The University of Chicago Press, Chicago, IL
Tilden JE (1968) The algae and their life relations. Fundamentals of phycology. Hafner Co, NY
Tiwari DN, Pandey AK, Mishra AK, Srivastava V (1982) Role of 2,4-D on growth and macromolecular synthesis in a cyanobacterium Anabaena cylindrica. Ind J Bot 5(2):111–114
U.S. Environmental Protection Agency. Algal assay procedure bottle test. National Eutrophication Research Program. Corvallis, OR, 1971
Vinyard WC (1979) Diatoms. Mad River Press, Eureka, CA
Vollenweider RA (1974) A manual on methods of measuring primary production in aquatic environments. Blackwell Scientific, London
Wetzel RG (1983) Limnology. WB Saunders, Philadelphia
Wojtalik TA, Hall TF, Hill L (1971) Monitoring ecological conditions associated with wide-scale applications of DMA 2,4-D to aquatic environments. Pest Monit J 4(4):184–203
Wong PK, Chang L (1988) The effects of 2,4-D herbicide and organophosphorus herbicides on growth, photosynthesis, and chlorophyll a synthesis of Chlamydomonas reinhardtii (mt+). Environ Pollut 55:179–189
Woodford EK, Holly K, McCready CC (1958) Herbicides. Ann Rev Plant Physiol 9:311–358
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Kobraei, M.E., White, D.S. Effects of 2,4-Dichlorophenoxyacetic acid on Kentucky algae: Simultaneous laboratory and field toxicity testings. Arch. Environ. Contam. Toxicol. 31, 571–580 (1996). https://doi.org/10.1007/BF00212442
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF00212442