Summary
Tactile spines are large cuticular sense organs that appear to provide insects with a sense of touch which is spatially coarse but of great sensitivity. Cockroach legs have a number of these spines on each leg and a particularly prominent spine on the end of each femur, the femoral tactile spine. The ease of recording afferent activity from this spine during mechanical stimulation has made it one of the most thoroughly studied insect mechanoreceptors and yet it has never been examined by electron microscopy.
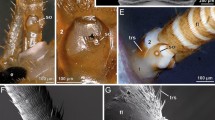
We report here the results of an examination of the femoral tactile spine by both scanning and transmission electron microscopy, as well as by light microscopy. The spine is shown to be innervated by a single sensory bipolar neuron with its soma located in the base of the spine. A canal through the wall of the spine leads to the outside and emerges just above the junction between the base of the spine and its articulating socket membrane. The sensory dendrite of the neuron passes from the soma through this canal and forms a modified ciliary sensory ending with the typical dendritic sheath and dense tubular body that is characteristic of insect mechanosensory cuticular sensilla. The tubular body is embedded in a cuticular terminal plug which closes the exterior end of the canal but appears to be fastened to the spine by a very flexible ring of cuticle. This plug is connected to the socket membrane by a specialized socket attachment which presumably serves to move the plug relative to the wall of the spine during movement of the spine within the socket. The morphology of this sensillum is discussed in terms of the possible ways in which it is stimulated by movements of the spine and also in light of the dynamic behaviour of the receptor which is now very well described.
Similar content being viewed by others
References
Albert PJ, Zacharuk RY, Wong L (1976) Structure, innervation and distribution of sensilla on the wings of the grasshopper. Can J Zool 54:1542–1553
Bassemir U, Hansen K (1980) Single-pore sensilla of Damsel-fly larvae: representatives of phylogenetically old contact chemoreceptors? Cell Tissue Res 207:307–320
Bromley AK, Dunn JA, Anderson M (1980) Ultrastructure of the antennal sensilla of aphids. II. Trichoid, chordotonal and campaniform sensilla. Cell Tissue Res 205:493–511
Chapman KM (1965) Campaniform sensilla on the tactile spines of the legs of the cockroach. J Exp Biol 41:191–203
Chapman KM, Smith RS (1963) A linear transfer function underlying impulse frequency modulation in a cockroach mechanoreceptor. Nature (London) 197:699–700
Chapman KM, Mosinger JL, Duckrow RB (1979) The role of viscoelastic coupling in sensory adaptation in an insect mechanoreceptor. J Comp Physiol 131:1–12
French AS (1980) Sensory transduction in an insect mechanoreceptor: linear and nonlinear properties. Biol Cybernetics 38:115–123
French AS, Wong RKS (1976) The responses of trochanteral hair plate sensilla in the cockroach to periodic and random displacements. Biol Cybernetics 22:33–38
French AS, Sanders EJ (1979) The mechanism of sensory transduction in the sensilla of the trochanteral hair plate of the cockroach, Periplaneta americana. Cell Tissue Res 198:159–174
French AS, Holden AV, Stein RB (1972) The estimation of the frequency response function of a mechanoreceptor. Kybernetik 11:15–23
Gaffal KP, Theiss J (1978) The tibial thread hairs of Acheta domesticus L. (Saltatoria, Gryllidae). The dependence of stimulus transmission and mechanical properties on the anatomical characteristics of the socket apparatus. Zoomorphologie 90:41–51
Gaffal KP, Tichy H, Theiss J, Seelinger G (1975) Structural polarities in mechanosensitive sensilla and their influence on stimulus transmission (Arthropoda). Zoomorphologie 82:79–103
Gnatzy W (1978) Development of the filiform hairs on the cerci of Gryllus bimaculatus Deg. (Saltatoria, Gryllidae). Cell Tissue Res 187:1–24
Karnovsky MJ (1965) A formaldehyde-glutaraldehyde fixative of high osmolarity for use in electron microscopy. J Cell Biol 27:137a
Lewis CT (1970) Structure and function in some external receptors. Symp Roy Entomol Soc London 5:59–76
Matsumoto DE, Farley RD (1978) Comparison of the ultrastructure of stimulated and unstimulated mechanoreceptors in the taste hairs of the blowfly, Phaenicia serricata. Tissue and Cell 10:63–76
McIver SB (1975) Structure of cuticular mechanoreceptors of arthropods. Ann Rev Entomol 20:381–397
Moore RK (1960) Travelling wave engineering. McGraw-Hill, New York
Moran DT, Varela FG (1971) Microtubules and sensory transduction. Proc Natl Acad Sci USA 68:757–760
Moran DT, Chapman KM, Ellis RA (1971) The fine structure of cockroach campaniform sensilla. J Cell Biol 48:155–173
Moran DT, Rowley JC, Varela FG (1975) Ultrastructure of the grasshopper proximal femoral chordotonal organ. Cell Tissue Res 161:445–457
Moran DT, Rowley JC, Zill SN, Varela FG (1976) The mechanism of sensory transduction in a mechanoreceptor. Functional stages in campaniform sensilla during the moulting cycle. J Cell Biol 71:832–847
Moran DT, Varela FG, Rowley JC (1977) Evidence for active role of cilia in sensory transduction. Proc Natl Acad Sci USA 74:793–797
Müller M, Honegger H-W, Nickel E, Westphal C (1978) The ultrastructure of campaniform sensilla on the eye of the cricket, Gryllus campestris. Cell Tissue Res 195:349–357
Pringle JW, Wilson VJ (1952) The response of a sense organ to a harmonic stimulus J Exp Biol 29:220–234
Rice MJ (1975) Insect mechanoreceptor mechanisms. In: Galun R, Hillman P, Parnas L, Werman R (eds) Sensory physiology and behavior. Plenum Press, New York
Rice MJ (1976) Contact chemoreceptors on the ovipositor of Lucilia cuprina (Wied.), the Australian sheep blowfly. Aust J Zool 24:353–360
Rice MJ, Galun R, Finlayson LH (1973) Mechanotransduction in insect neurones. Nature New Biol (London) 241:286–288
Schweidler ER von (1907) Studien über die Anomalien in Verhalten der Dielektrika. Ann Phys 24:711–770
Slifer EH (1961) The fine structure of insect sense organs. Int Rev Cytol 11:125–159
Spinola SM, Chapman KM (1975) Proprioceptive indentation of the campaniform sensilla of cockroach legs. J Comp Physiol 96:257–272
Thorson J, Biederman-Thorson M (1974) Distributed relaxation processes in sensory adaptation. Science 183:161–172
Wiederhold ML (1976) Mechanotransduction in sensory and motile cilia. Ann Rev Biophys Bioengng 5:39–62
Zacharuk RY (1980) Ultrastructure and function of insect chemosensilla. Ann Rev Entomol 25:27–47
Zacharuk RY, Albert PJ (1978) Ultrastructure and function of scolopophorous sensilla in the mandible of an elaterid larva (Coleoptera). Can J Zool 56:246–259
Zacharuk RY, Albert PJ, Bellamy FW (1977) Ultrastructure and function of digitiform sensilla on the labial palp of a larval elaterid (Coleoptera). Can J Zool 55:569–578
Author information
Authors and Affiliations
Additional information
Supported by the Canadian Medical Research Council. The authors gratefully acknowledge the expert technical assistance of Sita Prasad and Rodney Gramlich
Rights and permissions
About this article
Cite this article
French, A.S., Sanders, E.J. The mechanosensory apparatus of the femoral tactile spine of the cockroach, Periplaneta americana . Cell Tissue Res. 219, 53–68 (1981). https://doi.org/10.1007/BF00210018
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00210018