Abstract
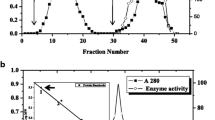
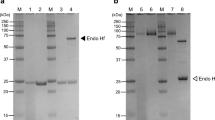
The α-xylosidase which is involved in the postgerminative mobilisation of xyloglucan in nasturtium seed cotyledons has now been purified to apparent homogeneity by a facile procedure involving lectin affinity chromatography. The purified enzyme, a glycoprotein, moved as a single band (apparent molecular weight 85000) on sodium dodecyl sulphate-gel electrophoresis, whilst isoelectric focusing gave a number of enzymatically active protein bands spanning the range pI = 5.0 to 7.1 (maximum activity at pI = 6.1). The enzyme did not hydrolyse the simple α-xylosides p-nitrophenyl-α-d-xylopyranoside and woprimeverose (α-d-Xyl(1→6)-d-Glc), or polymeric tamarind-seed xyloglucan. It released xylose from a complex mixture of oligosaccharides produced by exhaustive hydrolysis of tamarind seed xyloglucan using the xyloglucan-specific endo-(1→4)-β-d-glucanase from germinated nasturtium seeds (M. Edwards et al. 1986, J. Biol. Chem., 261. 9489–9494). The three xyloglucan oligosaccharides of lowest molecular size were purified from this mixture and were shown by 1H-nuclear magnetic resonance (1H-NMR) and enzymatic analysis to have the structures:
Similar content being viewed by others
Abbreviations
- Con A:
-
Concanavalin A
- DEAE:
-
diethylaminoethyl
- Gal:
-
galactose
- Glc:
-
glucose
- HPLC:
-
high-performance liquid chromatography
- M r :
-
apparent molecular mass
- NMR:
-
nuclear magnetic resonance
- pI:
-
isoelectric point
- SDS-PAGE:
-
sodium dodecyl sulphate-polyacrylamide gel electrophoresis
- Xyl:
-
xylose
References
Bergmeyer, H.U., Berndt, E., Schmidt, F., Stark, H. (1974) D-Glucose determination with hexokinase and glucose-6-phosphate dehydrogenase. In: Methods of enzymatic analysis, pp. 1196–1201, Bergmeyer, U., ed. Verlag Chemie, Weinheim
Clegg, J.C.S. (1982) Glycoprotein detection in nitrocellulose transfers of electrophoretically separated protein mixtures using Concanavalin A peroxidase: application to Arenavirus and Flavivirus proteins. Anal. Biochem. 127, 389–394
Cleland, W.W. (1979) Statistical analysis of enzyme kinetic data. Methods Enzymol. 63, 103–138
Dawson, R.M.C., Elliott, D.C., Elliott, W.H., Jones, K.M. (1982) Data for biochemical research. Clarendon Press, Oxford
Dische, Z. (1962) Color reactions of carbohydrates. In: Methods in carbohydrate chemistry, vol. 1, pp. 475–514, Whistler, R.L., Wolfrom, M.L., eds. Academic Press, New York
Dubois, M., Gilles, K.A., Hamilton J.K., Rebers, P.A., Smith F. (1956) Colorimetric method for determination of sugars and related substances. Anal. Chem. 28, 350–356
Edwards, M., Dea, I.C.M., Bulpin, P.V., Reid, J.S.G., (1985) Xyloglucan (amyloid) mobilisation in the cotyledons of Tropaeolum majus L. seeds following germination. Planta 163, 133–140
Edwards, M., Dea, I.C.M., Bulpin, P.V., Reid, J.S.G. (1986) Purification and properties of a novel xyloglucan-specific endo-(1→4)-β-d-glucanase from germinated nasturtium seeds (Tro-paeolum majus L.). J. Biol. Chem. 261, 9489–9494
Edwards, M., Bowman, Y.J.L., Dea, I.C.M., Reid, J.S.G. (1988) A β-d-galactosidase from nasturtium (Tropaeolum majus L.) cotyledons. Purification, properties and demonstration that xyloglucan is the natural substrate. J. Biol. Chem. 263, 4333–4337
Fairbanks, G., Steck, T.L., Wallach, D.F.H. (1971) Electrophoretic analysis of the major polypeptides of the human erythrocyte membrane. Biochemistry 10, 2606–2617
Fry, S.C. (1987) Formation of isodityrosine by peroxidase isozymes. J. Exp. Bot. 38, 853–862
Gidley, M.J., Lillford, P.J., Rowlands, D.W., Lang, P., Dentini, M., Crescenzi, V., Edwards, M., Fanutti, C., Reid, J.S.G. (1991) Structure and solution properties of tamarind seed polysaccharide. Carbohydr. Res., in press
Halliwell, G., Riaz, M. (1970) The formation of short fibres from native cellulose by components of Trichoderma koningii cellulase. Biochem. J. 116, 35–42
Hayashi, T. (1989) Xyloglucans in the primary cell wall. Annu. Rev. Plant Physiol. Plant Mol. Biol. 40, 139–168
Kirchner, J.G. (1967) Thin layer chromatography. In: Technique of organic chemistry, vol. 12, pp. 147–185, Perry, E.S., Weissberger, A., eds. Interscience, New York
Matsushita, J., Kato, Y., Matsuda, K. (1985) Purification and properties of an α-d-xylosidase from Aspergillus niger. J. Biochem. 98, 825–832
O'Neill, R.A., Albersheim, P., Darvill, A.G. (1989) Purification and characterization of a xyloglucan oligosaccharide-specific xylosidase from pea seedlings. J. Biol. Chem. 264, 20430–20437
Reid, J.S.G. (1985) Cell wall storage carbohydrates in seeds. Biochemistry of the seed “gums” and “hemicelluloses”. Adv. Bot. Res. 11, 125–155
Reid, J.S.G., Edwards, M., Dea, I.C.M. (1988) Enzymatic modification of natural seed gums. In: Gums and stabilisers for the food industry, vol. 4, pp 391–398, Phillips, G.O., Wedlock, D.J., Williams, P.A., eds. IRL Press, Oxford
Reis, D., Vian, B., Darzens, D., Roland, J.-C. (1987) Sequential patterns of intramural digestion of galactoxyloglucan in tamarind seedlings. Planta 170, 60–73
Roe, J.H., Rice, E.W. (1948) A photometric method for the determination of free pentoses in animal tissue. J. Biol. Chem. 173, 507–512
Saeman J.F., Buhl, J.L., Harris, E.E. (1945) Quantitative saccharification of wood and cellulose. Ind. Eng. Chem. Anal. Ed. 17, 35–37
Sedmak, J.J., Grossberg, S.E. (1977) A rapid, sensitive and versatile assay for protein using Coomassie Brilliant Blue G250. Anal. Biochem. 79, 544–552
Towbin, H., Staehlin, T., Gordon, J. (1979) Electrophoretic transfer of proteins from polyacrylamide gels to nitrocellulose sheets: procedure and some applications. Proc. Natl. Acad. Sci. USA 76, 9350–9359
Vian, B., McGowan, J., Reid, J.S.G. (1991) Enzyme-gold cytochemistry of seed xyloglucans using two xyloglucan-specific hydrolases. Importance of prior heat-deactivation of the enzymes. Histochem. J., in press
York W.S., Darvill, A.G., Albersheim, P. (1984) Inhibition of 2,4-dichlorophenoxyacetic acid-stimulated elongation of pea stem segments by a xyloglucan oligosaccharide. Plant Physiol. 75, 295–297
York, W.S., Oates J.E., van Halbeek, H., Darvill, A.G., Albersheim, P. (1988) Location of the O-acetyl substituents on a nonasaccharide repeating unit of sycamore extracellular xyloglucan. Carbohydr. Res. 173, 113–132
Zong, N., Yasui, T. (1989) Purification and some properties of an α-d-xylosidase from Bacillus sp. No. 693–1. Agric. Biol. Chem. 53, 187–195
Zong, N., Kamiyama, Y., Yasui, T. (1989) Substrate specificity of Bacillus α-d-xylosidase. Agric. Biol. Chem. 53, 2129–2139
Author information
Authors and Affiliations
Additional information
Much of the work reported in this paper was carried out with the aid of the European Community's “Science Stimulation Action” (Contract No. ST2P-0250-UK), and we wish to record our appreciation of this support.
Rights and permissions
About this article
Cite this article
Fanutti, C., Gidley, M.J. & Reid, J.S.G. A xyloglucan-oligosaccharide-specific α-d-xylosidase or exo-oligoxyloglucan-α-xylohydrolase from germinated nasturtium (Tropaeolum majus L.) seeds. Planta 184, 137–147 (1991). https://doi.org/10.1007/BF00208247
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00208247