Abstract
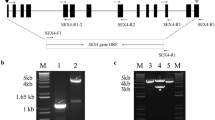
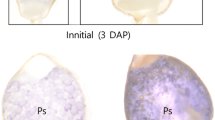
We have isolated and characterised cDNA sequences from a Vicia faba cotyledonary library encoding a plastidic isoform (VfPhol) and a cytosolic isoform (VfPho2) of an α-1,4-glucan phosphorylase (EC 2.4.1.1; Commission on Plant Gene Nomenclature 1994). The Phol isoform is characterized by the presence of a plastidial transit peptide and an 81-residue stretch of additional amino acids in the middle of the polypeptide which are not found in the Pho2 isoform. We define the position of this so-called insertion sequence differently from previous authors. The Phol transcripts were found predominantly in the early seed coat and in cotyledons, and accumulated until the late desiccation phase, whereas Pho2 transcripts were about equally abundant in all investigated tissues. Activity patterns of both enzymes in cotyledons roughly followed mRNA accumulation patterns, with the exception of the late desiccation phases when mRNAs were degraded but enzyme activities remained at high level, even in long-stored seeds. The distinct Phol and Pho2 gene expression patterns in seed coats coincided with the transient accumulation pattern of starch. Similarly, in-situ hybridisation revealed a relationship between Phol gene expression and starch granule formation in developing cotyledons. Expression data and enzyme activity patterns were associated with starch formation during seed development, and could simply reflect a continuous accumulation of enzyme protein, ensuring immediate participation in starch degradation during germination.
Similar content being viewed by others
Abbreviations
- DAF:
-
days after fertilisation
- RACE:
-
rapid amplification of cDNA ends
- RT-PCR:
-
reverse transcriptase-polymerase chain reaction
- VfPhol:
-
plastidic glucan phosphorylase of Vicia faba L.
- VfPho2:
-
cytosolic glucan phosphorylase of Vicia faba L.
References
Borisjuk L, Weber H, Panitz R, Manteuffel R, Wobus U (1995) Embryogenesis of Vicia faba L. Histodifferentiation in relation to starch and storage protein synthesis. J Plant Physiol 147: 203–218
Brisson N, Giroux H, Zollinger M, Camirand A, Simard C (1989) Maturation and subcellular compartmentation of potato starch phosphorylase. Plant Cell 1: 559–566
Camirand A, Benoit St-P, Marineau C, Brisson N (1990) Occurrence of a copia-like transposable element in one of the introns of the potato starch phosphorylase gene. Mol Gen Genet 224: 33–39
Church GM, Gilbert W (1984) Genomic sequencing. Proc Natl Acad Sci USA 81: 1991–1995
Commission on plant gene nomenclature (1994) Nomenclature of sequenced plant genes. Plant Mol Biol Rep 12, CPGN supplement
Conrads J, van Berkel J, Schachtele C, Steup M (1986) Non-chloroplast α-1,4 glucan phosphorylase from pea leaves: characterization and in situ localiztion by indirect immunofluorescence. Biophys Biochim Acta 882: 452–463
Gavel Y, von Heijne G (1990) A conserved cleavage-site motif in chloroplast transit peptides. FEBS Lett 261: 455–458
Goodall G, Filipowicz W (1989) The AU-rich sequences present in introns of plant nuclear pre-mRNAs are required for splicing. Cell 58: 473–483
Hariis N (1976) Starch grain breakdown in cotyledon cells of germinating mung bean seeds. Planta 129: 271–272
Heim U, Weber H, Bäumlein H, Wobus U (1993) A sucrose-synthase gene of Vicia faba L.: Expression pattern in developing seeds in relation to starch synthesis and metabolic regulation. Planta 191: 394–401
Kleczkowski LA, Villand P, Lüthi E, Plsen O-A, Preiss J (1993) Insensitivity of barley endosperm ADP-glucose pyrophosphorylase to 3-phosphoglycerate and orthophosphate regulation. Plant Physiol 101: 179–186
Lin C-T, Yeh K-W, Lee P-D, Su J-C (1991) Primary structure of sweet potato starch phosphorylase deduced from its cDNA sequence. Plant Physiol 95: 1250–1253
Lin C-T, Lin M-T, Chou H-Y, Lee P-D, Su J-C (1995) The gene structure of starch phosphorylase from sweet potato. Plant Physiol 107: 277–278
Matheson NK, Richardson RH (1976) Starch phosphorylase enzymes in developing and germinating pea seeds. Phytochemistry 15: 887–892
Matheson NK, Richardson RH (1978) Kinetic properties of two starch phosphorylases from pea seeds. Phytochemistry 17: 195–200
Mori H, Tanizawa K, Fukui T (1991) Potato tuber type H phosphorylase isozyme. J Biol Chem 266: 18446–18453
Mori H, Tanizawa K, Fukui T (1993) A chimeric α-glucan phosphorylase of plant type L and H isozymes. J Biol Chem 268: 5574–5581
Nakano K, Fukui T (1986) The complete amino acid sequence of potato α-glucan phosphorylase. J Biol Chem 261: 8230–8236
Nakano K, Mori H, Fukui T (1989) Molecular cloning of cDNA encoding potato amyloplast α-glucan phosphorylase and the structure of its transit peptide. J Biochem 106: 691–695
Newgard CB, Hwang PK, Fletterick RJ (1989) The family of glycogen phosphorylases: structure and function. Crit Rev Biochem Mol Biol 24: 69–99
Okita TW (1992) Is there an alternative pathway for starch synthesis? Plant Physiol 100: 560–564
Rochat C, Wuilleme S, Boutin J-P, Hedley CL (1995) A mutation at the rb gene, lowering the ADPGPPase activity, affects storage product metabolism of pea seed coats. J Exp Bot 46: 415–421
Shimomura S, Nagai M, Fukui T (1982) Comparative glucan specificities of two types of spinach leaf phosphorylase. J Biochem 91: 703–717
Smith AM, Martin C (1993) Starch biosynthesis and the potential for its manipulation. In: Grierson D (ed) Biosynthesis and manipulation of plant products. Chapman and Hall, London, pp 1–54
Sonnewald U, Basner A, Greve B, Steup M (1995) A second Ltype isozyme of potato glucan phosphorylase: cloning, antisense inhibition and expression analysis. Plant Mol Biol 27: 567–576
Steup M (1988) Starch degradation. In: Preiss J (ed) Biochemistry of plants, vol 14, Carbohydrates. Academic Press, New York, pp 255–296
Steup M (1990) Starch degrading enzymes. In: Methods Plant Biochem 3: 103–128
Steup M, Latzko E (1979) Intracellular localization of phosphorylases in spinach and pea leaves. Planta 145: 69–75
Steup M, Robeneck H, Melkonian M (1983) In-vitro degradation of starch granules isolated from spinach chloroplasts. Planta 158: 428–436
Van Berkel J, Conrads-Strauch J, Steup M (1991) Glucan-phosphorylase forms in cotyledons of Pisum sativum L.: Localisation, developmental change, in vitro translation, and processing. Planta 185: 432–439
Weber H, Heim U, Borisjuk L, Wobus U (1994) Cell-type specific, coordinate expression of two ADP-glucose pyrophosphorylase genes in relation to starch biosynthesis during seed development of Vicia faba L. Planta 195: 352–361
Yang Yi, Steup M (1990) Polysaccharide fraction from higher plants which strongly interacts with cytosolic phosphorylase isozyme. Plant Physiol 94: 960–969
Author information
Authors and Affiliations
Additional information
We thank S. König (IPK Gatersleben, Germany) for automatic sequencing and Drs. U. Sonnewald (IPK Gatersleben) and C. Martin (Norwich, UK) for making available the potato glucan phosphorylase cDNAs and the pea GBSS I and GBSS II cDNAs, respectively. We also thank Hans Weber (IPK Gatersleben) for stimulating discussions. This work was supported by the BMFT/BMBF. U.W. acknowledges additional support by the Fonds der Chemischen Industrie.
Rights and permissions
About this article
Cite this article
Buchner, P., Borisjuk, L. & Wobus, U. Glucan phosphorylases in Vicia faba L.: cloning, structural analysis and expression patterns of cytosolic and plastidic forms in relation to starch. Planta 199, 64–73 (1996). https://doi.org/10.1007/BF00196882
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00196882