Abstract
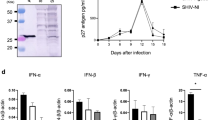
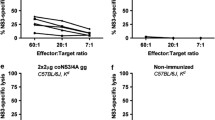
Safe vaccines should optimally induce both cell-mediated and humoral immunity. Recently, it has been shown that protective cytotoxic T cells (CTLs) can be induced not only with live vaccines, but also with recombinant viral proteins. This report shows in C57BL/6 (H-2b) mice that the recombinant nucleoprotein (N) of vesicular stomatitis virus (VSV) induced protective CTLs but no neutralizing antibodies in mice, whereas the recombinant glycoprotein (G) of VSV alone induced neutralizing antibodies but no CTLs. If the N and G of VSV were coinjected, both CTLs and a long-lasting neutralizing IgG response was measurable, demonstrating that mixed vaccines can be used to induce protective CTLs and antibodies with an efficiency comparable to live virus. In an attempt to define optimal conditions for CTL priming, the intravenous, intraperitoneal and subcutanous route of injection were compared. Intravenous injection of recombinant VSV-N induced up to 30 times higher responses than the latter two routes. Finally, we tried to define conditions inducing only CTLs and no antibodies binding to the native protein form, or vice versa, only antibodies and no CTLs. Intravenous injection of boiled VSV-N induced a CTL response but no antibodies specific for the native VSV-N, whereas VSV-N injected subcutanously in incomplete Freund's adjuvant induced high amounts of anti-VSV-N antibodies but virtually no CTLs. The conditions defined here permit vaccines to be designed which would function along selected and defined immunological effector pathways.
Article PDF
Similar content being viewed by others
References
Ada G (1991) Vaccine development: real and imagined dangers. Nature 349:369
Bachmann MF, Rohrer Hoffmann U, Kündig TM, Bürki K, Hengartner H, Zinkernagel RM (1993a) The influence of antigen organisation of B cell responsiveness. Science 262:1448–1451
Bachmann MF, Kündig TM, Kalberer C, Hengartner H, Zinkernagel RM (1993b) Formalin in-activation of vesicular stomatitis virus impairs T cellbut not T help independent B cell responses. J Virol 67:3917–3922
Bachmann MF, Bast C, Hengartner H, Zinkernagel RM (1994a) Immunogenicity of a viral model vaccine after different inactivation procedures. Med Microbiol Immunol 183:95–104
Bachmann MF, Kündig TM, Freer G, Li Y, Bishop DH, Hengartner H, Zinkernagel RM (1994b) Induction of protective cytotoxic T-cells with viral proteins. Eur J Immunol 24:2228–2236
Bachmann MF, Kündig TM, Kalberer CP, Hengartner H, Zinkernage RM (1994c) How many specific B cells are needed to protect against a virus? J Immunol 152:4235–4241
Battegay M, Kyburz D, Hengartner H, Zinkernagel RM (1993) Enhancement of disease by neutralizing antiviral antibodies in the absence of primed antiviral cytotoxic T cells. Eur J Immunol 23:3236–3241
Bevan MJ (1987) Class discrimination in the world of immunology. Nature 325:192–194
Binder D, Kündig TM (1991) Antiviral protection by DC8+ versus CD4+ T cells: CD8+ T cells correlating with cytotoxic activity in vitro are more efficient in antivaccinia virus protection than CD4-dependent interleukins. J Immunol 146:4301–4307
Blanden RV (1974) T cell response to viral and bacterial infection. Transplant Rev 19:56–84
Charan S, Zinkernagel RM (1986) Antibody mediated suppression of secondary IgM response in nude mice against vesicular stomatitis virus. J Immunol 136:3057–3061
Connors M, Kulkarni AB, Firestone C-Y, Holmes KL, Morse HC, III, Sotnikov AV, Murphy BR (1992) Pulmonary histopathology induced by respiratory syncytial virus (RSV) challenge of formalin-inactivated RSV-immunized BALB/c mice is abrogated by depletion of CD4+ T cells. J Virol 66:7444–7451
Corapi WV, Olsen CW, Scott FW (1992) Monoclonal antibody analysis of neutralization and antibody-dependent enhancement of feline infectious peritonitis virus. J Virol 66:6695–6705
Fenton RG, Taub DD, Kwak LW, Smith MR, Longo DL (1993) Cytotoxic T-cell response and in vivo protection against tumor cells harboring activated ras protooncogenes. J Natl Cancer Inst 85:1294–1302
Fields BN (1985) Virology. Raven Press, New York
Fulginiti VA, Eller JJ, Downie AW, Kempe CH (1967) Altered reactivity to measles virus: atypical measles in children previously immunized with inactivated measles virus vaccine. J Am Med Assoc 202:1075–1080
Germaine RN (1986) The ins and outs of antigen processing and presentation. Nature 322:687–689
Germain RN, Margulies DH (1993) The biochemistry and cell biology of antigen processing and presentation. Annu Rev Immunol 11:403–450
Halstead B (1988) The thesis that won't go away. Nature 331:497–498
Halstead SB (1988) Pathogenesis of dengue: challenges to molecular biology. Science 239:476–481
Horstmann DM (1979) Viral vaccines and their ways. Rev Infect Dis 1:502–516
Kapikian AZ, Mitchell RH, Chanock RM, Shvedoff RA, Stewart CE (1969) An epidemiologic study of altered clinical reactivity to respiratory syncytial (RS) virus infection in children previously vaccinated with an inactivated RS virus vaccine. Am J Epidemiol 89:405–421
Kelley JM, Emerson SU, Wagner RR (1972) The glycoprotein of vesicular stomatitis virus is the antigen that gives rise to and reacts with neutralizing antibodies. J Virol 10:1231–1235
Kovacsovics-Bankowski M, Clark K, Benacerraf B, Rock KL (1993) Efficient major histocompatibility complex class I representation of exogenous antigen upon phagocytosis by macrophages. Proc Natl Acad Sci USA 90:4942–4946
Kündig T, Castelmur I, Bachmann MF, Abraham D, Binder D, Hengartner H, Zinkernagel RM (1993) Fewer protective cytotoxic T cell epitopes than T helper cell epitopes on vesicular stomatitis virus. Virol 67:3680–3683
Lefrançois L (1984) Protection against lethal viral infection by neutralizing and nonneutralizing monoclonal antibodies: distinct mechanisms of action in vivo. J Virol 51:208–214
Leist TP, Cobbold SP, Waldmann H, Aguet M, Zinkernagel RM (1987) Functional analysis of T lymphocyte subsets in antiviral host defense. J Immunol 138:2278–2281
Li Y, Luo L, Schubert M, Wagner RR, Kang Y (1993) Viral liposomes released from insect cells infected with recombinant baculovirus expressing the matrix protein of vesicular stomatitis virus. J Virol 67:4415–4420
Mackett M, Yilma T, Rose JK, Moss B (1985) Vaccinia virus recombinants: expression of VSV genes and protective immunization of mice and cattle. Science 227:433–435
Matsuura Y, Possee RD, Overton HA, Bishop DHL (1987) Baculovirus expression vectors: the requirements for high level expression of proteins, including glycoproteins. J Gen Virol 68:1233–1250
McCaren LC, Holland JJ, Syverton JT (1959) The mammalien cell-virus realtionship. I. attachment of poliovirus to cultivated of primate and non-primate origin. J Exp Med 109:475–485
Morrison LA, Lukacher AE, Braciale VL, Fan DP, Braciale TJ (1986) Differences in antigen presentation to MHC class I- and class II-restricted influenza virus specific cytolytic T lymphocyte clones. J Exp Med 163:903–921
Moss B, Flexner C (1987) Vaccinia virus expression vectors. Annu Rev Immunol 5:305–324
Nkowane BM, Wassilak SG, Orenstein WA (1987) Vaccine-associated paralytic poliomyelitis in the United States: 1973 through 1984. JAMA 257:1335–1340
Oehen S, Hengartner H, Zinkernagel RM (1991) Vaccination for disease. Science 251:195–198
Oldstone MBA, Schwimmbeck P, Dyrberg T, Fujinami R (1986) Mimicry by virus of host molecules: implications for autoimmune disease. Prog Immunol 6:787–795
Prince GA, Jenson AB, Hemming VG, Murphy BR, Walsh EE, Horswood RL, Chanock RM (1986) Enhancement of respiratory syncytial virus pulmonary pathology in cotton rats by prior intramuscular inoculation of formalin-inactivated virus. J Virol 57:721–728
Puddington L, Bevan MJ, Lefrancois L (1986) N protein is the predominant antigen recognized by vesicular stomatitis virus-specific cytotoxic T cells. J Virol 60:708–717
Rock KL, Gamble S, Rothstein L (1990) Presentation of exogenous antigen with class I major histocompatibility complex molecules. Science 249:918–921
Rosenthal KL, Zinkernagel RM (1980) Cross-reactive cytotoxic T cells to serologically distinct vesicular stomatis viruses. J Immunol 124:2301–2308
Salk J, Salk D (1977) Control of influenza and poliomyelitis with killed virus vaccines. Science 195:834–847
Schirmbeck R, Zerrahn J, Kuhrober A, Kury A, Deppert W, Reimann J (1992) Immunization with soluble simian virus 40 large T antigen induces a specific response of CD3+ CD4− CD8+ cytotoxic T lymphocytes in mice. Eur J Immunol 22:759–766
Schirmbeck R, Melber K, Mertens T, Reimann J (1994a) Antibody and cytotoxic T-cell responses to soluble hepatitis B virus (HBV) S antigen in mice: implication for the pathogenesis of HBV-induces hepatitis. J Virol 68:1418–1425
Schirmbeck R, Melber K, Mertens T, Reimann J (1994b) Selective stimulation of murine cytotoxic T cell and antibody responses by particulate or monomeric hepatitis B virus surface (S) antigen. Eur J Immunol 24:1088–1096
Scott DW, Gershon RK (1970) Determination of total and mercaptoethanol-resistant antibody in the serum sample. Clin Exp Immunol 6:13–18
Staerz UD, Karasuyama H, Garner AM (1987) Cytotoxic T lymphocytes against a soluble protein. Nature 329:449–451
Vennema H, De Groot RJ, Barbour DA, Dalderup M, Gruffydd-Jones T, Horzinek MC, Spaan WJM (1990) Early death after feline infectious peritonitis virus challenge due to recombinant vaccinia virus immunization. J Virol 64:1407–1409
Wagner RR (1987) The rhabdoviruses. Plenum Press, New York
Yewdell JW, Bennink JR, Mackett M, Lefrancois L, Lyles DS, Moss B (1986) Recognition of cloned vesicular stomatitis virus internal and external gene products by cytotoxic T lymphocytes. J Exp Med 163:1529–1538
Zinkernagel RM, Althage A, Adler B, Blanden RV, Davidson WF, Kees U, Dunlop MBC, Shreffler DC (1977) H-2 restriction of cell-mediated immunity to an intracellular bacterium. Effector T cells are specific for Listeria antigen in association with H-2I region coded selfmarkers. J Exp Med 145:1353–1367
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Bachmann, M.F., Hengartner, H. & Zinkernagel, R.M. Immunization with recombinant protein: conditions for cytotoxic T cell and/or antibody induction. Med Microbiol Immunol 183, 315–324 (1994). https://doi.org/10.1007/BF00196682
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00196682