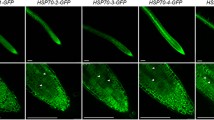
Abstract
We have characterized several heat-shock-induced genes in rice (Oryza sativa L.) and compared their expression under a variety of conditions. Three of these genes, which are analogs of the hsp82/90 family, lie within a cloned 18-kilobase (kb) region of the genome. The middle member of this cluster, designated hsp82B, has been fully sequenced. The gene uses a promoter containing six putative heat-shock elements as well as several unusual sequence motifs including a stretch of 11 thymidines alternating with 11 adenosines. The mRNA for this gene reaches its highest relative level of expression within 120 min after plants are shifted to 42° C; no other conditions induce this gene. By contrast, we found that during heat stress the expression of hsp70 correlates well with increases in internal ion concentrations, and can also be induced by excess salt or ethanol at normal growth temperatures. These results appear to indicate that whereas hsp70 is induced by all stresses that lead to protein denaturation — including heat stress — HSP82 mRNA accumulates only upon heat stress.
Similar content being viewed by others
Abbreviations
- HSF:
-
heat shock factor
- HSP:
-
heat shock protein
- kb:
-
kilobase
References
Ananthan, J., Goldberg, A.L., Voellmy, R. (1986) Abnormal proteins serve as eukaryotic stress signals and trigger the activation of heat shock genes. Science 232, 522–524
Angelidis, C.E., Lazaridis, J., Pagoulatos, G.N. (1991) Constitutive expression of heat-shock protein 70 in mammalian cells confers thermoresistance. Eur. J. Biochem. 199, 35–39
Ashburner, M., Bonner, J.J. (1979) The induction of gene activity in Drosophila by heat shock. Cell 17, 241–254
Baler, R., Welch, W.J., Voellmy, R. (1992) Heat shock gene regulation by nascent polypeptides and denatured proteins: hsp70 as a potential autoregulatory factor. J. Cell Biol. 117, 1151–1159
Bardwell, J.C.A., Craig, E.A. (1987) Eukaryotic M r 83,000 heat shock protein has a homologue in Escherichia coli. Proc. Natl. Acad. Sci. USA 84, 5177–5181
Baulieu, E.-E., Binart, N., Cadepond, F., Catelli, M.-G., Chambraud, B., Garnier, J., Gasc, J.-M., Groyer-Schweizer, G., Rafestin-Oblin, M.-E., Radanyi, C., Redeuilh, G., Renoir, J.-M., Sabbah, M. (1990) Are receptor-associated nuclear proteins associated with the earliest effects of steroid hormones? In: Hormone perception and signal transduction in animals and plants (Proceedings of the Symposia of the Society for Experimental Biology, vol. 44), pp. 3–20, Roberts, J., Kirk, C., Venis, M., eds. Society for Experimental Biology, Cambridge
Beckmann, R.P., Mizzen, L.A., Welch, W.J. (1990) Interactions of Hsp 70 with newly synthesized proteins: implications for protein folding and assembly. Science 248, 850–854
Benjamin, I.J., Horie, S., Greenberg, M.L., Alpern, R.J., Williams, R.S. (1992) Induction of stress proteins in cultured myogenic cells. Molecular signals for the activation of heat shock transcription factor during ischemia. J. Clin. Invest. 89, 1685–1689
Blackman, R.K., Meselson, M. (1986) Interspecific nucleotide sequence comparisons used to identify regulatory and structural features of the Drosophila hsp82 gene. J. Mol. Biol. 188, 499–515
Borkird, C., Simoens, C., Villarroel, R., Van Montagu, M. (1991) Gene expression associated with water-stress adaptation of rice cells and identification of two genes as hsp 70 and ubiquitin. Physiol. Plant. 82, 449–457
Borkovich, K.A., Farrelly, F.W., Finkelstein, D.B., Taulien, J., Lindquist, S. (1989) hsp82 is an essential protein that is required in higher concentrations for growth of cells at higher temperatures. Mol. Cell. Biol. 9, 3919–3930
Cadepond, F., Schweizer-Groyer, G., Segard-Maurel, I., Jibard, N., Hollenberg, S.M., Giguère, V., Evans, R.M., Baulieu, E.-E. (1991) Heat shock protein 90 as a critical factor in maintaining glucocorticosteroid receptor in a nonfunctional state. J. Biol. Chem. 266, 5834–5841
Catelli, M.G., Radanyi, C., Renoir, J.M., Binart, N., Baulieu, E.E. (1988) Definition of domain of HSP 90 interacting with steroid receptors. J. Cell. Biochem. Suppl. 12D, 276
Cheng, L., Hirst, K., Piper, P.W. (1992) Authentic temperature-regulation of a heat shock gene inserted into yeast on a high copy number vector. Influences of overexpression of HSP90 protein on high temperature growth and thermotolerance. Biochim. Biophys. Acta 1132, 26–34
Chirico, W.J., Waters, M.G., Blobel, G. (1988) 70K heat shock related proteins stimulate protein translocation into microsomes. Nature 332, 805–810
Conner, T.W., LaFayette, P.R., Nagao, R.T., Key, J.L. (1990) Sequence and expression of a HSP83 from Arabidopsis thaliana. Plant Physiol. 94, 1689–1695
Craig, E.A., Gross, C.A. (1991) Is hsp70 the cellular thermometer? Trends Biochem. Sci. 16, 135–140
Csermely, P., Kahn, C.R. (1991) The 90-kDa heat shock protein (hsp-90) possesses an ATP binding site and autophosphorylating activity. J. Biol. Chem. 266, 4943–4950
Dalley, B.K., Golomb, M. (1992) Gene expression in the Caenorhabditis elegans dauer larva: developmental regulation of Hsp90 and other genes. Dev. Biol. 151, 80–90
Dellaporta, S.L., Wood, J., Hicks, J.B. (1983) A plant DNA minipreparation: version II. Plant Mol. Biol. Reporter 1, 19–21
Denhardt, D. (1966) A membrane filter technique for the detection of complementary DNA. Biochem. Biophys. Res. Comm. 23, 641–646
Edelman, L., Czarnecka, E., Key, J.L. (1988) Induction and accumulation of heat shock-specific poly(A+) RNAs and proteins in soybean seedlings during arsenite and cadmium treatments. Plant Physiol. 86, 1048–1056
Farrelly, F.W., Finkelstein, D.B. (1984) Complete sequence of the heat shock-inducible HSP90 gene of Saccharomyces cerevisiae. J. Biol. Chem. 259, 5745–5751
Felsheim, R.F., Das, A. (1992) Structure and expression of a heatshock protein 83 gene of Pharbitis nil. Plant Physiol. 100, 1764–1771
Ferguson, D.L., Guikema, J.A., Paulsen, G.M. (1990) Ubiquitin pool modulation and protein degradation in wheat roots during high temperature stress. Plant Physiol. 92, 740–746
Gasc, J.M., Renoir, J.M., Faber, L.E., Delahaye, F., Baulieu, E.E. (1990) Nuclear localization of two steroid receptor-associated proteins, hsp90 and p59. Exp. Cell Res. 186, 362–367
Hanley, B.A., Schuler, M.A. (1988) Plant intron sequences: evidence for distinct groups of introns. Nucleic Acids Res. 16, 7159–7176
Hickey, E., Brandon, S.E., Smale, G., Lloyd, D., Weber, L.A. (1989) Sequence and regulation of a gene encoding a human 89-kilodalton heat shock protein. Mol. Cell. Biol. 9, 2615–2626
Hoagland, D.T., Arnon, D.I. (1938) Water culture method for growing plants without soil. Univ. Calif. Agric. Exp. Stu. Circ. 347, 1–39
Jones, J.D.G., Dunsmuir, P., Bedbrook, J. (1985) High level expression of introduced chimaeric genes in regenerated transformed plants. EMBO J. 4, 2411–2418
Kang, S.-M., Titus, J.S. (1989) Increased proteolysis of senescing rice leaves in the presence of NaCl and KCl. Plant Physiol. 91, 1232–1237
Kobayashi, N., McEntee, K. (1990) Evidence for a heat shock transcription factor-independent mechanism for heat shock induction of transcription in Saccharomyces cerevisiae. Proc. Natl. Acad. Sci. USA 87, 6550–6554
Koning, A.J., Rose, R., Comai, L. (1992) Developmental expression of tomato heat-shock cognate protein 80. Plant Physiol. 100, 801–811
Kouzarides, T., Ziff, E. (1989) Leucine zippers of fos, jun and GCN4 dictate dimerization specificity and thereby control DNA binding. Nature 340, 568–571
Koyasu, S., Nishida, E., Kadowaki, T., Matsuzaki, F., Iida, K., Harada, F., Kasuga, M., Sakai, H., Yahara, I. (1986) Two mammalian heat shock proteins, HSP90 and HSP100, are actinbinding proteins. Proc. Natl. Acad. Sci. USA 83, 8054–8058
Landschulz, W.H., Johnson, P.F., McKnight, S.L. (1988) The leucine zipper: a hypothetical structure common to a new class of DNA binding proteins. Science 240, 1759–1764
Li, G.C., Li, L., Liu, R.Y., Rehman, M., Lee, W.M.F. (1992) Heat shock protein hsp70 protects cells from thermal stress even after deletion of its ATP-binding domain. Proc. Natl. Acad. Sci. USA 89, 2036–2040
Lindquist, S. (1980) Varying patterns of protein synthesis during heat shock; implications for regulation. Dev. Biol. 77, 463–419
Lindquist, S., Craig, E.A. (1988) The heat-shock proteins. Annu. Rev. Genet. 22, 631–677
Loenen, W.A.M., Blattner, F.R. (1983) Lambda Charon vectors (Ch32, 33, 34 and 35) adapted for DNA cloning in recombination-deficient hosts. Gene 26, 171–179
Maniatis, T., Fritsch, E.F., Sambrook, J., eds. (1982) Molecular cloning, a laboratory manual. Cold Spring Harbor Laboratory, Cold Spring Harbor
Maxam, A.M., Gilbert, W. (1980) Sequencing end-labeled DNA with base-specific chemical cleavages. Methods Enzymol. 65, 499–559
Melton, D.A., Krieg, P.A., Rebagliati, M.R., Maniatis, T., Zinn, K., Green, M.R. (1984) Efficient in vitro synthesis of biologically active RNA and RNA hybridization probes from plasmids containing a bacteriophage SP6 promoter. Nucleic Acids Res. 12, 7035–7056
Miyata, Y., Yahara, I. (1992) The 90-kDa heat shock protein, HSP90, binds and protect casein kinase II from self-aggregation and enhances its kinase activity. J. Biol. Chem. 267, 7042–7047
Murashige, T., Skoog, F. (1962) A revised medium for rapid growth and bio assays with tobacco tissue cultures. Physiol. Plant. 15, 473–4197
Nover, L., Neumann, D., Scharf, K.-D., eds. (1990) Heat shock and other stress response systems of plants. Results and problems in cell differentiation, vol. 16. Springer-Verlag, Berlin
Olazabal, U.E., Pfaff, D.W., Mobbs, C.V. (1992) Estrogenic regulation of heat shock protein 90 kDa in the rat ventromedial hypothalamus and uterus. Mol. Cell. Endocrinol. 84, 175–183
Peattie, D.A., Harding, M.W., Fleming, M.A., DeCenzo, M.T., Lippke, J.A., Livingston, D.J., Benasutti, M. (1992) Expression and characterization of human FKBP52, an immunophilin that associates with the 90-kDa heat shock protein and is a component of steroid receptor complexes. Proc. Natl. Acad. Sci. USA 89, 10974–10978
Pelham, H.R.B. (1982) A regulatory upstream promoter element in the Drosophila Hsp 70 heat-shock gene. Cell 30, 517–528
Perdew, G.H. (1988) Association of the Ah receptor with the 90-kDa heat shock protein. J. Biol. Chem. 263, 13802–13805
Picard, D., Khursheed, B., Garabedian, M.J., Fortin, M.G., Lindquist, S., Yamamoto, K.R. (1990) Reduced levels of hsp90 compromise steroid receptor action in vivo. Nature 348, 166–168
Rebbe, N.F., Hickman, W.S., Ley, T.J., Stafford, D.W., Hickman, S. (1989) Nucleotide sequence and regulation of a human 90-kDa heat shock protein gene. J. Biol. Chem. 264, 15006–15011
Rose, D.W., Wettenhall, R.E.H., Kudlicki, W., Kramer, G., Hardesty, B. (1987) The 90-kilodalton peptide of the heme-regulated eIF-2α kinase has sequence similarity with the 90-kilodalton heat shock protein. Biochemistry 26, 6583–6587
Rose, D.W., Welch, W.J., Kramer, G., Hardesty, B. (1989) Possible involvement of the 90-kDa heat shock protein in the regulation of protein synthesis. J. Biol. Chem. 264, 6239–6244
Saiki, R.K., Scharf, S., Faloona, F., Mullis, K.B., Horn, G.T., Erlich, H.A., Arnheim, N. (1985) Enzymatic amplification of β-globin genomic sequences and restriction site analysis for diagnosis of sickle cell anemia. Science 230, 1350–1354
Sanchez, E.R., Toft, D.O., Schlesinger, M.J., Pratt, W.B. (1985) Evidence that the 90-kDa phosphoprotein associated with the untransformed L-cell glucocorticoid receptor is a murine heaft shock protein. J. Biol. Chem. 260, 12398–12401
Scharf, K.-D., Rose, S., Zott, W., Schöff, F., Nover, L. (1990) Three tomato genes code for heat stress transcription factors with a region of remarkable homology to the DNA-binding domain of the yeast HSF. EMBO J. 9, 4495–4501
Scherrer, L.C., Dalman, F.C., Massa, E., Meshinchi, S., Pratt, W.B. (1990) Structural and functional reconstitution of the glucocorticoid receptor-Hsp90 complex. J. Biol. Chem. 265, 21397–21400
Schwartz, J.A., Mizukami, H., Skafar, D.F. (1993) A metal-linked gapped zipper model is proposed for the hsp90-glucocorticoid receptor interaction. FEBS Lett. 315, 109–113
Takahashi, T., Naito, S., Komeda, Y. (1992) Isolation and analysis of the expression of two genes for the 81-kilodalton heat-shock proteins from Arabidopsis. Plant Physiol. 99, 383–390
Tsang, E.W.T., Bowler, C., Hérouart, D., Van Camp, W., Villarroel, R., Genetello, C., Van Montagu, M., Inzé, D. (1991) Differential regulation of superoxide dismutases in plants exposed to environmental stress. Plant Cell 3, 783–792
Vierling, E. (1991) The roles of heat shock proteins in plants. Annu. Rev. Plant Physiol. Plant Mol. Biol. 42, 579–620
von Gromoff, E.D., Treier, U., Beck, C.F. (1989) Three light-inducible heat shock genes of Chlamydomonas reinhardtii. Mol. Cell. Biol. 9, 3911–3918
Wiech, H., Buchner, H., Zimmermann, R., Jakob, Y. (1992) Hsp90 chaperones protein folding in vitro. Nature 358, 169–170
Yanisch-Perron, C., Vieira, J., Messing, J. (1985) Improved M13 phage cloning vectors and host strains: nucleotide sequences of the M13mp18 and pUC19 vectors. Gene 33, 103–119
Ziemiecki, A., Catelli, M.-G., Joab, I., Moncharmont, B. (1986) Association of the heat shock protein hsp90 with steroid hormone receptors and tyrosine kinase oncogene products. Biochem. Biophys. Res. Comm. 138, 1298–1307
Author information
Authors and Affiliations
Additional information
The authors thank R. Villarroel (Laboratorium voor Genetica, Universiteit Gent, Gent, Belgium) for help with the sequencing, both Dr. Van Der Straeten and D. Van den Broeck (Laboratorium voor Genetica, Universiteit Gent, Gent, Belgium) for critical reading of the manuscript, Martine De Cock for typing it, and Karel Spruyt and Vera Vermaercke for drawings and photographs. This work was supported by grants from the Belgian Programme on Interuniversity Poles of Attraction (Prime Minister's Office, Science Policy Programming #38), the Commission of the European Communities TS2-0053-B (GDF), and the Rockefeller Foundation (FR 86058 #59). A.B.G. is indebted to Conselho Nacional de Desenvolvimento Científico e Tecnológico (Brasil; RHAE proc. no. 260015188.1) for a predoctoral fellowship.
Rights and permissions
About this article
Cite this article
Van Breusegem, F., Dekeyser, R., Garcia, A.B. et al. Heat-inducible rice hsp82 and hsp70 are not always co-regulated. Planta 193, 57–66 (1994). https://doi.org/10.1007/BF00191607
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00191607