Summary
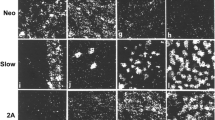
The late fetal development of rat extra-ocular and masticatory muscles was examined by myosin immunohistochemistry. The pattern of slow and neonatal myosin isoform expression in primary and secondary myotubes in these muscles was generally similar to that seen by others in limb muscles. We observed a consistent difference between the Sprague-Dawley and Wistar rats in the degree of maturity reached by all muscles studied at a particular age. In both strains, extra-ocular muscles were also about one day in advance of the masticatory muscles. Thus, secondary myotubes were first seen at E17 in Wistar extraocular muscles, at E18 in Sprague-Dawley extra-ocular muscles and Wistar masticatory muscles, and at E19 in Sprague-Dawley masticatory muscles. There was a strikingly early and complete type differentiation of primary myotubes in extraocular muscles, and tonic myosin first appeared before birth in presumptive extrafusal tonic fibres in the orbital layer of the oculorotatory muscles. Throughout the late fetal period, retractor bulbi was composed of fast myotubes only, but these myotubes were not arranged in classical clusters. In the masticatory muscles at E17/E18 some slow primary myotubes started to express tonic myosin, and these presumptive spindle bag2 fibres were located only in regions of the muscles known to contain spindles in the adult. Presumptive bag1 fibres appeared about a day later (initially without tonic myosin), and in the region of the spindle cluster in anterior deep masseter extrafusal secondary myotube production appeared to be suppressed.
Similar content being viewed by others
References
d'Albis A, Janmot C, Bechet J-J (1986) Comparison of myosins from the masseter muscle of adult rat, mouse and guinea-pig. Persistence of neonatal-type isoforms in the murine muscle. Eur J Biochem 156:291–296
d'Albis A, Janmot C, Conteaux R (1991) Species and muscle type-dependence of perinatal isomyosin transitions. Int J Dev Biol 35:53–56
Antoniou A, Shafi AI, Rowlerson A (1990) Soleus muscle fibre and spindle development in Swiss White and C3H brown mice. J Physiol (Lond) 429:32P
Bondi AY, Chiarandini DJ, Jacoby J (1986) Induction of action potentials by denervation of tonic fibres in rat extraocular muscles. J Physiol (Lond) 374:165–178
Boyd IA (1976) The response of fast and slow nuclear bag fibres and nuclear chain fibres in isolated cat muscle spindles to fusimotor stimulation, and the effect of intrafusal contraction on the sensory endings. Q J Exp Physiol 61:203–254
Butler-Browne GS, Bugaisky LB, Cuenoud S, Schwarz K, Whalen RG (1982) Denervation of newborn rat muscles does not block the appearance of adult fast myosin heavy chain. Nature 299:830–833
Condon K, Silberstein L, Blau HM, Thompson WJ (1990a) Development of muscle fibre types in the prenatal rat hindlimb. Dev Biol 138:256–274
Condon K, Silberstein L, Blau HM, Thompson WJ (1990b) Differentiation of fibre types in aneural musculature of the prenatal rat hindlimb. Dev Biol 138:275–295
Dhoot GK (1988) Identification and changes in the pattern of expression of slow-skeletal-muscle-like myosin heavy chains in a developing fast muscle. Differentiation 37:53–61
Duxson MJ, Usson Y, Harris AJ (1989) The origin of secondary myotubes in mammalian skeletal muscles: ultrastructural studies. Development 107:743–750
Gambke B, Rubinstein NA (1984) A monoclonal antibody to the embryonic myosin heavy chain of rat skeletal muscle. J Biol Chem 259:12092–12100
Harris AJ (1981) Embryonic growth and innervation of rat skeletal muscles. 1. Neural regulation of muscle fibre numbers. Philos Trans R Soc Lond [Biol] 293:257–277
Harris AJ, Fitzsimmons RB, McEwan JC (1989) Neural control of the sequence of expression of myosin heavy chain isoforms in foetal mammalian muscles. Development 107:751–769
Hoh JFY, Hughes S (1989) Immunocytochemical analysis of the perinatal development of cat masseter muscle using anti-myosin antibodies. J Mus Res Cell Motil 10:312–325
Hoh JFY, Hughes S, Hale PT, Fitzsimmons RB (1988) Immunocytochemical and electrophoretic analysis of changes in myosin gene expression in cat limb fast and slow muscles during post-natal development. J Mus Res Cell Motil 9:30–47
Jacoby J, Ko K, Weiss C, Rushbrook JI (1989) Systematic variation in myosin expression along extraocular muscle fibres of the adult rat. J Mus Res Cell Motil 11:25–40
Jones SP, Ridge RMAP, Rowlerson A (1987) The non-selective innervation of muscle fibres and mixed composition of motor units in a muscle of neonatal rat. J Physiol (Lond) 386:377–394
Karlsen K (1965) The location of motor end-plates and the distribution and histological structure of muscle spindles in jaw muscles of the rat. Acta Odont Scand 23:521–547
Kelly AM, Zacks SI (1969) The histogenesis of rat intercostal muscle. J Cell Biol 42:135–153
Kozeka K, Ontell M (1981) The three-dimensional cyto-architecture of developing murine muscle spindles. Dev Biol 87:133–147
Kucera J, Walro JM (1989) Non-uniform expression of myosin heavy chain isoforms along the length of cat intrafusal muscle fibres. Histochemistry 92:291–299
Kucera J, Walro JM (1990) Origin of intra-fusal muscle fibres in the rat. Histochemistry 93:567–580
Kucera J, Walro JM, Reichler J (1989) Role of nerve and muscle factors in the development of rat muscle spindles. Am J Anat 186:144–160
Lennartsson B (1980) Number and distribution of muscle spindles in the masticatory muscles of the rat. J Anat 130:279–288
Maier A (1979) Occurrence and distribution of muscle spindles in masticatory and suprahyoid muscles of the rat. Am J Anat 55:483–506
Maier A, Gambke B, Pette D (1988) Immunohistochemical demonstration of embryonic myosin heavy chains in adult mammalian intrafusal fibres. Histochemistry 88:267–271
Mascarello F, Carpene E, Veggetti A, Rowlerson A, Jenny E (1982) The tensor tympani muscle of cat and dog contains IIM and slow-tonic fibres: an unusual combination of fibre types. J Mus Res Cell Motil 3:363–374
Milburn A (1973) The early development of muscle spindles in the rat. J Cell Sci 12:175–195
Milburn A (1984) Stages in the development of cat muscle spindles. J Embryol Exp Morphol 82:177–216
Morgan DL, Proske U (1984) Vertebrate slow muscle: its structure, pattern of innervation and mechanical properties. Physiol Rev 64:103–169
Narusawa M, Fitzsimmons RB, Izumo S, Nadal-Ginard B, Rubinstein NA, Kelly AM (1987) Slow myosin in developing rat skeletal muscle. J Cell Biol 104:447–459
Navarette R, Vrbova G (1980) Electromyographic activity of rat slow and fast muscles during post-natal development. J Physiol (Lond) 305:33–34P
Ontell M, Kozeka K (1984) The organogenesis of murine striated muscle: a cytoarchitectural study. Am J Anat 171:133–148
Ontell M, Bourke D, Hughes D (1988) Cytoarchitecture of the fetal murine soleus muscle. Am J Anat 181:267–278
Pedrosa F, Thornell LE (1990) Expression of myosin heavy chain isoforms in developing rat muscle spindles. Histochemistry 94:231–244
Pedrosa F, Butler-Browne GS, Dhoot GK, Fischman DA, Thornell LE (1989) Diversity in expression of myosin heavy chain isoforms and M-band proteins in rat muscle spindles. Histochemistry 92:185–194
Pierobon-Bormioli S, Torresan P, Sartore S, Moschini GB, Schiaffino S (1979) Immunohistochemical identification of slow-tonic fibres in human extrinsic eye muscles. Invest Ophthalmol Vis Sci 18:303–306
Pierobon-Bormioli S, Sartore S, Vitadello M, Schiaffino S (1980) “Slow” myosins in vertebrate skeletal muscle. J Cell Biol 85:672–681
Ridge RMAP (1967) The differentiation of conduction velocities of slow twitch and fast twitch muscle motor innervation in kittens and cats. Q J Exp Physiol 52:293–304
Ridge RMAP (1989) Motor unit organisation in developing muscle. Comp Biochem Physiol 93A:115–123
Rowlerson A (1988) Early type-differentiation of intrafusal fibres. In: Hnik P, Soukup T, Vejsada R, Zelena J (eds) Mechanoreceptors: development, structure and function. Plenum Press, New York, pp 45–50
Rowlerson A, Mascarello F, Veggetti A, Carpene E (1983) The fibre-type composition of the first branchial arch muscles in Carnivora and Primates. J Mus Res Cell Motil 4:443–472
Rowlerson A, Gorza L, Schiaffino S (1985) Immunohistochemical identification of spindle fibre types in mammalian muscle using type-specific antibodies to isoforms of myosin. In: Boyd IA, Gladden MH (eds) The muscle spindle. Macmillan, London, pp 29–34
Rowlerson A, Mascarello F, Barker D, Saed H (1988) Muscle spindle distribution in relation to the fibre type composition of masseter in mammals. J Anat 161:37–60
Sartore S, Mascarello F, Rowlerson A, Gorza L, Ausoni S, Vianello M, Schiaffino S (1987) Fibre types in extraocular muscles: a new myosin isoform in the fast fibres. J Mus Res Cell Motil 8:161–172
Scapolo PA, Rowlerson A, Mascarello F, Veggetti A (1991) Neonatal myosin in bovine and pig tensor tympani muscle fibres. J Anat (in press)
Thornell LE, Eriksson PO, Fischman DA, Grove BK, Butler-Browne GS, Virtanen I (1988) Human muscle spindle development. In: Hnik P, Soukup T, Vejsada R, Zelena J (eds) Mechanoreceptors: development, structure and function. Plenum Press, New York, pp 39–44
Vivarelli E, Brown WE, Whalen RG, Cossu G (1988) The expression of slow myosin during mammalian somitogenesis and limb bud differentiation. J Cell Biol 107:2191–2197
Whalen RG, Sell SM, Butler-Browne GS, Schwarz K, Bouveret P, Pinset-Harstrom I (1981) Three myosin heavy-chain isozymes appear sequentially in rat muscle development. Nature 292:805–809
Wieczorek DF, Periasamy M, Butler-Browne GS, Whalen RG, Nadal-Ginard B (1985) Co-expression of multiple myosin heavy chain genes, in addition to a tissue-specific one, in extraocular musculature. J Cell Biol 101:618–629
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Mascarello, F., Rowlerson, A.M. Myosin isoform transitions during development of extra-ocular and masticatory muscles in the fetal rat. Anat Embryol 185, 143–153 (1992). https://doi.org/10.1007/BF00185915
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00185915