Summary
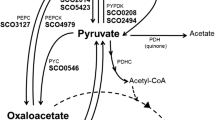
The intracellular α-aminoadipic acid pool in Streptomyces glavuligerus mycelium growing in a starch-peptone medium decreased during the late exponential and stationary phases when cephamycin was being produced; however, the amino acid accumulated extracellularly. Although the specific activity of lysine ɛ-aminotransferase (LAT) decreased during this period, there was no indication that the extracellular α-aminoadipic acid functioned as a precursor reserve for synthesis of the β-lactam antibiotic. Measurement of LAT activity in cultures grown in defined media with starch and various nitrogen sources indicated that the enzyme was synthesized preferentially only during early growth. In its insensitivity to induction by a precursor, and in its susceptibility to carbon catabolite repression, LAT behaved as a secondary metabolic pathway enzyme. Unexpectedly, however, the enzyme increased in specific activity when cultures were supplemented with excess phosphate. Unlike LAT, cadeverine aminotransferase was inducible by lysine or cadaverine and insensitive to phosphate; its features were consistent with a role in the catabolism of lysine by S. clavuligerus.
Similar content being viewed by others
References
Aharonowitz Y, Demain AL (1977) Influence of inorganic phosphate and organic buffers on cephalosporin production by Streptomyces clavuligerus. Arch Microbiol 115:169–173
Aharonowitz Y, Demain AL (1978) Carbon catabolite regulation of cephalosporin production in Streptomyces clavuligerus. Antimicrob Agents Chemother 14:159–164
Braña AF, Wolfe S, Demain AL (1985) Ammonium repression of cephalosporin production by Streptomyces clavuligerus. Can J Microbiol 31:736–743
Braña AF, Wolfe S, Demain AL (1986) Relationship between nitrogen assimilation and cephalosporin synthesis in Streptomyces clavuligerus. Arch Microbiol 146:46–51
Castro JM, Liras P, Cortés J, Martin JF (1985) Regulation of α-aminoadipylcysteinylvaline, isopenicillin N synthetase, isopenicillin N isomerase, and deacetoxycephalosporin C synthetase by nitrogen sources in Streptomyces lactamdurans. Appl Microbiol Biotechnol 22:32–40
Demain AL (1989) Carbon source regulation of idiolite biosynthesis in actinomycetes. In: Shapiro S (ed) Regulation of secondary metabolism in actinomycetes. CRC, Boca Raton, Fla., pp 127–134
Fothergill JC, Guest JR (1977) Catabolism of l-lysine by Pseudomonas aeruginosa. J Gen Microbiol 99:139–155
Goulden SA, Chattaway FW (1968) Lysine control of α-aminoadipate and penicillin synthesis in Penicillium chrysogenum. Biochem J 110:55P-56P
Herbert D, Phipps PJ, Strange RE (1971) Chemical analysis of microbial cells. Methods Microbiol 5B:209–344
Hu W-S, Braña AF, Demain AL (1984) Carbon source regulation of cephem antibiotic production by resting cells of Streptomyces clavuligerus and its reversal by protein synthesis inhibitors. Enzyme Microb Technol 6:155–160
Kern BA, Hendlin D, Inamine E (1980) l-Lysine ɛ-aminotransferase involved in cephamycin C synthesis in Streptomyces lactamdurans. Antimicrob Agents Chemother 17:679–685
Kirkpatrick JR, Doolin LE, Godfrey OW (1973) Lysine biosynthesis in Streptomyces lipmanii: implications in antibiotic biosynthesis. Antimicrob Agents Chemother 4:542–550
Lebrihi A, Lefebvre G, Germain P (1988) A study on the regulation of cephamycin C and expandase biosynthesis by Streptomyces clavuligerus. Appl Microbiol Biotechnol 26:130–135
Lilley C, Clark AE, Lawrence GC (1981) Control of the production of cephamycin C and thienamycin by Streptomyces cattleya NRRL8057. J Chem Technol Biotechnol 31:127–134
Lowry OH, Rosebrough NJ, Farr AL, Randall RJ (1951) Protein measurement with the Folin phenol reagent. J Biol Chem 193:265–275
Lubbe C, Wolfe S, Demain AL (1985) Repression and inhibition of cephalosporin synthetases in Streptomyces clavuligerus by inorganic phosphate. Arch Microbiol 140:317–320
Madduri K, Stuttard C, Vining LC (1989) Lysine catabolism in Streptomyces spp. in primarily through cadaverine: β-lactam producers also make α-aminoadipate. J Bacteriol 171:299–302
Martín JF (1989) Molecular mechanisms for the control by phosphate of the biosynthesis of antibiotics and other secondary metabolites. In: Shapiro S (ed) Regulation of secondary metabolism in actinomycetes. CRC, Boca Raton, Fla., pp 213–237
Rosen H (1957) A modified ninhydrin colorimetric analyses for amino acids. Arch Biochem Biophys 67:10–15
Author information
Authors and Affiliations
Additional information
Offprint requests to: L. C. Vining
Rights and permissions
About this article
Cite this article
Madduri, K., Shapiro, S., DeMarco, A.C. et al. Lysine catabolism and α-aminoadipate synthesis in Streptomyces clavuligerus . Appl Microbiol Biotechnol 35, 358–363 (1991). https://doi.org/10.1007/BF00172726
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/BF00172726