Summary
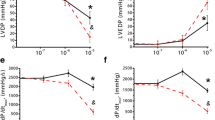
The effects of the new inotropic agents saterinone, sulmazole, UD-CG 212.C1 and milrinone at A1 adenosine receptors and m-cholinoceptors were evaluated in human myocardium from patients with heart failure. At A1 adenosine receptors, all compounds inhibited 3H-DPCPX-binding to ventricular membrane preparations at micromolar concentrations. As judged from the Ki-values, the rank order of potency was saterinone > sulmazole > UD-CG 212.C1 > milrinone. The new inotropic agents also displaced the binding of 3H-QNB at m-cholinoceptors. Except for saterinone, the concentration ranges of mean Ki-values were considerably higher at m-cholinoceptors than at A1 adenosine receptors. The rank order of potency was saterinone > sulmazole > UD-CG 212.Cl > milrinone. Competition of the A1 adenosine receptor agonist R-PIA to 3H-DPCPX-binding showed a biphasic curve with a shallow slope (Hill coefficient nH = 0.63) and revealed two affinity states of the A1 adenosine receptor. In the presence of guanine nucleotides [Gpp(NH)p], the competition curve showed one low affinity class of binding sites and was shifted to the right. In contrast, the competition curves of the new inotropic agents were characterized by a monophasic, steeper slope (mean Hill coefficient nH = 0.98). Guanine nucleotides had no effect. Similar results were obtained with saterinone and carbachol at m-cholinoceptors. Competition with carbachol revealed three affinity states of the m-cholinoceptor, the superhigh affinity binding was reversed by Gpp(NH)p. Competition with saterinone revealed one class of binding sites which was not influenced by Gpp(NH)p. Accordingly, in isolated, electrically driven human atrial trabeculae, the negative inotropic effect of adenosine was antagonized concentration-dependently by saterinone, sulmazole and UD-CG 212.Cl. Similarly the negative inotropic effect of carbachol was antagonized concentration-dependently by saterinone. It is concluded that the new inotropic agents bind to A1 adenosine receptors and that their interaction is of antagonist nature. This mechanism might contribute to their capacity to enhance force of contraction by stimulation of cAMP-formation in addition to phosphodiesterase inhibition. The effects of saterinone may be partially due to antagonism at m-cholinoceptors. This is presumably not the case with the other inotropic agents studied given their low affinity for this receptor.
Similar content being viewed by others
References
Ahn HS, Eardley D, Watkins R, Prioli N (1986) Effects of several newer cardiotonic drugs on cardiac cyclic AMP metabolism. Biochem Pharmacol 35:1113–1121
Belardinelli L, Isenberg G (1983) Isolated atrial myocytes: adenosine and acetylcholine increase potassium conductance. Am J Physiol 244:H734-H737
Berne RM (1963) Cardiac nucleotides in hypoxia: possible role in regulation of coronary bloodflow. Am J Physiol 204:317–322
Böhm M, Brückner R, Hackbarth I, Haubitz B, Linhart R, Meyer W, Schmidt B, Schmitz W, Scholz H (1984) Adenosine inhibition of catecholamine-induced increase on force of contraction in guinea-pig atrial and ventricular heart preparations. Evidence against a cyclic AMP and cyclic GMP-dependent effect. J Pharmacol Exp Ther 230:483–492
Böhm M, Meyer W, Mügge A Schmitz W, Scholz H (1985) Functional evidence for the existence of adenosine receptors in the human heart. Eur J Pharmacol 116:323–326
Böhm M, Diet F, Kemkes B, Erdmann E (1988) Enhancement of the effectiveness of milrinone to increase force of contraction by stimulation of cardiac beta-adrenoceptors in the failing human heart. Klin Wochenschr 66:957–962
Böhm M, Pieske B, Ungerer M, Erdmann E (1989a) Characterization of A1 adenosine receptors in atrial and ventricular myocardium from diseased human hearts. Circ Res 65:1201–1211
Böhm M, Gierschik P, Ungerer M, Erdmann E (1989b) Coupling of adenosine receptors to a pertussis toxin-sensitive G-protein in the human heart. Eur J Pharmacol 172:407–411
Brown JH (1979) Depolarization-induced inhibition of cyclic AMP accumulation: cholinergic-adrenergic antagonism in murine atria. Mol Pharmacol 16:841–850
Brunkhorst D, von der Leyen H, Meyer W, Nigbur R, Schmidt-Schumacher C, Scholz H (1989) Relation of positive inotropic and chronotropic effects of pimobendan, UD-CG 212 Cl, milrinone and other phosphodiesterase inhibitors to phosphodiesterase III inhibition in guinea-pig heart. Naunyn-Schmiedeberg's Arch Pharmacol 339:575–583
Cheng YC, Prusoff WH (1973) Relationship between the inhibition constant (ki) and the concentration of inhibitor which causes 50% inhibition (IC50) of an enzymatic reaction. Biochem Pharmacol 22:3099–3108
Chiba S, Himori N (1975) Different inotropic responses to adenosine on the atrial and ventricular muscle of the dog heart. Jpn J Pharmacol 25:489–491
DeLean A, Hancock AA, Lefkowitz RJ (1982) Validation and statistical analysis of a computer modeling method for quantitative analysis of radioligand binding data for mixtures of pharmacological receptor subtypes. Mol Pharmacol 21:5–16
Dorigo P, Maragno H (1986) Interaction of amrinone with endogenous adenosine in guinea-pig atria. Br J Pharmacol 87:623–629
Earl CQ, Linden J, Weglicki WB (1986) Biochemical mechanisms for the inotropic effect of the cardiotonic drug milrinone. J Cardiovasc Pharmacol 8:864–872
Endoh M, Maruyama M, Taira N (1983) Modification by islet-activating protein of direct and indirect inhibitory actions of adenosine on rat atrial contraction in relation to cyclic nucleotide metabolism. J Cardiovasc Pharmacol 5:131–142
Feldman AM, Cates AE, Veazey WB, Hershberger RE, Bristow MR, Baughman KL, Baumgartner WA, VanDoop C (1988) Increase of the 40,000-mol wt pertussis toxin substrate (G protein) in the failing human heart. J Clin Invest 82:189–197
Fox AC, Reed GE, Glassman E, Kaltman AJ, Silk BB (1974) Release of adenosine from human hearts during angina induced by rapid atrial pacing. J Clin Invest 53:1447–1457
Goyal RK (1989) Muscarinic receptor subtypes. Physiology and clinical implications. N Engl J Med 321:1022–1029
Hazeki O, Ui M (1981) Modification by islet-activating protein of receptor-mediated regulation of cyclic AMP accumulation in isolated rat heart cells. J Biol Chem 256:2856–2862
Herzig JW, Feile K, Rüegg JC (1981) Activating effects of AR-L 115 BS on the Ca2+ sensitive force, stiffness and unloaded shortening velocity (Vmax) in isolated contractile structures from mammalian heart muscle. Arzneimittelforschung 31(1):188–191
Honerjäger P, Schafer-Korting M, Reiter M (1981) Involvement of cyclic AMP in the direct inotropic action of amrinone. Biochemical and functional evidence. Naunyn-Schmiedeberg's Arch Pharmacol 318:112–120
Jakob H, Oelert H, Rupp J, Nawrath H (1989) Functional role of cholinoceptors and purinoceptors in human isolated atrial and ventricular heart muscle. Br J Pharmacol 97:1199–1208
Jochem G, Nawrath H (1983) Adenosine activates a potassium conductance in guinea-pig atrial muscle. Experientia 39:1347–1349
Linden J (1989) Adenosine deaminase for removing adenosine: how much is enough? TIPS 10:260–262
Newman WH, Grossman SJ, Francis MB, Webb JG (1984) Increased myocardial adenosine release in heart failure. J Mol Cell Cardiol 16:577–580
Parsons WJ, Ramkumar V, Stiles GS (1988) The new cardiotonic drug sulmazole is an A1 adenosine receptor antagonist and functionally blocks the inhibitory regulator, G1. Mol Pharmacol 33:441–448
Pieske B, Böhm M, Erdmann E (1988) Effect of saterinone on force of contraction and phosphodiesterase activity in isolated human myocardium. Naunyn-Schmiedeberg's Arch Pharmacol 338: R43
Ransnäs L, Gjörstrup P, Hjalmarsson A, Sjögren CG, Jacobsson B (1986) Muscarinic receptors in mammalian myocardium: effects of atrial and ventricular receptors on phosphatidylinositol metabolism and adenylate cyclase. J Mol Cell Cardiol 18:807–814
Renard M, Jacobs P, Dechamps P, Dresse A, Bernard R (1983) Hemodynamic and clinical response to three-day infusion of sulmazol (AR-L 115 BS) in severe congestive heart failure. Chest 84:408–413
Rockoff JB, Dobson JG (1980) Inhibition by adenosine of catecholamine-induced increase in rat atrial contractility. Am J Physiol 239:H365-H370
Schipke J, Heusch G, Thämer V (1987) Evidence against the adenosine-catecholamine antagonism in the canine heart in situ. Arzneimittelforschung 37:1345–1347
Schrader J, Baumann G, Gerlach E (1977a) Adenosine as inhibitor of myocardial effects of catecholamines. Pflügers Arch 372:29–35
Schrader J, Haddy FJ, Gerlach E (1977b) Release of adenosine, inosine and hypoxanthine from isolated guinea pig heart during hypoxia, flow-autoregulation and reactive hyperemia. Pflügers Arch 369:1–6
Seitelberger R, Schütz W, Schlappack O, Raberger G (1984) Evidence against the adenosine-catecholamine antagonism under in vivo conditions. Naunyn-Schmiedeberg's Arch Pharmacol 325:234–239
Sorota S, Adam LP, Pappano AJ (1986) Comparison of muscarinic receptor properties in hatched chick atrium and ventricle. J Pharmacol Exp Ther 236:602–609
van Meel JCA, Zimmermann R, Diederen W, Erdmann E, Mrwa U (1988) Increase in calcium sensitivity of cardiac myofibrils contributes to the cardiotonic action of sulmazole. Biochem Pharmacol 37:213–220
von der Leyen H, Colberg H, Meyer W, Scholz H, Wenzlaff H (1988) Phosphodiesterase III inhibition by new cardiotonic agents in failing human heart. Naunyn-Schmiedeberg's Arch Pharmacol 338:R40
Weishaar RE, Burrows SD, Kobylarz DC, Quade MM, Evans DB (1986) Multiple molecular forms of cyclic nucleotide phosphodiesterase in cardiac and smooth muscle and in platelets. Biochem Pharmacol 35:787–800
Author information
Authors and Affiliations
Additional information
Send offprint requests to M. Böhm at the above address
Supported by the Deutsche Forschungsgemeinschaft
Rights and permissions
About this article
Cite this article
Ungerer, M., Böhm, M., Schwinger, R.H.G. et al. Antagonism of novel inotropic agents at A1 adenosine receptors and m-cholinoceptors in human myocardium. Naunyn-Schmiedeberg's Arch Pharmacol 341, 577–585 (1990). https://doi.org/10.1007/BF00171739
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00171739