Abstract
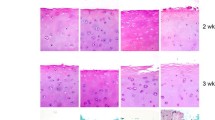
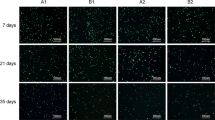
Although transplantation of preserved cartilage has assumed a role of great importance in reconstructive surgery, there are many divergent and contradictory opinions with regard to the outcome of cryopreserved cartilage. This study was formulated to assess the functional state of chondrocytes after cryopreservation. Freeze injury and survival were studied using the trypan blue dye exclusion test, functional assay for cell adhesion and transmission electron microscopy. The methods applied clearly proved that a greater part of the cartilage cells was irreversibly damaged by cryopreservation. Findings demonstrated that cryopreserved cartilage remained non-viable and was not ablt to originate new cartilage. Thus, such cartilage will be reconstruction of parts of the skeleton subject to mechanical stress. The feasibility of cryopreservation techniques for providing vital cartilage substitutes needs further evaluation.
Similar content being viewed by others
References
Adlington P, Anscombe AJ, Phillips JJ (1992) Influence of the mode of preparation on the long-term eficacy of homologous costal cartilage implants. J Laryngol Otol 106:511–517
Amos DB, Bashir H, Boyle W, MacQueen M, Tilikainen A (1969) A simple micro cytotoxicity test. Transplantation 7:220–223
Billingham RE, Medawar PB (1952) The freezing, drying and storage of mammalian skin. J Exp Biol 29:454–457
Bujfa J, Pitzke P, Wilmes E, Hammer C (1992) Culture and cryopreservation of chondrocytes from human cartilage allografting in otolaryngology. ORL 54:80–84
Bujía J, Osete JM, Medina A, Sprekelsen C, Wilmes E, Hammer C (1994) Prolonged vital cartilage graft preservation using tissue culture methods. Eur J Plast Surg 17:20–22
Curran R, Gibson T (1956) The uptake of labelled sulphate by human cartilage cells and its use as a test for viability. Proc R Soc Lond[B] 144:572–576
Gastpar H (1980) Biology of the articular cartilage in health and disease. Schattauer, Stuttgart
Gibson T (1957) Viability of cartilage after freezing. Proc R Soc Br 147:528–531
Gibson T, Davis WB (1958) The distortion of autogenous cartilage grafts: its cause and prevention. Br J Plast Surg 10:257–274
Hagerty RF, Calhoon TB, Lee WH, Cuttino JT (1960) Characteristics of fresh human cartilage. Surg Gynecol Obstet 110:3–9
Hellmich S (1982) Fehler und Gefahren bei der freien Knorpeltransplantation im Gesichtsbereich. HNO 30:140–144
Heyner S (1960) The survival of embryonic mammalian cartilage after freezing to −79°C. J Exp Zool 144:165–177
Kastenbauer ER (1983) Konservierung und Anwendungsmöglichkeiten allogener (homologer) Transplantate im Hals-Nasen-Ohrenbereich HNO 31:371–380
Kawabe N, Yoshinao M (1990) Cryopreservation of cartilage. Int Orthop 14:231–235
Luyet B, Keane J (1955) A critical temperature range apparently characterized by sensitivity of bull semen to high freezing velocity. Biodynamica 7:281–292
Mazur P (1984) Freezing of living cells: mechanisms and implications. Am J Physiol 247:125–142
Mazur P, Leibo SP, Chu EHY (1972) A two-factor hypothesis of freezing injury. Exp Cell Res 71:345–355
Meryman HT, Williams RJ, Donglas MSTJ (1977) Freezing injury from “Solution Effects” and its preservation by natural or artifical cryopreservation. Cryobiology 14:287–302
Schachar NS (1987) Cryopreservation of articular cartilage for transplantation. Sapporo Med J 56:157–161
Searl JC, Kinser DD (1972) Radioautographic study of chondrocytic proliferation in nasal septal cartilage of the 10-day-old rat. J Dent Res 51:812–818
Stevenson S, Dannuci GA, Sharkey NY, Pool RR (1989) The fate of articular cartilage after transplantation of fresh and cryopreserved tissue-antigen-matched and mismatched osteochondrical allografts in dogs. J Bone Joint Surg [Am] 71:1297–1307
Stulberg CC, Soule HD, Berman L (1958) Preservation of human-epithelial-like and fibroblast-like cell strains at low temperatures. Proc Soc Exp Biol Med 98:428–431
Thyberg J, Friberg U (1979) The lysosonal system in endochondral growth. Prog Histochem Cytochem (Monogr) 10 (4)
Thyberg J, Moskalewski S, Friberg U (1975) Electron microscopic studies on the uptake of colloidal thorium dioxide particles by isolated fetal guinea-pig chondrocytes and the distribution of labeled lysosomes in cartilage formed by transplanted chondrocytes. Cell Tissue Res 156:317–344
Weinstein RA, Rubinstein AS, Choukus N (1976) Sequential electron microscopic healing study of grafted palatal mucosa. J Dent Res 55:16–21
Weiss D, Mirow S (1973) An ultrastructural characteristics of osteoarthritis. Fed Proc 32:1459–1466
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Bujía, J., Kremer, D., Wilmes, E. et al. Determination of viability of cryopreserved cartilage grafts. Eur Arch Otorhinolaryngol 252, 30–34 (1995). https://doi.org/10.1007/BF00171437
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00171437