Abstract
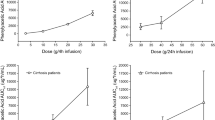
The toxicity of the glutamine antagonist 6-diazo-5-oxo-L-norleucine (DON) administered as a 24 hour infusion has been evaluated. Studies of the clinical pharmacology of the drug have also been performed in 3 patients. The limiting toxicity of the drug was acute nausea, vomiting and diarrhea that was dose dependent in its severity and duration. The maximum tolerated dose was 600 mg/m2 over 24 hours. The other major toxicity was thrombocytopenia that was maximal 7–10 days after the completion of the infusion. The drug does not exhibit renal, hepatic or central nervous system toxicity. DON achieves steady state levels during these infusions and is eliminated by first order kinetics when the infusion is completed (t1/2α = 1.81 h). The principal route of excretion is renal. A starting dose of 400 mg/m2 would be acceptable for Phase II studies of this drug administered on this schedule.
Similar content being viewed by others
References
Levintow L: The glutamyltransferase activity of normal and neoplastic tissues. JNCI 15:347–352, 1954
Ghosh S, Blumenthal HG, Davidson E et al.: Glucosamine metabolism. V. Enzymatic synthesis of glucosamine-6-phosphate. J Biol Chem 235:1265–1273, 1960
Moore EC, Lepage GA: In vivo sensitivity of normal and neoplastic mouse tissues to azaserine. Cancer Res 17:804–808, 1957
Edidinoff ML, Knoll JE, Marano B et al.: Effect of DON (6-diazo-5-oxo-L-norleucine) on incorporation of precursors into nucleic acid pyrimidines. Cancer Res 18:105–109, 1985
Barclay RK, Phillips MA: Effects of 6-diazo-5-oxo-L-norleucine on the incorporation of precursors into nucleic acids. Cancer Res. 22:809–814, 1962
Tiemeier DC, Milman G: Chinese hamster liver glutamine synthetase. J Biol Chem 247:2272–2277, 1972
Overjera A, Houchens DP, Catane R et al.: Efficacy of 6-diazo-5-oxo-L-norleucine against experimental tumors in conventional and nude mice. Cancer Res 39:3220–3224, 1979
Magill GB, Myers WPL, Reilly HC et al.: Pharmacological and initial therapeutic observations on 6-diazo-5- oxo-L-norleucine (DON) in human neoplastic disease. Cancer 10:1138–1150, 1957
Veterans Administration Cancer Chemotherapy Study Group. A clinical study of the comparative effects of nitrogen mustard and DON in patients with bronchogenic carcinoma, Hodgkin's disease, lymphosarcoma, and melanoma, JNCI 22:433–439, 1959
Lim C: Management of choriocarcinoma and related tumors of uterus and testis. Med Clin North Am 45:661–676, 1961
Pittillo RF, Woolley C, Brockman RW et al.: Azotomycin (NSC-56654): biologic fate in mice and man. Cancer Chemother Rep 55:47–52, 1971
Weiss AF, Ramirez G, Grace T et al.: Phase II study of azotomycin (NSC-56654). Cancer Chemother Rep 52:611–614, 1968
Sklaroff RB, Casper ES, Magil GB, Young CW: Phase I study of 6-diazo-5-oxo-L-norleucine (DON) Cancer Treat Rep 64:1247–1251, 1980
Holford NHG: DRUGFUN, in Prophet Public Procedures Notebook (HM Perry, ed) Bolt Beranek and Newman Inc., Cambridge Ma. January, 1982
Knott CD: MLAB, A Mathematical Modelling Tool. Computer Programs Biomed 10:271–280, 1979
Cooney DA, Jayaram HN, Milman HA, Homan ER, Pittillo R, Geran RI, Ryan J, Rosenbluth RJ: DON, CONV and DONV, III. Pharmacologic and Toxicologic Studies. Biochem Pharmacol 25:1859–1870, 1976
Colowick SP, Womack FC: Binding of diffusible molecules by macromolecules: rapid measurement by rate of dialysis. J Biol Chem 244:774–777, 1969
Rubin J, Sorensen S, Schutt AJ, Van Hazel GA, O'Connell MJ, Moertel CG: A phase II study of 6-diazo-5-oxo-L-norleucine (DON, NSC 7365) in advanced large bowel carcinoma. Am J Clin Oncol 6:325–326, 1983
Luc VT, Rosenoff S, Woolley PV: Interaction of mAMSA with serum protein. (Abstract) Proc Am Assoc Cancer Res 22:176, 1981
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Rahman, A., Smith, F.P., Luc, PV.T. et al. Phase I study and clinical pharmacology of 6-diazo-5-oxo-L-norleucine (DON). Invest New Drugs 3, 369–374 (1985). https://doi.org/10.1007/BF00170760
Issue Date:
DOI: https://doi.org/10.1007/BF00170760