Abstract
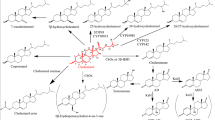
An intensive and systematic investigation of the oxidation of cholesterol (CL) to cholest-4-en-3-one (CN) by Rhodococcus erythropolis was undertaken in the presence of natural and chemically modified cyclodextrins (CDs) in a stirred bioreactor. The biotransformation was found to be strongly affected by the mode of addition of the natural CDs. While simultaneous addition of CL with either β- or λ-CD led to a limited enhancement effect, the microbial oxidation of β- and λ-CD complexes of CL was totally inhibited. In contrast, the alkylated CDs- dimethyl-, trimethyl- and hydroxypropyl-β-CD exhibited a remarkable enhancement of the microbial oxidation, irrespective of their mode of addition. The performance of the alkylated CDs was interpreted in the light of the measured phase solubility diagrams of CL and CN. It was thus shown that unlike the low solubilising power of hydroxypropyl-β-CD, dimethyl- and trimethyl-β-CD at 90 mm each, dissolved 9.3 and 8.7 g/l of CL and CN, respectively. Further investigation focused on the formation of CD complexes with CL and CN, analysed by X-ray powder diffractometry, differential scanning calorimetry and 1H-nuclear magnetic resonance. It was thus shown that β-CD forms a 2:1 CD:CL and CD:CN water-insoluble complexes. A mechanism of the biotransformation in homogeneous and heterogeneous CD media was presented while suggesting a direct interaction of the CD-substrate complex with microbial cells.
Similar content being viewed by others
References
Atrat P, Huller E, Hörhold C (1980) Steroid Transformationen mit immobilisierten Mikroorganismen I. Transformation von Cholesterin zu Cholestenon in organischen Lebensmitteln. Z Allg Mikrobiol 20:79–84
Bar R (1988) Ultrasound enhanced bioprocesses: cholesterol oxidation by Rhodococcus erythropolis. Biotechnol Bioeng 32:655–663
Bar R (1989a) Cyclodextrin-aided bioconversions and fermentations. Trends Biotechnol 7:2–4
Bar R (1989b) Cyclodextrin-aided microbial transformation of aromatic aldehydes by Saccharomyces cerevisiae. Appl Microbiol Biotechnol 31:25–28
Buckland BC, Dunnill P, Lilly MD (1975) The enzymatic transformation of water insoluble reactants in nonaqueous solvents: conversion of cholesterol to cholest-4-en-3-one by a Nocardia sp. Biotechnol Bioeng 17:815–826
Claudy P, Letoffe JM, Germain P, Bastide JP, Bayol A, Blasquez S, Rao RC, Gonzalez B (1991) Physicochemical characterization of cholesterol-beta cyclodextrin inclusion complexes. J Therm Anal 37:2497–2506
Fridrich R, Mehnert W, Fromming KH (1990) Studies of the inclusion compound between β-cyclodextrin and cholesterol. In: Duchene D (ed) Fifth International Symposium on Cyclodextrins (Editions de Santé). March 28–30, Paris, pp 299–302
Frijlink HW (1990) Biopharmaceutical aspects of cyclodextrins. Ph. D. Thesis, Rijksuniversiteit, Groningen, pp 139–155
Goetschel R, Bar R (1992a) Formation of mixed crystals in microbial conversion of sterols and steroids. Enzyme Microb Technol 14:462–469
Goetschel R, Bar R (1992b) Microbial conversions in a liposomal medium. Part 2. Cholesterol oxidation by Rhodococcus erythropolis. Enzyme Microb Technol 14:390–395
Haberland ME, Reynolds IA (1973) Self-association of cholesterol in aqueous solutions. Proc Nat Acad Sci USA 70:2313–2316
Hesselink PGM, Vliet S van, Vries H de, Witholt B (1989) Optimization of steroid side chain cleavage by Mycobacterium sp. in the presence of cyclodextrins. Enzyme Microb Technol 11:398–404
Higuchi T, Connors K (1965) Phase solubility techniques. Adv Anal Chem Instrum 4:117–212
Imai T, Irie T, Otagiri M, Uekama K, Yamasaki M (1984) Comparative study on inclusion complexation of anti-inflammatory drug flurbiprofen with β-cyclodextrin and methylated β-cyclodextrin. J Inclusion Phenom 2:597–604
Saha BC, Zeikus JG (1992) Cyclodextrin degrading enzymes. Starch/Starke 44:312–315
Singer Y, Shity H, Bar R (1991) Microbial transformations in a cyclodextrin medium. Part 2. Reduction of androstenedione to testosterone by Saccharomyces cerevisiae. Appl Microbiol Biotechnol 35:731–737
Smith AG, Brooks CJW (1976) Cholesterol oxidases: properties and application. J Steroid Biochem 7:705–713
Sneath P (1984) Bergey's manual of systematic bacteriology vol. 2. Williams and Wilkins, Baltimore, pp 1472–1478
Szejtli J (1988) Cyclodextrin technology, Kluwer, Dordrecht
Uekama K, Irie T (1987) Pharmaceutical application of methylated cyclodextrin derivatives. In: Duchene (ed) Cyclodextrin and their industrial uses. Editions de Santé, Paris, pp 395–402
Uekama K, Fujinaga T, Hirayama F, Otagiri M, Yamasaki M (1982) Inclusion complexations of steroid hormones with cyclodextrins in water and in solid phase. Int J Pharm 10:1–15
Uvardy NW, Bartho I, Mantos G, Trinn M, Vida ZS, Szejtli J, Stadler-Szoke A, Haven I, Balas-Czurda M (1983) Belgian patent no. 894501 (Chem Abs 99:4069)
Zabaneh M, Bar R (1991) Ultrasound enhanced bioprocesses. II. dehydrogenation of hydrocortisone by Arthrobacter simplex. Biotechnol Bioeng 37:998–113
Author information
Authors and Affiliations
Additional information
Correspondence to: R. Bar
Rights and permissions
About this article
Cite this article
Jadoun, J., Bar, R. Microbial transformations in a cyclodextrin medium. Part 3. Cholesterol oxidation by Rhodococcus erythropolis . Appl Microbiol Biotechnol 40, 230–240 (1993). https://doi.org/10.1007/BF00170372
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00170372