Summary
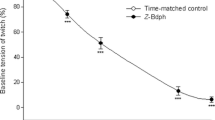
The effect of botulinum C2 toxin was studied on the contractions of the guinea pig ileum myenteric plexus longitudinal muscle preparation. Botulinum C2 toxin inhibited the muscle contraction induced by electrical stimulation (60 V; 0.5 ms; 0.33 Hz) in a time and concentration dependent manner. The inhibitory effect occurred with a time lag of about 1 h, and depended on the presence of both toxin components. After 4 h of incubation with 1.7 µg/ml of component I and 6.7 µ/ml of component II of botulinum C2 toxin, the smooth muscle contraction was inhibited by about 60%. At these toxin concentrations, about 55% of the modifiable smooth muscle actin was ADP-ribosylated. Smooth muscle contraction induced by bradykinin and bethanechol were similarly inhibited. Moreover, the C2 toxin inhibited muscle contraction induced by Bat2+, and by direct muscle membrane depolarization (60 V; 10 ms; 0.33 Hz) after suppression of acetylcholine release by normorphine. Also cytochalasin D inhibited the electrically evoked contraction of the ileum longitudinal muscle. In contrast to botulinum C2 toxin, inhibition of contractility by cytochalasin D occurred without a lag phase, and was reversed by washing off the toxin. In contrast of guinea pig ileum longitudinal muscle, botulinum C2 toxin did not reduce the contraction of the rabbit aortic smooth muscle stimulated by K+-depolarization or noradrenaline.
Similar content being viewed by others
References
Adler KB, Krill J, Alberghini TV, Evans JN (1983) Effect of cytochalasin D on smooth muscle contraction. Cell Motil 3:545- 551
Aktories K, Bärmann M, Chhatwal GS, Presek P (1986) New class of microbial toxins ADP-ribosylates actin. Trends Pharmacol Sci 8:158–160
Aktories K, Ankenbauer T, Schering B, Jakobs KH (1986) ADP-ribosylation of platelet actin by botulinum C2 toxin. Eur J Biochem 161:155–162
Aktories K, Barmann M, Ohishi I, Tsuyama S, Jakobs KH, Habermann E (1986) Botulinum C2 toxin ADP-ribosylates actin. Nature 322:390–392
Böttinger H, Renner K-H, Aktories K (1987) Inhibition of histamine release from rat mast cells by botulinum C2 toxin. Intern Archs Allergy appl Immun 84:380–384
Bradford M (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254
Burgoyne RD, Cheek TR (1987) Reorganisation of peripheral actin filaments as a prelude to exocytosis. Biosci Rep 7:281–288
Cooper JM (1987) Effects of cytochalasin and phalloidin on actin. J Cell Biol 105:1473–1478
Fatigati V, Murphy RA (1984) Actin and tropomyosin variants in smooth muscle. J Biol Chem 259:14383–14388
Gabella G (1984) Structural apparatus for force transmission in smooth muscle. Physiol Rev 64:455–477
Habermann E, Dreyer F (1986) Clostridial neurotoxins: Handling and action at the cellular and molecular level. Curr Top Microbiol Immun 129:93–179
Korn ED (1982) Actin polymerization and its regulation by proteins from nonmuscle cells. Physiol Rev 62:672–737
Kosterlitz HW, Lydon RJ, Watt AJ (1970) The effects of adrenaline, noradrenaline and isoprenaline on inhibitory c-and β-adrenoceptors in the longitudinal muscle of the guinea-pig ileum. Br J Pharmacol 39:398–413
Kuroda M (1985) Change of actin isomers during differentiation of smooth muscle. Biochim Biophys Acta 843:208–213
Lammli UK (1970) Cleavage of structural proteins during the assembly of the head of bacteriophages T4. Nature 227:680–685
Matter K, Dreyer F, Aktories K (1989) Actin involvement in exocytosis from PC12 cells: studies on the influence of botulinum C2 toxin on stimulated noradrenaline release. J Neurochem 52:370 -376
Norgauer J, Kownatzki E, Seifert R, Aktories K (1988) Botulinum C2 toxin ADP-ribosylates actin and enhances O2-production and secretion but inhibits migration of activted human neutrophils. J Clin Invest 82:1376–1382
North RA, Egan TM (1983) Actions and distributions of opioid peptides in peripheral tissues. Br Med Bull 39:71–75
Ohishi I (1983) Response of mouse intestinal loop to botulinum C2 toxin: Enterotoxin activity induced by cooperation of nonlinked protein components. Infect Immun 40:691–695
Ohishi I, Iwasaki M, Sakaguchi G (1980) Purification and characterization of two components of botulinum C2 toxin. Infect Immun 30:668–673
Ohishi I, Miyaki I, Ogura H, Nakamura S (1984) Cytopathic effect of botulinum C2 toxin on tissue-culture cells. FEMS Lett 23:281–284
Omann GM, Allen RA, Bokoch GM, Painter RG, Traynor AE, and Sklar LA (1987) Signal transduction and cytoskeletal activation in neutrophil. Physiol Rev 67:285–322
Reuner KH, Presek P, Boschek CB, Aktories K (1987) Botulinum C2 toxin ADP-ribosylates actin and disorganizes the microfilament network in intact cells. Eur J Cell Biol 43:134–140
Schulz R, Goldstein A (1972) Inactivity of narcotic glucuronides as analgesics and on guinea pig ileum. J Pharmacol Exp Ther 183:404–410
Singer SJ, Kupfer A (1986) The directed migration of eukaryotic cells. Ann Rev Cell Biol 2:337–365
Schering B, Bärmann M, Chhatwal GS, Geipel U, Aktories K (1988) ADP-ribosylation of skeletal muscle and non-muscle actin by Clostridium perfringens iota toxin. Eur J Biochem 171:225–229
Simpson LL (1982) A comparison of the pharmacological properties of Clostridium Botulinum type C1 and C2 toxins. J Pharmacol Exp Ther 223:695–701
Simpson LL (1984) Molecular basis for the pharmacological actions of Clostridium Botulinum type C2 toxin. J Pharmacol Exp Ther 230:665–669
Skalli O, Vandekerckhove J, Gabbiani G (1987) Actin-isoforms pattern as a marker of normal or pathological smooth-muscle and fibroblastic tissues. Differentiation 33:232–238
Vandekerckhove J, Weber, K (1979) The complete amino acid sequence of actins from bovine aorta, bovine heart, bovine fast skeletal muscle and rabbit slow skeletal muscle. Differentiation 14:123–133
Vandekerckhove J, Schering B, Bärmann M, Aktories K (1988) Botulinum C2 toxin ADP-ribosylates cytoplasmic \/γ-actin in arginine 177. J Biol Chem 263:696–700
Weeds A (1982) Actin-binding proteins — regulators of cell architecture and motility. Nature 296: 811–816
Wegner A, Aktories K (1988) ADP-ribosylated actin caps the barbed ends of actin filaments. J Biol Chem 263:13739–13742
Yashuda Y, Cabral AM, Pires JGP, Antonio A (1983) Dissociation of Calcium- and barium-induced contractions of the rat duodenum smooth muscle. Gen Pharmacol 14:643–647
Author information
Authors and Affiliations
Additional information
Send offprint requests to K. Aktories at the above address
Rights and permissions
About this article
Cite this article
Mauss, S., Koch, G., Kreye, V.A.W. et al. Inhibition of the contraction of the isolated longitudinal muscle of the guinea-pig ileum by botulinum C2 toxin: Evidence for a role of G/F-actin transition in smooth muscle contraction. Naunyn-Schmiedeberg's Arch Pharmacol 340, 345–351 (1989). https://doi.org/10.1007/BF00168521
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00168521