Abstract
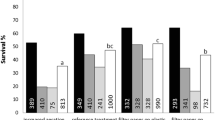
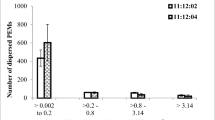
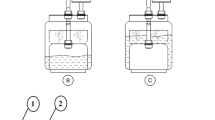
Somatic embryogenesis of pepper, Capsicum annuum var. Ace, was performed in an airlift bioreactor and a magnetically stirred hanging-stirrer-bar bioreactor, each with 1.81 working volume. All stages of embryogenesis, from growth of embryogenic suspension cultures to embryo maturation, were performed in the bioreactor as a series of drain-and-fill batches, keeping the cells and embryos in the bioreactor all the time. When two bioreactors were compared in terms of percentage embryogenesis and visually observed quality of mixing, under different rates of aeration and stirring, the performance of the magnetically stirred bioreactor was better. The effects of inoculum type and inoculum level on the percentage embryogenesis were also investigated. Under the optimum conditions, embryogenesis was 98%, with 57 embryos/ml. Oxygen-uptake rates of cultures in different stages of embryogenesis were different, the highest being in the embryogenic suspension culture and the lowest during embryo maturation.
Similar content being viewed by others
References
Ammirato PV, Styer D (1985) Strategies for large scale manipulation of somatic embryos in suspension culture. In: Zeatlin M, Day P, Hollaender A (eds) Biotechnology in plant science. Academic Press, New York, pp 161–178
Carman J (1988) Somatic embryogenesis in wheat: factors affecting competence induction and differentiation. In Vitro Cell Dev Biol 24:71A
Cazzulino D, Pedersen H, Chin C (1991) Bioreactors and image analysis for scale-up and plant propagation. In: Vasil K (ed) Cell culture and somatic cell genetics of plants, vol 8. Academic Press, New York, pp 147–177
Chen T, Thompson B, Gerson D (1987) In vitro production of alfalfa somatic embryos in fermentation systems. J Ferment Technol 65:353–357
Fowler MW (1982) The large-scale cultivation of plant cells. Prog Ind Microbiol 17:209–220
Halperin W (1967) Population density effects on embryogenesis in carrot cell culture. Exp Cell Res 48:170–173
Kessel RHJ, Carr AH (1972) The effect of dissolved oxygen concentration on growth and differentiation of carrot (Daucus carota) tissue cultures. J Exp Bot 23:996–1007
Murashige T, Skoog F (1962) A revised medium for rapid growth and bioassays with tobacco tissue cultures. Physiol Plant 15:473–497
Pirt SJ (1975) Principles of microbe and cell cultivation. Blackwell, Oxford, p 108
Preil W, Florek P, Wix U, Beck A (1988) Towards mass propagation by use of bioreactors. Acta Hortic (Wageningen) 226:99–105
Scragg AH (1992) Bioreactors for the mass cultivation of plant cells. In: Fowler MW, Warren GS (eds) Moo-Young M (ed in cheif) Plant biotechnology. Comprehensive biotechnology, 2nd suppl. Pergamon, Oxford, p 51
Street HE (1977) Cell (suspension) cultures — techniques. In: Street HE (ed) Plant tissue and cell culture, 2nd edn. University of California Press, Berkeley, Calif, pp 61–102
Stuart DA, Strickland SG, Walker KA (1987) Bioreactor production of alfalfa somatic embryos. Hortscience 22:800–809
Styer D (1985) Bioreactor technology for plant propagation. In: Henke R, Hughes K, Constantin M, Hollaender A (eds) Tissue culture in forestry and agriculture. Plenum, New York, pp 117–130
Takayama S (1991) Mass propagation of plants through shake- and bioreactor-culture techniques. In: Bajaj YPB (ed) Biotechnology in agriculture and forestry, vol 17. High-Tech and micropropagation I. Springer, Berlin, Heidelberg, New York, pp 495–515
Tanaka H (1981) Technological problems in cultivation of plant cell at high density. Biotechnol Bioeng 23:1203–1218
Widholm J (1972) The use of fluorescein diacetate and phenosefranine for determining viability of cultured plant cells. Stain Technol 47:189–194
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Mavituna, F., Buyukalaca, S. Somatic embryogenesis of pepper in bioreactors: a study of bioreactor type and oxygen-uptake rates. Appl Microbiol Biotechnol 46, 327–333 (1996). https://doi.org/10.1007/BF00166225
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00166225