Summary
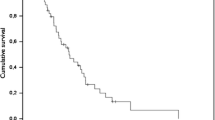
Cisplatin (DDP) is a chemotherapeutic agent that has shown efficacy against primary CNS malignancies. Intraarterial (IA) administration of DDP to patients with brain tumors should produce higher peak levels of drug than intravenous (IV) administration of an identical dose and reduce systemic toxicity. Twelve patients with malignant glioma were entered into the study. All had failed irradiation, 11 had failed IA BCNU. Each patient received IA DDP, 58–100 mg/m2, into the internal carotid artery at four to six week intervals. One of 12 patients had a partial response of 6 months. The remaining 11 patients had progressive disease [10] or severe complications [1]. Toxicity included seizures in four patients, weakness and/or aphasia in four patients, coma in two patients, and visual deterioration in two patients. IA DDP has very limited efficacy in patients with malignant gliomas after failure of nitrosoureas and is associated with an unacceptable level of toxicity. IA DDP may be more effective when used as initial chemotherapy of malignant gliomas.
Similar content being viewed by others
References
Zulch KJ: Brain tumors: Their biology and pathology. 3rd edition. Springer-Verlag Publ., Berlin, 1986
Walker M, Alexander E, Hunt WE, MacCarty CS, Mahaley MS, Mealey J, Norrell HA, Owens G, Ransahoff J, Wilson CB, Gehan EA, Strike TA: Evaluation of BCNU and/or radiotherapy in the treatment of anaplastic glioma. J Neurosurg 49:333–343, 1978
Walker M, Green SB, Byar DP, Alexander E, Batzdorf FU, Brooks WH, Hunt WE, MacCarty CS, Mahaley MS, Meayley, Owens G, Ransahoff J, Robertson JT, Shapiro WR, Smith KR, Wilson CB, Strike TA: Randomized comparisons of radiotherapy and nitrosoureas for the treatment of malignant glioma after surgery. N Eng J Med 303:1323–1329, 1980
Green SB, Byar DP, Walker MD, Pistenmaa DA, Alexander E, Batzdorf U, Brooks WH, Hunt WE, Mealey J, Odom GL, Paoletti P, Ransohoff J, Robertson JT, Selker RG, Shapiro WR, Smith KR, Wilson CB, Strike TA: Comparisons of carmustine, procarbazine, and high-dose methylprednisolone as additions to surgery and radiotherapy for the treatment of malignant gliomas. Cancer Treat Rep 67:121–132, 1983
Crafts DC, Levin VA, Neilson SA: Intracarotid BCNU. A toxicity study in six Rhesus monkeys. Cancer Treat Rep. 60:541–545, 1976
Fenstermacher JP, Cowles AL: Theoretic limitation of intracarotid infusions in brain tumor chemotherapy. Cancer Treat Rep 61:519–526, 1977
Einhorn LH, Donohue J: Cis-diamminedichloroplatinum, vinblastine, and bleomycin combination chemotherapy in disseminated testicular cancer. Ann Int Med 87:293–298, 1977
Yagoda A, Watson RC, Gonzalez-Vitale JC, Grabstold H, Whitmore WF: Cis-dichlorodiammineplatinum (II) in advanced bladder cancer. Cancer Treat Rep 60:917–923, 1976
Wittes RE, Cvitkovic E, Shah J, Gerold FP, Strong EW. Cis-dichlorodiammineplatinum (II) in the treatment of epidermoid carcinoma of the head and neck. Cancer Treat Rep 61:359–366, 1977
Wiltshaw E, Kroner T: Phase II study of cis-dichlorodiammineplatinum (II) in advanced adenocarcinoma of the ovary. Cancer Treat Rep 60:55–60, 1976
Lippard SJ: New chemistry of an old molecule: cis (Pt(NH3)2-CL2). Science 218:1075–1082, 1982
Khan A, McCollough D, Barts F, Sinks LF: Update on use of cisplatin in CNS malignancies. Proc Am Assoc Cancer Res 21:390, 1980
Levanthal BG, Freeman A: Cis-diamminedichloroplatinum — a phase II study in pediatric malignancies. Proc Am Assoc Cancer Res 20:197, 1979
Stewart DJ, Wallace S, Feun L, Leavens M, Young SE, Handel S, Mavligit G, Benjamin RS: A phase I study of intracarotid cis-diamminedichloroplatinum (II) in patients with recurrent malignant intracerebral tumors. Cancer Res 42:2059–2062, 1982
Fenn LG, Wallace S, Stewart DJ, Chuang VP, Yung WA, Leavens ME, Burgess MA, Savaraj N, Benjamin RS, Young SE, Tang RA, Handel S, Mavligit G, Fields WS: Intracarotid infusion of cis-diamminedichloroplatinum in the treatment of recurrent malignant brain tumors. Cancer 54:794–799, 1984
Lehane DE, Bryan RN, Horowitz B, DeSantos L, Ehni G, Zubler MA, Moiel R, Rudolph L, Aldama-Leubbert A, Mahoney D, Harper R: Intra-arterial cisplatinum chemotherapy for patients with primary and metastatic brain tumors. Cancer Drug Delivery 1:69–77, 1983
Kapp JP, Vance RB: Supraophthalmic carotid infusion for recurrent glioma: rationale, technique, and preliminary results for cisplatin and BCNU. J Neuro-oncol 3:5–11, 1985
Vance RB, Kapp JP: Supraophthalmic carotid infusion with low dose cisplatin and BCNU for malignant glioma. J Neuro-oncol 3:287–290, 1986
Phillips TW, Chandler WF, Kindt GW, Ensminger WD, Greenberg HS, Seeger JF, Doan KM, Gyves JW: New implantable continuous administration and bolus dose intracarotid drug delivery system for the treatment of malignant gliomas. Neurosurgery 11:213–218, 1982
Eckman WW, Pattack CS, Fenstermacher JD: A critical evaluation of the principles governing the advantages of IA infusion. J Pharmacokinet Biopharmacokinet 2:257–285, 1974
Levin VA, Kubra PM, Freeman-Dove MA: Pharmacokinetics of intracarotid artery14C-BCNU in the squirrel monkey. J Neurosurg 48:587–593, 1978
Campbell TL, Howell SB, Pfeifle CE, Wung WE, Boostein J: Clinical pharmacokinetics of intra-arterial cisplatin in humans. J Clin Oncol 1:755–762, 1983
Rottenberg DA, Dhawan V, Cooper SC, Strother SC, Alcock N, Ginos JZ: Assessment of the pharmacologic advantage of intra-arterial versus intravenous chemotherapy using N-cisplatin and positron emission tomography (PET). Neurology 37(S1):335, 1987
Cairncross JG, Macdonald DR: Successful chemotherapy for recurrent malignant oligodendroglioma. Ann Neurol 23:360–364, 1988
Yung WKA, Shapiro JR, Shapiro WR: Heterogeneous chemosensitivies of subpopulations of human glioma cells in culture. Cancer Res 42:992–998, 1982
Schnipper LE: Clinical implications of tumor cell heterogeneity. N Engl J Med 314:1423–1430, 1986
Teicher BA, Cucchi CA, Lee JB, Flatow JL, Rosowsky A, Frei E: Alkylating agents: In vitro studies of cross-resistance patterns in human cell lines. Cancer Res 46:4379–4383, 1986
Bracken WM, Klaassen CD: Induction of metallothionein by steroids in rat primary hepatocyte cultures. Tox Appl Pharm 87:381–388, 1987
Endresen L, Schjerven L, Rugstad HE: Tumours from a cell strain with a high content of metallothionein show enhanced resistance against cisdichlorodiammineplatinum. ACTA Pharm Toxicol 55:183–187, 1984
Bermann IJ, Mann MP: Seizures and transient cortical blindness associated with cisplatinum (II) diamminedichloride (PDD) therapy in a 30-year-old man. Cancer 45:764–766, 1980
Kapp JP, Sanford RA: Neurological deficit after carotid infusion of cisplatin and 1,3-bis(2-chloroethyl)-1-nitrosourea (BCNU) for malignant glioma: an analysis of risk factors. Neurosurgery 19:779–783, 1986
Wilding G, Caruso R, Lawrence TS, Ostchega Y, Ballintine EJ, Young RC, Ozols RF: Retinal toxicity after high-dose cisplatin therapy. J Clin One 12:1683–1689, 1985
Kapp JP, Ross RL, Tucker EM: Supraophthalmic carotid infusion for brain tumor chemotherapy. J Neurosurg 58:616–618, 1983
Granowetter L, Rosenstock JG, Packer RJ: Enhanced cis-platinum neurotoxicity in pediatric patients with brain tumors. J Neuro-oncol 1:293–297, 1983
Reddell RR, Kefford RF, Grant JM, Coates AS, Fox RM, Tattersall MHN: Ototoxicity in patients receiving cisplatin. Importance of dose and method of drug administration. Cancer Treat Rep 66:19–23, 1982
Thompson SW, Davis LE, Kornfeld M, Hilgers RD, Standefer JC: Cisplatin neuropathy: clinical, electrophysiology, morphologic, and toxicologic studies. Cancer 54:1269–1275, 1984
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Newton, H.B., Page, M.A., Junck, L. et al. Intra-arterial cisplatin for the treatment of malignant gliomas. J Neuro-Oncol 7, 39–45 (1989). https://doi.org/10.1007/BF00149377
Issue Date:
DOI: https://doi.org/10.1007/BF00149377