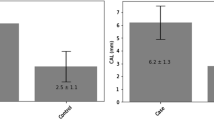
A case-control study of oral cancer was conducted in Beijing, People's Republic of China. The study was hospitalbased and controls were hospital in-patients matched to the cases by age and gender. A total of 404 case/control pairs were interviewed. This paper provides data regarding oral conditions as risk factors for oral cancer, with every patient having an intact mouth examined (pre-operation among cases) using a standard examination completed by trained oral physicians. After adjustment for tobacco smoking and alcohol consumption, poor dentition—as reflected by missing teeth—emerged as a strong risk factor for oral cancer: the odds ratio (OR) for those who had lost 15 – 32 teeth compared to those who had lost none was 5.3 for men and 7.3 for women and the trend was significant (P <0.01) in both genders. Those who reported that they did not brush their teeth also had an elevated risk (OR =6.9 for men, 2.5 for women). Compared to those who had no oral mucosal lesions on examination (OR=1.0), persons with leukoplakia and lichen planus also showed an elevated risk of oral cancer among men and women. Denture wearing per se did not increase oral cancer risk (OR=1.0 for men, 1.3 for women) although wearing metal dentures augmented risk (OR=5.5 for men). These findings indicate that oral hygiene and several oral conditions are risk factors for oral cancer, independently of the known risks associated with smoking and drinking.
Similar content being viewed by others
References
Martin H, Pflueger OH. Cancer of the cheek (buccal mucosa). Arch Surg 1935; 30: 731–47.
Wynder EL, Bross IJ, Feldman R. A study of the etiology of factors in cancer of the mouth. Cancer 1957; 6: 1300–23.
Graham S, Dayal H, Rohrer T, et al. Dentition, diet, tobacco and alcohol in the epidemiology of oral cancer. JNCI 1977; 59: 1611–8.
Silverman S, Gorsky M, Lazada F. Oral leukoplakia and malignant transformation: a follow-up study of 257 patients. Cancer 1984; 53: 563–8.
Browne RM, Camsey MC, Waterhouse JAH. Etiological factors in oral squamous cell carcinoma. Community Dent Oral Epidemiol 1977; 5: 301–6.
Gupta PC, Mehta FS, Daftary DK, et al. Incidence rates of oral cancer and natural history of oral precancerous lesion in a 10-year follow-up study of India villagers. Community Dent Oral Epidemiol 1980; 8: 287–333.
Malaowalla AM, Silverman S, Mani NJ, et al. Oral cancer in 57518 industrial workers of Gujarat. India. A prevalence and follow-up study. Cancer 1976; 37: 1882–6.
Silverman S, Bhargava R, Mani NJ, et al. Malignant transformation and natural history of oral leukoplakia in 57518 industrial workers in Gujarat, India. Cancer 1976; 38: 1790–5.
Pindborg JJ, Jolst O, Renstrup G, et al. Studies in oral leukoplakia: A preliminary report on the period prevalence of malignant transformation in leukoplakia based on a follow-up study of 248 patients. J Am Dent Assoc 1968; 78: 767–71.
Silverman S, Rosen RD. Observations on the clinical characteristics and natural history of oral leukoplakia. J Am Dent Assoc 1968; 76: 772–6.
Boyle P, Zheng T, Macfarlane GJ, et al. Etiology and epidemiology of head and neck cancer. Current Opinion in Oncology 1990; 2: 539–45.
Boyle P, Macfarlane GJ, McGinn R, et al. Epidemiology of head and neck cancer. In: DeVries N and Gluckman J, eds. Second Primary Cancers of Head and Neck. Heidelberg: Springer-Verlag, 1990.
Zheng T, Boyle P, Hu H, et al. Cigarette smoking, alcohol consumption, and risk of oral cancer: a case-control study in Beijing, People's Republic of China. Cancer Causes and Control 1990; 1: 173–9.
World Health Organization. International Classification of Diseases, 9th Revision. Geneva: WHO, 1974.
Mantel N, Haenszel W. Statistical aspects of the analysis of data from retrospective studies of disease. JNCI 1959; 22: 719–48.
Breslow NE, Day NE. Statistical Methods in Cancer Research. Vol I. The Analysis of Case-control Studies. Lyon: International Agency for Research on Cancer, 1980; IARC Sci Pub 32: 248–79.
Marshall J, Graham S, Haughey BP. Smoking, alcohol, dentition and diet in the epidemiology of oral cancer. Submitted for publication.
Kabat GC, Hebert JR, Wynder EL. Risk factors for oral cancer in women. Cancer Res 1989; 49: 2803–6.
Franco EL, Kowalski LP, Oliveira BV, et al. Risk factors for oral cancer in Brazil: a case-control study. Int J Cancer 1989; 43: 992–1000.
Additional information
From the Department of Epidemiology, National Institute of Environmental Health and Engineering, Chinese Academy of Preventive Medicine, Beijing, China (ZT; HH; NS); Unit of Analytical Epidemiology, International Agency for Research on Cancer, Lyon, France (PB; ZT); Beijing Union Hospital (DJ); Cancer Institute, Chinese Academy of Medical Science (JP); Beijing Medical University Stomatological Hospital (MD); Beijing Municipal Stomatological Hospital (SL); University Department of Oral Medicine and Oral Surgery, Bristol Dental Hospital and School, UK (CS); Department of Epidemiology, Harvard School of Public Health (BM; ZT). Address correspondence to Dr Zheng at the Cancer Prevention Research Unit, Yale University, School of Medicine, 26 High Street, New Haven, CT 06510, USA. Dr Zheng was partly supported by a grant from the DuPont Company.
Rights and permissions
About this article
Cite this article
Zheng, T., Boyle, P., Hu, H. et al. Dentition, oral hygiene, and risk of oral cancer: a case-control study in Beijing, People's Republic of China. Cancer Causes Control 1, 235–241 (1990). https://doi.org/10.1007/BF00117475
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00117475