Abstract
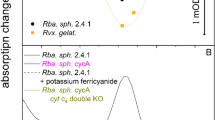
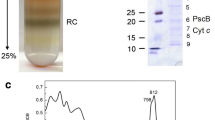
In this paper we propose that the reduction of the bacteriochlorophyl dimer cation (P+) by cytochrome c in the photosynthetic bacteria Rps. viridis and Chromatium vinosum proceeds via two parallel electron transfer (ET) processes from two distinct cytochrome c molecules. The dominating ET process at high temperatures involves the activated oxidation of the high-potential cytochrome c at closest proximity to P, while the dominating low-temperature process involves activationless ET from a low-potential cytochrome c, which is further away from P. The available data for the effects of blocking the low-potential cytochrome c on ET dynamics are consistent with this model, which results in reasonable nuclear reorganization and electronic coupling parameters for the parallel cytochrome oxidation processes. The lack of universality in the cytochrome oxidation in reaction centres of various bacteria is emphasized.
Similar content being viewed by others
References
Alberding N, Austin RH, Beeson K, Chan SS, Eisenstein L, Frauenfelder H and Nordlund TM (1976) Tunneling in ligand binding to heme proteins. Science 192: 1002–1004
Applebury ML, Peter KS and Rentzepid PM (1978) Primary intermediates in the photochemical cycle of bacteriorodopsin. Biophys J 23: 375–393
Bixon M and Jortner J (1986a) On the mechanism of cytochrome oxidation in bacterial photosynthesis. Quantum tunnelling effects revisited. FEBS Lett 200: 303–308
Bixon M and Jortner J (1986b) Does cytochrome oxidation in bacterial photosynthesis manifest tunneling effects? In: Jortner J and Pullman B (eds.) Tunneling, pp 361–371, Dordrecht: D. Reidel
Bixon M and Jortner J (1986c) Coupling of protein modes to electron transfer in bacterial photosynthesis. J Phys Chem 90: 3755–3800
Bixon M and Jortner (1988) Comment on: ‘Cytochrome oxidation in bacterial photosynthesis’, J Chem Phys 89: 3392–3393
Buhks E, Bixon M and Jortner J (1981) Effects of medium modes on electron transfer in biological systems. Chem Phys 55: 41–48
Case GD and Parson WW (1971) Thermodynamics of the primary and secondary photochemical reactions in chromatium. Biochim Biophys Acta 253: 187–202
Chamorovsky SK, Konovenko AA, Remeknnikov SM and Rubin AB (1980) The oxidation rate of high-potential c-type cytochrome in the photochemical reaction center is temperature independent. Biochim Biophys Acta 589: 151–155
Chance B, Kihara T, DeVault D, Hildreth W, Nishimura M and Hiyama T (1969) Temperature-insensitive electron transfer in photosynthetic systems. In: Metzner H (ed) Progress in Photosynthesis Research. Vol. III, pp 1321–1346. Dordrecht: Martinus Nijhoff
Cusanovich MA, Bartsch RG and Kamen MD (1968) Light-induced electron transport in chromatium strain D. II. Light induced absorbance changes in chromatium chromophores. Biochim Biophys Acta 153: 397–417
Deisenhofer J, Epp O, Miki K, Huber R and Michel H (1984) X-ray structure analysis of a membrane protein complex electron density map at 3 Å resolution and a model of the chromophores of the photosynthetic reaction center from Rhodopseudomonas viridis. J Mol Biol 180: 385–398
Deisenhofer J, Epp O, Miki K, Huber R and Michel H (1985) Structure of the protein subunits in the photosynthetic reaction centre of Rhodopseudomonas viridis at 3 Å resolution. Nature 318: 618–624
DeVault D and Chance B (1966) Studies of photosynthetis using a pulsed laser I. Temperature dependence of cytochrome oxidation rate in Chromatium. Evidence for tunneling. Biophys J 6: 825–847
DeVault D (1980) Quantum mechanical tunneling in biological systems. Quart Rev Biophys 13: 387–564
Dogonadze RR and Zakaraya MG (1987) Mechanism of electron transfer between cytochrome and photo-oxidized pigment at the photosynthetic reaction center. Biophysics 32: 7–12
Dracheva SM, Drachev LA, Zaberezhnaya SM, Konstantinov AA, Semenov A.Yu. and Skulachev VP (1986) Spectral, redox and kinetic characteristics of high-potential cytochrome c hemes in Rhodopseudomonas viridis reaction center. FEBS Lett 205: 41–46
Dutton PL, Kihara T, McCray JA and Thornber JP (1971) Cytochrome C553 and bacteriochlorophyll interaction at 77 K in chromatophore and subchromatophore preparation from chromatium D. Biochim Biophys Acta 225: 81–87
Godanskii VI (1986) Quantum chemical reaction in the deep cold. Scientific American 254: 38–44
Gunner MR, Robertson DE and Dutton PL (1986) Kinetic studies of the reaction center protein from Rhodopseudomonas sphaeroides: The temperature and free energy dependence of electron transfer between various quinones in the QA site and the oxidized bacteriochlorophyll dimer. J Phys Chem 90: 3783–3795
Hales B (1976) Temperature dependency of the rate of electron transport as monitor of protein motion. Biophys J 16: 471–480
Hopfield JJ (1974) Electron transfer between biological molecules by thermally activated tunneling. Proc Natl Acad Sci USA 71: 3640–3644
Jortner J (1976) Temperature dependent activation energy for electron transfer between biological molecules. J Chem Phys 64: 4860–4867
Jortner J (1980) Dynamics of the primary events in bacterial photosynthesis. J Am Chem Soc 102: 6676–6686
Kestner NR, Logan J and Jortner J (1974) Thermal electron transfer reactions in polar solvents. J Phys Chem 78: 2148–2166
Knapp EW and Fischer SF (1987) Electron transfer and protein dynamics. J Chem Phys 87: 3880–3887
Kuznetsov AM, Søndergard NC and Ulstrup J (1978) Low temperature electron transfer in bacterial photosynthesis. Chem Phys 29: 383–390
Levich L (1965) Present state of the theory of oxidation-reduction in solution (bulk and electrode reactions). In: Delahay P (ed) Advances in Electrochemistry and Electrochemical Engineering, Vol. 4, p 249, New York: Interscience
Marcus RA (1956) On the theory of oxidation-reduction reactions involving electron transfer. I. J Chem Phys 24: 979–986
Milosavljeric BH and Thomas JK (1986) Photochemistry of compounds in the constrained medium cellulose. 7. The effect of temperature on photoinduced electron transfer from tris (2,2-bipyridine) ruthenium (II) to methyliologen solubilized in cellophane. J Am Chem Soc 108: 2513–2517
Peterson-Kennedy SE, McGourty JL and Hoffman BM (1984) Temperature dependence of long-rante electron transfer in [Zn, FeIII] hybrid hemoglobin. J Am Chem Soc 106: 5010–5012
Prince RC, Leigh JSJr and Dutton PL (1976) Thermodynamic properties of the reaction center of Phodopseudomonas viridis. In vivo measurement of the reaction center bacteriochlorophyll-primary acceptor intermediary electron carrier. Biochim Biophys Acta 400: 622–636
Rosen D, Okamura MY, Abresch EC, Valkirs GE and Feher G (1983) Interaction of cytochrome c with reaction centers of Rhodopseudomonas sphaeroides R-26: Localization of the binding site by chemical cross-linking and immunochemical studies. Biochemistry 22: 335–341
Sarai A (1980) Possible role of protein in photosynthetic electron transfer. Biochim Biophys Acta 589: 71–83
Siebrand W, Wildman TA and Zgierski MZ (1984) Golden rule treatment of hydrogen-transfer reactions. 2. Applications. J Am Chem Soc 106: 4089–4096
Shopes RJ, Levine LMA, Holten D and Wraight CA (1987) Kinetics of oxidation of the bound cytochromes in reaction centers from Rhodopseudomonas viridis. Photosyn Res 12: 165–180
Shopes RJ and Wraight CA (1987) Charge recombination from P+ QA − state in reaction centers from Rhodopseudomonas viridis. Biochim Biophys Acta 893: 409–425
Tiede DM, Leigh JS and Dutton PL (1978) Structural organization of the Chromatium vinosum reaction center associated c-cytochromes. Biochim Biophys Acta 503: 524–544
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Bixon, M., Jortner, J. Cytochrome oxidation in bacterial photosynthesis. Photosynth Res 22, 29–37 (1989). https://doi.org/10.1007/BF00114764
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00114764