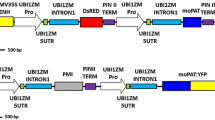
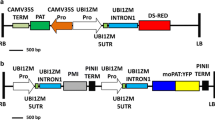
Abstract
The effectiveness of four phosphinothricin (PPT)-based selective agents were evaluated for use in maize transformation: glufosinate, bialaphos, Basta® and Herbiace®. Glufosinate and its commercial formulation, Basta®, were less effective in controlling growth of non-transgenic corn callus than the tripeptide, bialaphos, or its commercial formulation, Herbiace®. Addition of 25 mM l-proline had no significant effect on selection when using bialaphos. However, when l-proline was included with the selective agent glufosinate, selection was inhibited and callus growth was enhanced. At four weeks, callus growth on 0.3, 1.0 and 3.0 mg l-1 glufosinate in the presence of proline was 76, 43, and 21% of control growth, respectively, and in the absence of proline was only 32, 9, and 6% of control growth. Optimized selection protocols for Basta® and bialaphos yielded comparable numbers of transformants. Using these protocols, fertile transgenic plants were regenerated from transformed callus cultures.
Similar content being viewed by others
Abbreviations
- AA:
-
amino acid
- GS:
-
glutamine synthetase
- PAT:
-
phosphinothricin acetyl transferase
- PPT:
-
2-amino-(methylphosphinyl)-butanoic acid (phosphinothricin)
- 2,4-d :
-
2,4-dichlorophenoxyacetic acid
References
Armstrong CL & Green CE (1985) Establishment and maintenance of friable, embryogenic maize callus and the involvement of l-proline. Planta 164: 207–214
Callis J, Fromm M & Walbot V (1987) Introns increase gene expression in cultured maize cells. Genes & Development 1: 1183–1200
Chu CC, Wang CC, Sun CS, Hsu C, Yin KC, Chu CY & Bi FY (1975) Establishment of an efficient medium for anther culture of rice through comparative experiments on the nitrogen sources. Sci. Sinica 18: 659–668
DeBlock M, Botterman J, Vandewiele M, Dockx J, Thoen C, Gosselé V, Movva NR, Thompson C, Van Montagu M & Leemans J (1987) Engineering herbicide resistance in plants by expression of a detoxifying enzyme. EMBO J 6: 2513–2518
De Block M (1990) Factors influencing the tissue culture and the Agrobacterium tumefaciens-mediated transformation of hybrid aspen and poplar clones. Plant Physiol. 93: 1110–1116
Dekeyser R, Claes B, Marichal M, Van Montagu M & Caplan A (1989) Evaluation of selectable markers for rice transformation. Plant Physiol. 90: 217–223
Escandón AS & Hahne G (1991) Genotype and composition of culture medium are factors important in the selection for transformed sunflower (Helianthus annuus) callus. Physiol. Plant. 81: 367–376
Fromm ME, Morrish F, Armstrong C, Williams R, Thomas J & Klein TM (1990) Inheritance and expression of chimeric genes in the progeny of transgenic maize plants. Bio/Technology 8: 833–839
Gordon-Kamm WJ, Spencer TM, Mangano ML, Adams TR, Daines RJ, Start WG, O'Brien JV, Chambers SA, Adams WRJr, Willetts NG, Rice TB, Mackey CJ, Krueger RW, Kausch AP & Lemaux PG (1990) Transformation of maize cells and regeneration of fertile transgenic plants. Plant Cell 2: 603–618
Jefferson RA, Kavanagh TA & Bevan MW (1987) GUS fusions: β-glucuronidase as a sensitive and versatile gene fusion marker in higher plants. EMBO J 6: 3901–3907
Kay R, Chan A, Daly M & McPherson J (1987) Duplication of the CaMV 35S promoter creates a strong enhancer for plants. Science 236: 1299–1302
Kinraide TB (1981) Interamino acid inhibition of transport in higher plants: Evidence for two transport channels with ascertainable affinities for amino acids. Plant Physiol. 68: 1327–1333
Petersen WL, Armstrong CL & Sulc S (1992) Effect of nurse cultures on the production of macro-calli and fertile plants from maize embryogenic suspension culture protoplasts. Plant Cell Rep. 10: 591–594
Songstad DD, Armstrong CL & Petersen WL (1991) AgNO3 increases type II callus production from immature embryos of maize inbred B73 and its derivatives. Plant Cell Rep. 9: 699–702
Songstad DD, Petersen WL & Armstrong CL (1992) Establishment of friable embryogenic (Type II) callus from immature tassels of Zea mays (Poaceae). Amer. J. Bot. 79(7): 761–764
Spencer TM, Gordon-Kamm WJ, Daines RJ, Start WG & Lemaux PG (1990) Bialaphos selection of stable transformants from maize cell culture. Theor. Appl. Genet. 79: 625–631
Stryer L (1988) Biochemistry, Third Edition. W.H. Freeman and Company, New York (pp 590–591)
Tachibana K, Watanabe T, Sekizawa Y & Takematsu T (1986) Action mechanism of bialaphos. J. Pest. Sci. 11: 27–31
Thompson CJ, Movva NR, Tizard R, Crameri R, Davies JE, Lauwereys M & Botterman J (1987) Characterization of the herbicide-resistance gene bar from Streptomyces hygroscopicus. EMBO J 6: 2519–2523
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Dennehey, B.K., Retersen, W.L., Ford-Santino, C. et al. Comparison of selective agents for use with the selectable marker gene bar in maize transformation. Plant Cell Tiss Organ Cult 36, 1–7 (1994). https://doi.org/10.1007/BF00048308
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00048308