Abstract
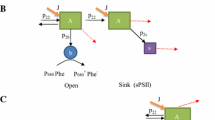
The building up of the two types of reaction centers, PS IIα and PS IIβ, was investigated during the greening of Euglena gracilis Z cells in resting medium. The maximal values in the proportion of PS IIα centers (55%) and in the oxygen evolved per chlorophyll were reached at the outbreak of greening, when accumulation of galactolipids (MGDG and DGDG) rich in unsaturated fatty acids occurred, and when anionic lipids (SQDG and PG) emerged. As the greening progressed, the chlorophyll accumulation corresponded to a secondary enrichment in PS IIβ centers, which built up more rapidly than PS IIα centers; correlatively, a general saturation of the fatty acids constitutive of all lipid classes took place.
Similar content being viewed by others
Abbreviations
- DCMU:
-
3-(3,4-dichlorophenyl)-1,1-dimethylurea
- DGDG:
-
digalactosyldiacylglycerol
- FAME:
-
Tatty acid methyl esters
- HEPES:
-
acide (N-[2-hydroxyethyl]piperazine-N′-[2-ethane sulfonic]
- MGDG:
-
monogalactosyldiacylglycerol
- PC:
-
phosphatidylcholine
- PE:
-
phosphatidylethanolamine
- PG:
-
phosphatidylglycerol
- PQ:
-
plastoquinone
- PS I:
-
Photosystem I
- PS II:
-
Photosystem II
- QA :
-
primary quinone electron acceptor of PS II
- QB :
-
secondary quinone electron acceptor of PS II
- SQDG:
-
sulfoquinovosyldiacylglycerol
References
Albertsson, P-A, Andreasson, E, Svensson, P and Yu, S-G (1992) The domains of the plant thylakoid membrane. In: Barber, J, Guerrero, M-G and Medrano, H (eds) Trends in Photosynthesis Research, pp 45–57 Intercept Ltd, Andover, UK
Anderson, JM and Melis, A (1983) Localization of different photosystems in separate regions of chloroplast membranes. Proc Natl Acad Sci USA 80: 745–749
Armond, PA, Staehelin, LA and Arntzen, CJ (1977) Special relationship of photosystem I, Photosystem II and the light harvesting complex in chloroplast membranes. J Cell Biol 73: 400–418
Block, MA, Dorne, AJ, Joyard, J and Douce, R (1983) Preparation and characterization of membrane fractions enriched in outer and inner envelope membranes from spinach chloroplasts. J Biol Chem 258: 13281–13286
Ban-Dar, Hsu and Jee-Yau, Lee (1991) Characterization of the Photosystem II centers inactive in plastoquinone reduction by fluorescence induction. Photosynth Res 27: 145–150
Calvayrac, R (1970) Relation entre les substrats, la espiration et la structure mitochondriale chez Euglena gracilis Z Arch Microbiol 73: 308–314
Christie, WW (1982) The analysis of complex lipids. In: Wheatson, A (ed) Lipid Analysis, pp 107–134, Pergamon Press, Oxford
Dhal, M and Biswal, B (1990) Excision induced modification in the kinetics of chlorophyll accumulation during greening of clusterbean cotyledons. Biochem Physiol Pflanz (BPP) 186 (1): 11–18
Dorne, AJ, Joyard, J and Block, MA (1985) Localization of phosphatidylcholine in outer envelope membrane of spinach chloroplasts. J Cell Biol 100: 1690–1697
Dorne, AJ, Jouard, J and Douce, R (1990) Do thylakoids really contain phosphatidylcholine? Proc Natl Acad Sci USA 87: 71–74
Dubertret, G and Joliot, P (1974) Structure and organisation of system II photosynthetic units during the greening of dark grown Chlorella mutant. Biochim Biophys Acta 357: 399–411
El Kaoua M (1992) Mise en place des lipides et des centres réactionnels PS IIα et PS IIβ, au cours du verdissement d'Euglena gracilis. Comparaison entre la souche témoin Z et Zt42 et Zt45, cellules ayant subi des chocs de température. Thèse de Doctorat, Université Paris VII
El, Kaou, M, Duval, E and Laval-Martin, D (1991) Long-term effect of diuron on chlorophyllous callus of Bromus erectus: Lipid composition. Z Naturforsch 46c: 569–574
Farineau, J and Laval-Martin, D (1992) Oxygen-evolving system and secondary quinonic acceptors are highly reduced in dark adapted Euglena cells: A thermoluminescence study. Photosynth Res 32: 167–180
Farineau, J and Laval-Martin, D (1995) Characteristics of thermoluminescence bands of Euglena cells belonging to 2 lines presenting different degrees of diuron-resistance. Z Naturforsch 50c: 86–92
Guenther, JE, Nemson, JA and Melis, A (1988) Photosystem stoichiometry and chlorophyll antenna size in Dunaliella salina (green algea). Biochim Biophys Acta 934: 108–117
Henrysson, T and Sundby, C (1990) Characterization of photosystem II in stroma thylakoid membranes. Photosynth Res 25: 107–117
Hodges, M and Barber, J (1986) Analysis of chlorophyll fluorescence induction kinetics exhibited by DCMU-inhibited thylakoids and the origin of α and β centers. Biochim Biophys Acta 848: 239–246
Horton, P and Crose, E (1979) Characterization of two quenchers of chlorophyll fluorescence with different mid-point oxidation-reduction potentials in chloroplasts. Biochim Biophys Acta 545: 188–201
Laval-Martin, D (1985) Spectrophotometric method of controlled pheophytinization for the determination of both chlorophylls and pheophytins in plant extracts. Anal Biochem 149: 121–129
Laval-Martin, D and Troton, D (1990) Chilling-resistance of photosynthetic performances in diuron-adapted Euglena gracilis. Plant Sci 72: 213–222
Laval-Martin, D, Dubertret, G and Calvayrac, R (1977) Photosynthetic properties of a DCMU resistant strain of Euglena gracilis Z. Plant Sci Lett 10: 185–195
Melis, A (1978) Oxidation-reduction potential dependence of the two kinetic components in chloroplast system II primary photochemistry. FEBS Lett 95: 202–206
Melis, A (1985) Functional properties of PS IIβ in spinach chloroplast. Biochim Biophys Acta 808: 334–342
Melis, A and Akoyunoglou, G (1977) Development of the two heterogeneous Photosystem II units in etiolated bean leaves. Plant Physiol 59: 1156–1160
Melis, A and Anderson, JM (1983) Structural and functional organisation of the photosystems in spinach chloroplasts. Antanna size, relative electron-transport capacity, and chlorophyll composition. Biochim Biophys Acta 724: 473–484
Melis, A and Homann, P (1975) Heterogeneity of the photochemical centers in system II of chloroplasts. Photochem Photobiol 23: 343–350
Melis, A and Homann, P (1978) Selective effect of Mg2+ on the photochemistry at one type of reaction center in Photosystem II of chloroplasts. Arch Biochem Biophys 23: 523–530
Salvador, G, Lefort-Tran, M, Nigon, V and Jourdan, F (1971) Structure et évolution du corps prolamellaire dans les proplastes d'Euglena gracilis. Exp Cell Res 64: 457–462
Sommerville, C and Browse, J (1991) Plant lipids: Metabolism, mutants, and membranes. Science 252: 80–87
Thielen, APGM and Van, Gorkum, HJ (1981) Quantum efficiency and antenna size of Photosystem IIα, β and I in tobacco chloroplast. Biochim Biophys Acta 635: 111–120
Troton D (1987) Modifications de la composition lipidique des thylakoides intervenant au cours de l'adaptation d'Euglena gracilis au diuron. Thèse de doctorat, Université Paris VII
Troton, D, Calvayrac, R and Laval-Martin, D (1986a) Adaptative plasticity of the thylakoid lipids in diuron-resistant Euglena gracilis. Biochim Biophys Acta 878: 71–78
Troton, D, Calvayrac, R, Phan-Thi, AT and Laval-Martin, D (1986b) Modification of thylakoid lipids in Euglena gracilis during diuron-adaptation. Photochem 25: 393–399
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Kaoua, M.E., Laval-Martin, D.L. Evolution of PS IIα and PS IIβ centers during the greening of Euglena gracilis Z: Correlations with changes in lipid content. Photosynth Res 43, 155–163 (1995). https://doi.org/10.1007/BF00042973
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00042973