Abstract
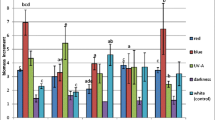
Anthocyanins isolated and characterized from the wild carrot suspension cultures used here were 3-O-β-D-glucopyranosyl-(1→6)-[β-D-xylopyranosyl-(1→2)-]β-D<-galactopyranosylcyanidin (1), 3-O-[β-D- xylopyranosyl-(1→2)-β-D-galactopyranosyl]cyanidin (2), 3-O-(6-O-sinapoyl)-β-D-glucopyranosyl-(1→6)-[β-D- xylopyranosyl-(1→2)-]β-D-galactopyranos ylcyanidin (3), 3-O-(6-O-feruoyl)-β-D-glucopyranosyl-(1→6)-[β- D-xylopyranosyl-(1→2)-]β-D-galactopyranosylcyanidin (4), 3-O-(6-O-coumaroyl)-β-D-glucopyranosyl-(1→6)- [β-D-xylopyranosyl-(1→2)-]β-D-galactopyrano sylcyanidin (5), 3-O-[6-O-(3,4,5-trimethoxycinnamoyl)]-β- D-glucopyranosyl-(1→6)-[β-D-xylopyranosyl-(1→2)-]β-D-galactopyranosylcyanidin (6), 3-O-[6-O-(3,4-dime- thoxycinnamoyl)]-β-D-glucopyranosyl-(1→6)-[β-D-xylopyranosyl-(1→2)-]β-D-galactopyranosylcyanidin (7), 3-O-[(6-O-sinapoyl)-β-D-glucopyranosyl-(1→6)-β-D-galactopyranosyl]cyanidin (8), and 3-O-(β-D-galactopyranosyl)cyanidin (9). Except when cinnamic acids were provided in the culture medium, the major anthocyanin present in the two clones examined was 2. When the naturally occurring and some non-naturally occurring cinnamic acids were provided individually in the medium, 1 and 2 were minor components and the anthocyanin acylated with the supplied cinnamic acid, namely 3, 4, 5, 6, or 7 was the major anthocyanin present in the tissue. When caffeic acid was provided the major anthocyanin in the tissue was 4, thereby suggesting that the caffeic acid was methylated before its use in anthocyanin biosynthesis. Other cinnamic acids supplied had limited effects on the anthocyanins accumulated and appeared not to result in the accumulation of new anthocyanins by the tissue. Thus the tissue can use some but not all analogues of sinapic acid to acylate anthocyanins. Additional anthocyanins were detected in extracts of the wild carrot tissue cultures using mass spectrometry (both MS/MS and HPLC/MS). The additional compounds detected have also been found in cultures of black carrot, an Afghan cultivar of Daucus carota ssp. sativa and the flowers of wild carrot giving no evidence for qualitative differences in the anthocyanins synthesized by subspecies, cell cultures from subspecies, or clones from cell cultures. There are major differences in the amounts of individual anthocyanins found in cultures from different subspecies and in different clones from cell cultures. Here anthocyanins without acyl groups were usually found in the tissues and their accumulation is discussed. On the basis of the structures of the isolated anthocyanins, a likely pathway from cyanidin to the accumulated anthocyanins is proposed and discussed.
Similar content being viewed by others
Abbreviations
- Sin:
-
sinapoyl
- Fer:
-
feruoyl
- 4-Coum.:
-
4-coumaroyl
- 3,4-MeO2Cin:
-
3,4-dimethoxyeinnamoyl
- 3,4,5-MeO3Cin:
-
3,4,5-trimethoxycinnamoyl
- Cya:
-
cyanidin
References
Cheng CL, Wetherell DF & Dougall DK (1985) 4-Coumarate: CoA ligase in wild carrot cell culture clones which accumulate different amounts of anthocyanin. In: Neuman et al. (Eds.) Primary and Secondary Metabolism of Plant Cell Cultures, (pp. 87–98). Springer-Verlag, Berlin, Heidelberg
Do CB & Cormier F (1991) Accumulation of peonidin 3-glucoside enhanced by osmotic stress in grape (Vitis vinifera L.) cell suspension. Plant Cell Tiss. Org. Cult. 24: 49–54
Dougall DK (1989) Sinapic acid stimulation of anthocyanin accumulation in carrot cell cultures. Plant Science 60: 259–262
Dougall DK, Johnson JM & Whitten GH (1980) A clonal analysis of anthocyanin accumulation by cell cultures of wild carrot. Planta 149: 292–297
Dougall DK & Vogelien DL (1990) Anthocyanin yields of clonal wild carrot cell cultures: Effects of serial cloning plus selection for high or low yield. Plant Cell Tiss. Org. Cult. 23: 79–91
Dougall DK & Weyrauch KW (1980a) Abilities of organic acids to support growth and anthocyanin accumulation by suspension cultures of wild carrot cells using ammonium as the sole nitrogen source. In Vitro 16: 969–975
Dougall DK & Weyrauch KW (1980b) Growth and anthocyanin production by carrot suspension cultures grown under chemostat conditions with phosphate as the limiting nutrient. Biotechnology and Bioengineering XXII, 337–352
Folch J, Lees M & Stanley GHS (1957) A simple method for the isolation and purification of total lipids from animal tissues. J. Biol. Chem. 226: 497–509
Gläßgen WE & Seitz HU (1992) Acylation of anthocyanins with hydroxycinnamic acids via 1-O-acylglucosides by protein preparations from cell cultures of Daucus carota L. Planta 186: 582–585
Gläßgen WE, Hofmann R, Emmerling M, Neumann DG & Seitz HU (1992a) Structure elucidation of saccharides in anthocyanins and flavonols by means of methylation analysis and gas chromatography. J. Chromatography 598: 81–87
Gläßgen WE, Seitz HU & Metzger JW (1992b) High-performance liquid chromatography/electrospray mass spectrometry and tandem mass spectrometry of anthocyanins from plant tissues and cell cultures of Daucus carota. Biological Mass Spectrometry 21: 271–277
Gläßgen WE, Wray V, Strack D, Metzger JW & Seitz HU (1992c) Anthocyanins from cell suspension cultures of Daucus carota. Phytochemistry 31: 1593–1601
Gross GG (1981) Phenolic acids. In: Con EE (Ed.) The Biochemistry of Plants Vol 7, Secondary Plant Products (pp. 301–316) Academic Press, New York
Halaweish FT & Dougall DK (1990) Sinapoyl glucose synthesis by extracts of wild carrot cell cultures. Plant Science 71: 179–184
Harborne JB (1976) A unique pattern of anthocyanins in Daucus carota and other umbelliferae. Biochemical Systematics and Ecology 4: 31–35
Harborne JB, Mayer AM & Bar-Nun N (1983) Identification of the major anthocyanin of carrot cells in tissue culture as cyanidin 3- (sinapoylxylosylglucosylgalactoside). Naturforsch. 38C: 1055–1056
Hopp W & Seitz HU (1987) The uptake of acylated anthocyanin into isolated vacuoles from a cell suspension culture of Daucus carota. Planta 170: 74–85
Sawardeker JS, Sloneker JH & Jeanes A (1965) Quantitative determination of monosaccharides as their alditol acetates by gas liquid chromatography. Analytical Chemistry 37: 1602–1605
Terahara N, Saito N, Toki K, Sakata Y & Honda T (1992) Cyanidin 3-lathyroside from berries of Fatsia japonica. Phytochemistry 31: 1446–1448
Yoshida K, Kondo T, Kameda K, Kawakishi S, Lubag AJM, Mendoza EMT & Goto T (1991) Structures of alatanin A, B and C isolated from edible purple yam Dioscorea alata. Tetrahedron Letters 32: 5575–5578
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Baker, D.C., Dougall, D.K., Gläßgen, W.E. et al. Effects of supplied cinnamic acids and biosynthetic intermediates on the anthocyanins accumulated by wild carrot suspension cultures. Plant Cell Tiss Organ Cult 39, 79–91 (1994). https://doi.org/10.1007/BF00037595
Issue Date:
DOI: https://doi.org/10.1007/BF00037595