Abstract
Two green algal species, Chlamydomonas reinhardtii and Scenedesmus obliquus, exhibited a relative maximum during the decay of luminescence, when adapted to low CO2 conditions that was not observed in high CO2 adapted cells.
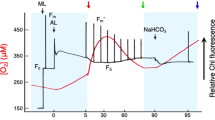
From the kinetics of transient changes in the level of dark fluorescence, after illumination and parallel to the luminescence maxima, it was concluded that the maximum in Scenedesmus was mainly related to a decrease in nonphotochemical quenching, whereas in Chlamydomonas the maximum was mainly related to a dark reduction of the primary PS II acceptor QA.
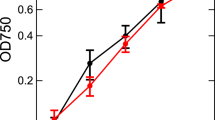
ATP/ADP ratios from low CO2 adapted Scenedesmus showed transient high levels after a dark/light transition that was not observed in high CO2 adapted cells. After 30 s of illumination the ATP/ADP ratios however stabilized at the same steady state level as in high CO2 adapted cells.
Dark addition of HCO3 - to low CO2 adapted cells of Chlamydomonas resulted in a rapid transient quenching of luminescence that was not observed in low CO2 adapted cells of neither species.
It is concluded that the luminescence maxima present in both low CO2 adapted Scenedesmus and Chlamydomonas reflect adaptation of the cells to low CO2 conditions. It is further suggested that the difference in mechanistic origin of luminescence maxima in the two species reflects differences in adaptation.
Similar content being viewed by others
Abbreviations
- ADP:
-
adenosine-diphosphate
- ATP:
-
adenosine-triphosphate
- Ci :
-
inorganic carbon
- FD :
-
dark fluorescence recorded under dark adapted conditions
- F0 :
-
fluorescence with all reaction centers open
- FV:
-
variable fluorescence
- PS I:
-
photosystem I
- PS II:
-
photosystem II
- QA :
-
the first quinone acceptor of PS II
References
Badger MR, Kaplan A and Berry JA (1980) Internal inorganic carbon pool of Chlamydomonas reinhardtii. Plant Physiol 66: 407–413
Bertsch WF and Azzi JR (1965) A relative maximum in the decay of long term delayed light emission from the photosynthetic apparatus. Biochim Biophys Acta 94: 15–26
Bilger W and Schreiber V (1986) Energy-dependent quenching of dark-level chlorophyll fluorescence in intact leaves. Photosynth Res 10: 303–308
Bishop NI (1971) Preparations and properties of mutants: Scenedesmus. Methods Enzymol 23: 372–408
Björn LO (1971) Far red induced, long lived afterglow from photosynthetic cells. Size of afterglow unit and paths of energy accumulation and dissipation. Photochem Photobiol 13: 5–20
Coleman JR, Berry JA, Togasaki RK and Grossman AR (1984) Identification of extracellular carbonic anhydrase of Chlamydomonas reinhardtii. Plant Physiol 76: 472–477
Crofts AR, Wraight CA and Fleischmann DE (1971) Energy conservation in the photochemical reactions of photosynthesis and its relation to delayed fluorescence. FEBS Lett 15 (2): 89–100
Desai TS, Rane SS, Tatake VG and Sane PV (1983) Identification of far-red induced relative increase in the decay of delayed light emission from photosynthetic membranes with thermoluminescence peak V appearing at 321 K. Biochim Biophys Acta 724: 485–489
Heil WG and Senger H (1986) Thylakoid-protein phosphorylation during the life cycle of Scenedesmus obliquus in synchronous culture. Planta 167: 233–239
Krause GH and Weis E (1984) Chlorophyll fluorescence as a tool in plant physiology. II Interpretation of fluorescence signals. Photosynth Res 5: 139–157
Lavorel J (1975) Luminescence. In: Govindjee (ed) Bioenergetics of Photosynthesis, pp 223–317. New York: Academic Press
Lundin A, Rickardsson A and Thore A (1976) Continuous monitoring of ATP-converting reactions by purified firefly luciferase. Anal Biochem 75: 611–620
Malkin S (1977) Delayed luminescence. In: JBarber (ed) Primary Processes of Photosynthesis, pp 349–431. North-Holland: Biomedical Press, Elsevier
Mellvig S and Tillberg J-E (1986) Transient peaks in the delayed luminescence from Scenedesmus obtusiculus induced by phosphorous starvation and carbon dioxide deficiency. Physiol Plantarum 68: 180–188
Miller AG and Canvin DT (1987) The quenching of chlorophyll a fluorescence as a consequence of the transport of inorganic carbon by the cyanobacterium Syneochoccus UTEX 625. Biochim Biophys Acta 894: 407–413
Ogawa T, Omata T, Miyano A and Inoue Y (1985) Photosynthetic reactions involved in the CO2-concentration mechanism in the cyanobacterium Anacystis nidulans. In: Lucas WJ and Berry JA (eds), Inorganic Carbon Uptake by Aquatic Photosynthetic Organisms. American Society for Plant Physiologists
Palmqvist K, Sjöberg S and Samuelsson G (1988) Induction of inorganic carbon accumulation in the unicellular green algae Scenedesmus obliquus and Chlamydomonas reinhardtii. Plant Physiol 87: 437–442
Palmqvist K, Sundblad L-G, Samuelsson G and Sundbom E (1986) A correlation between changes in luminescence decay kinetics and the appearance of a CO2-accumulating mechanism in Scenedesmus obliquus. Photosynth Res 10: 113–123
Quick WP and Horton P (1984) Studies on the induction of chlorophyll fluorescence in barley protoplasts. II. Resolution of fluorescence quenching by redox state and transthylakoid pH gradient. Proc R Soc Lond B220: 371–382
Rubin AB, Fokht AS and Venediktov PS (1966) Investigation of the kinetics of attenuation of the afterglow of photosynthesizing organism. Biofizika 11: 299–305
Schmidt W and Senger H (1987a) Long term delayed luminescence in Scenedesmus obliquus. I. Spectral and kinetic properties. Biochim Biophys Acta 890: 15–22
Schmidt W and Senger H (1987b) Long-term delayed luminescence in Scenedesmus obliquus. II. Influence of exogenous factors. Biochim Biophys Acta 891: 22–27
Schreiber U (1986) Detection of rapid induction kinetics with a new type of high frequency modulated chlorophyll fluorometer. Photosynth Res 9: 261–272
Schreiber U, Schliwa U and Bilger W (1986) Continuous recording of photochemical and non-photochemical chlorophyll fluorescence quenching with a new type of modulation fluorometer. Photosynth Res 10: 51–62
Spalding MH, Critchley C, Govindjee and Ogren WL (1984) Influence of carbon dioxide concentration during growth on fluorescence induction characteristics of the green algae Chlamydomonas reinhardtii. Photosynth Res 5: 169–176
Sundblad L-G (1988) Dark reduction of QA in intact barley leaves, as an effect of lowered CO2 concentration, monitored by chorophyll a luminescence and chlorophyll a F0 dark fluorescence. Biochim Biophys Acta 936: 429–434
Sundblad L-G, Palmqvist K and Samuelsson G (1986a) An energy dependent, transient peak in the minute range decay of luminescence, present in CO2 accumulating aells of Scenedesmus obliquus. FEBS Lett 199: 75–79
Sundblad L-G, Palmqvist K and Samuelsson G (1986b) Luminescence decay kinetics in relation to the relaxation of the transthylakoid ΔpH from high and low CO2 adapted cells of Scenedesmus obliquus. FEBS Lett 209: 28–32
Weis E and Berry JA (1987) Quantum efficiency of Photosystem II in relation to energy-dependent quenching of chlorophyll fluorescence. Biochim Biophys Acta 894: 198–208
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Sundblad, LG., Samuelsson, G., Wigge, B. et al. Luminescence decay kinetics in relation to quenching and stimulation of dark fluorescence from high and low CO2 adapted cells of Scenedesmus obliquus and Chlamydomonas reinhardtii . Photosynth Res 23, 269–282 (1990). https://doi.org/10.1007/BF00034857
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00034857