Abstract
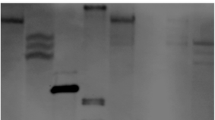
A 3.4-kilobase genomic DNA fragment from Pisum sativum L. containing the LegA gene, which encodes a major legumin storage protein, was transferred to Nicotiana plumbaginifolia using an Agrobacterium tumefaciens strain containing the Bin 19 binary vector system. Northern hybridisation analysis of legA-transformed plants demonstrated that legumin-specific RNA was present in developing seeds but not in developing leaves. Legumin protein was immunologically detected in the mature seeds of legA-transformed plants, and was present as the correct-size protein composed of disulphide-bonded polypeptides. It is concluded that the transferred pea genomic fragment contains all the information necessary for seed-specific expression of the legA gene, and for correct processing of the primary transcript and the precursor legumin protein.
Similar content being viewed by others
References
Amasino RM, Powell ALT, Gordon MP: Changes in T-DNA methylation and expression are associated with phenotypic variation and plant regeneration in a crown gall tumor line. Mol Gen Genet 197: 437–446 (1984).
Bäumlein H, Wobus U, Pustell J, Kafatos FC: The legumin gene family: structure of a B type gene of Vicia faba and a possible legumin gene specific regulatory element. Nucl Acids Res 14: 2707–2720 (1986).
Beachy RN, Chen Z-L, Horsch RB, Rogers SG, Hoffmann NJ, Fraley RT: Accumulation and assembly of soybean β-conglycinin in seeds of transformed petunia plants. EMBO J 4: 3047–3053 (1985).
Bevan M: Binary Agrobacterium vectors for plant transformation. Nucl Acids Res 12: 8711–8721 (1984).
Bevan MW, Flavell RB, Chilton M-D: A chimaeric antibiotic resistance gene as a selectable marker for plant cell transformation. Nature 304: 184–187 (1983).
Boulter D, Evans IM, Ellis JR, Shirsat A, Gatehouse JA, Croy RRD: Differential gene expression in the development of Pisum sativum. Plant Physiol Biochem 25(3): 283–289 (1987).
Casey R: Immunoaffinity chromatography as a means of purifying legumin from Pisum (pea) seeds. Biochem J 177: 509–520 (1979).
Casey R, Sharman JE, Wright DJ, Bacon JR, Guldager P: Quantitative variability in Pisum seed globulins: its assessment and significance. Qual Plant Plant Foods Hum Nutr 31: 333–346 (1982).
Chen Z-L, Schuler MA, Beachy RN: Functional analysis of regulatory elements in a plant embryo specific gene. Proc Natl Acad Sci (USA) 83: 8560–8564 (1986).
Croy RRD, Gatehouse JA: Genetic engineering of seed proteins: Current and potential applications. In: Dodds JH (ed.) Plant Genetic Engineering. Cambridge University Press, Cambridge/London/New York (1985) pp. 143–268.
Croy RRD, Gatehouse JA, Evans IM, Boulter D: Characterisation of the storage protein subunits synthesised in vitro by polyribosomes and RNA from developing pea (Pisum sativum L.) I. Legumin. Planta 148: 49–56 (1980).
Croy RRD, Lycett GW, Gatehouse JA, Yarwood JN, Boulter D: Cloning and analysis of cDNAs encoding plant storage protein precursors. Nature 295: 76–79 (1982).
Deblaere R, Bytebier B, DeGreve H, Deboeck F, Schell J, VanMontagu M, Leemans J: Efficient octopine Ti plasmidderived vectors for Agrobacterium-mediated gene transfer to plants. Nucl Acid Res 13: 4777–4788 (1985).
DeBlock M, Herrera-Estrella L, VanMontagu M, Schell J, Zambryski P: Expression of foreign genes in regenerated plants and in their progeny. EMBO J 3: 1681–1689 (1984).
Derbyshire E, Wright DJ, Boulter D: Legumin and vicilin, storage proteins of legume seeds. Phytochemistry 15: 3–24 (1976).
Ditta G, Stanfield S, Corbin D, Helinski DR: Broad host range DNA cloning system for Gram-negative bacteria: Construction of a gene bank of Rhizobium meliloti. Proc Natl Acad Sci (USA) 77: 7347–7351 (1980).
Domoney C, Casey R: Measurement of gene number for seed storage proteins in Pisum. Nucl Acids Res 13: 687–699 (1985).
Evans IM, Gatehouse JA, Croy RRD, Boulter D: Regulation of the transcription of storage-protein mRNA in nuclei isolated from developing pea (Pisum sativum L.) cotyledons. Planta 160: 559–568 (1984).
Gatehouse JA, Croy RRD, Boulter D: Isoelectric-focusing properties and carbohydrate content of pea (Pisum sativum) legumin. Biochem J 185: 497–503 (1980).
Gatehouse JA, Croy RRD, Boulter D: The synthesis and structure of pea storage proteins. In: CRC Critical Reviews in Plant Sciences, Vol. 1. CRC Press, Boca Raton, Florida (1985) pp. 287–314.
Gatehouse JA, Evans IM, Bown D, Croy RRD, Boulter D: Control of storage-protein synthesis during seed development in pea (Pisum sativum L.). Biochem J 208: 119–127 (1982).
Gatehouse JA, Evans IM, Croy RRD, Boulter D: Differential expression of genes during legume seed development. Phil Trans R Soc London Ser B 314: 367–384 (1986).
Goldberg RB: Regulation of plant gene expression. Phil Trans R Soc London Ser B 314: 343–353 (1986).
Goldsbrough PB, Gelvin SB, Larkins A: Expression of maize zein genes in transformed sunflower cells. Mol Gen Genet 202: 374–381 (1986).
Graham DE: The isolation of high molecular weight DNA from whole organisms or large tissue masses. Anal Biochem 85: 609–613 (1978).
Hepburn AG, Clarke LE, Pearson L, White J: The role of cytosine methylation in the control of nopaline synthase gene expression in a plant tumor. J Mol Appl Genet 2: 315–329 (1983).
Hoekema A, Hirsch PR, Hooykaas PJJ, Schilperoort RA: A binary plant vector strategy based on separation of vir and T-region of the Agrobacterium tumefaciens Ti plasmid. Nature 303: 179–180 (1983).
Horsch RB, Fraley RT, Rogers SG, Sanders PR, Lloyd A, Hoffmann N: Inheritance of functional foreign genes in plants. Science 223: 496–498 (1984).
Jones JDG, Dunsmuir P, Bedbrook J: High level expression of introduced chimaeric genes in regenerated transformed plants. EMBO J 4: 2411–2418 (1985).
Krishna TB, Croy RRD, Boulter D: Heterogeneity in subunit composition of the legumin of Pisum sativum. Phytochemistry 18: 1879–1880 (1979).
Lycett GW, Croy RRD, Shirsat AH, Boulter D: The complete nucleotide sequence of a legumin gene from pea (Pisum sativum L.). Nucl Acids Res 12: 4493–4506 (1984).
Maniatis T, Fritsch EF, Sambrook J: Molecular Cloning: A Laboratory Manual. Cold Spring Harbor Laboratory, Cold Spring Harbor, NY (1982).
Matta NK, Gatehouse JA, Boulter D: Molecular and subunit heterogeneity of legumin of Pisum sativum L. (Garden Pea) — A multi-dimensional gel electrophoretic study. J Exp Bot 32: 1295–1307 (1981).
McCormick S, Niedermeyer J, Fry J, Barnason A, Horsch R, Fraley R: Leaf disc transformation of cultivated tomato (L. esculentum) using Agrobacterium tumefaciens. Plant Cell Rep 5: 81–84 (1986).
Osborne TB: The Vegetable Proteins, 2nd edn. Longmans-Green, London (1924).
Otten LABM, Schilperoort RA: A rapid micro scale method for the detection of lysopine and nopaline dehydrogenase activities. Biochim Biophys Acta 527: 497–500 (1978).
Peerbolte R, Leenhouts K, Hooykaas-vanSlogteren GMS, Hoge JHC, Wullems GJ, Schilperoort RA: Clones from a shooty tobacco crown gall tumor I: deletions, rearrangements and amplifications resulting in irregular T-DNA structures and organizations. Plant Mol Biol 7: 265–284 (1986).
Peerbolte R, Leenhouts K, Hooykaas-vanSlogteren GMS, Wullems GJ, Schilperoort RA: Clones from a shooty tobacco crown gall tumor II: irregular T-DNA structures and organization, T-DNA methylation and conditional expression of opine genes. Plant Mol Biol 7: 285–299 (1986).
Prescott A, Martin C: A rapid method for the quantitative assessment of levels of specific mRNAs in plants. Plant Mol Biol Reporter 4: 219–224 (1987).
Sengupta-Gopalan C, Reichert NA, Barker RF, Hall TC, Kemp JD: Developmentally regulated expression of the bean β-phaseolin gene in tobacco seed. Proc Natl Acad Sci (USA) 82: 3320–3324 (1985).
Shaw CH, Watson MD, Carter GH, Shaw CH: The righthand copy of the nopaline Ti-plasmid 25 bp repeat is required for tumour formation. Nucl Acids Res 12: 6031–6041 (1984).
Spielmann A, Simpson RB: T-DNA structure in transgenic tobacco plants with multiple independent integration sites. Mol Gen Genet 205: 34–41 (1986).
Thomas PS: Hybridisation of denatured RNA transferred or dotted to nitrocellulose paper. Methods Enzymol 100: 255–266 (1983).
Towbin H, Staehelin T, Gordon J: Electrophoretic transfer of proteins from polyacrylamide gels to nitrocellulose sheets: Procedure and some applications. Proc Natl Acad Sci (USA) 76: 4350–4354 (1979).
Velten J, Schell J: Selection-expression plasmid vectors for use in genetic transformation of higher plants. Nucl Acids Res 13: 6981–6998 (1985).
Zambryski P, Joos H, Genetello C, Leemans J, VanMontagu M, Schell J: Ti plasmid vector for the introduction of DNA into plant cells without alteration of their normal regeneration capacity. EMBO J 2: 2143–2150 (1983).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Ellis, J.R., Shirsat, A.H., Hepher, A. et al. Tissue-specific expression of a pea legumin gene in seeds of Nicotiana plumbaginifolia . Plant Mol Biol 10, 203–214 (1988). https://doi.org/10.1007/BF00027397
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00027397