Abstract
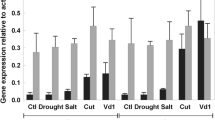
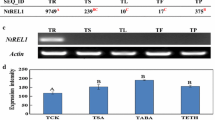
In a recent publication (Plant Molecular Biology 16: 547–565 (1991)) Showalter et al. described the isolation and initial characterization of fifteen extensin and extensin-like tomato cDNAs. These cDNAs were determined to fall into five distinct classes; class I and II clones encoded extensins, class III and V clones encoded glycine-rich proteins (GRPs), and class IV clones encoded a portion of a GRP sequence on one DNA strand and a portion of an extensin sequence on the other DNA strand. In this publication, a more detailed analysis of the expression of these cDNA classes was performed with respect to wounding in various tomato organs, development, kinetics and systemic extent of the wound response, ethylene treatment, abscisic acid (ABA) treatment, and drought stress by using RNA gel blot hybridizations. In general, extensin gene expression was readily detected in stems and roots, but not in leaves. With both class I and II extensin cDNA probes, wound-induced accumulation of mRNA in stems was first detected between 4 and 8 h after wounding with maximal accumulation occurring after 12 h. Moreover, these extensin wound responses were detected locally at the wound site but not systemically. Expression of the class III GRP was largely limited to wounded stem tissue. Initial detection and maximal accumulation of the class III GRP mRNA was similar to the extensins mRNAs; however, this GRP wound response occurred both locally and systemically. Additionally, abscisic acid treatment and drought stress resulted in the marked accumulation of the class III GRP mRNA in tomato stems, but did not alter the expression of the other cDNA classes. In contrast, expression of the class V GRP occurred in stems and roots and to a lesser extent in leaves and decreased in response to wounding over a 24 h time period. The class V GRP wound response was further characterized by an early, transient accumulation of mRNA occurring 2–4 h after wounding in stems and by its local nature.
Similar content being viewed by others
References
Bray EA: Drought and ABA-induced changes in polypeptide and mRNA accumulation in tomato leaves. Plant Physiol 88: 1210–1214 (1988).
Cassab GI, Varner JE: Cell wall proteins. Annu Rev Plant Physiol Plant Mol Biol 39: 321–353 (1988).
Condit CM, Keller B: The glycine-rich cell wall proteins of higher plants. In: Adair WS, Mecham RP (eds) Organization and Assembly of Plant and Animal Extracellular Matrix, pp. 119–135. Academic Press, New York (1990).
Condit CM, McClean BG, Meagher RB: Characterization of the expression of the petunia glycine-rich protein-1 gene product. Plant Physiol 93: 596–602 (1990).
Condit CM, Meagher RB: A gene encoding a novel glycine-rich structural protein of petunia. Nature 323: 178–181 (1986).
Condit CM, Meagher RB: Expression of a gene encoding a glycine-rich protein petunia. Mol Cell Biol 7: 4273–4279 (1987).
Corbin DR, Sauer N, Lamb CJ: Differential regulation of a hydroxyproline-rich glycoprotein gene family in wounded and infected plants. Mol Cell Biol 7: 4337–4344 (1987).
de Oliveira DE, Seurinck J, Inzé D, Van Montagu M, Botterman J: Differential expression of five Arabidopsis genes encoding glycine-rich proteins. Plant Cell 2: 427–436 (1990).
Epstein L, Lamport DTA: An intramolecular linkage involving isodityrosine in extensin. Phytochemistry 23: 1241–1246 (1984).
Gómez J, Sánchez-Martínez D, Stiefel V, Rigau J, Puigdomènech P, Pagès M: A gene induced by the plant hormone abscisic acid in response to water stress encodes a glycine-rich protein. Nature 334: 262–264 (1988).
Haffner MH, Chin MB, Lane BG: Wheat embryo ribonucleates: XII. formal characterization of terminal and penultimate nucleoside residues at 5′-ends of ‘capped’ RNA from imbibing wheat embryos. Can J Biochem 56: 729–733 (1978).
Kahl G: Molecular biology of wound healing: the conditioning phenomenon. In: Kahl G, Schell JS (eds) Molecular Biology of Plant Tumors, pp. 211–267. Academic Press, New York (1982).
Keller B, Sauer N, Lamb CJ: Glycine-rich cell wall proteins in bean: gene structure and association of the protein with the vascular system. EMBO J 7: 3625–3633 (1988).
Keller B, Schmid J, Lamb CJ: Vascular expression of a bean cell wall glycine-rich protein-β-glucuronidase gene fusion in transgenic tobacco. EMBO J 8: 1309–1314 (1989).
Keller B, Templeton MD, Lamb CJ: Specific localization of a plant cell wall glycine-rich protein in protoxylem cells of the vascular system. Proc Natl Acad Sci USA 86: 1529–1533 (1989).
Lamport DTA: Structure, biosynthesis and significance of cell wall glycoproteins. Recent Adv Phytochem 11: 79–115 (1977).
Lawton MA, Lamb CJ: Transcriptional activation of plant defense genes by fungal elicitor, wounding, and infection. Mol Cell Biol 7: 335–341 (1987).
Maniatis T, Fritsch EF, Sambrook J: Molecular Cloning: A Laboratory Manual. Cold Spring Harbor Laboratory, Cold Spring Harbor, NY (1982).
Mazau D, Esquerré-Tugayé MT: Hydroxyproline-rich glycoprotein accumulation in the cell walls of plants infected by various pathogens. Physiol Mol Plant Path 29: 147–157 (1986).
Mundy J, Chua N-H: Abscisic acid and water-stress induce the expression of a novel rice gene. EMBO J 7: 2279–2286 (1988).
Peña-Cortés H, Sánchez-Serrano JJ, Mertens R, Willmitzer L, Prat S: Abscisic acid is involved in the wound-induced expression of the proteinase inhibitor II gene in potato and tomato. Proc Natl Acad Sci USA 86: 9851–9855 (1989).
Rumeau D, Mazau D, Panabieres F, Delseny M, Esquerré-Tugayé MT: Accumulation of hydroxyproline-rich glycoprotein mRNAs in response to fungal elicitor and infection. Physiol Mol Plant Path 33: 419–429 (1988).
Sauer N, Corbin DR, Keller B, Lamb CJ: Cloning and characterization of a wound-specific hydroxyproline-rich glycoprotein in Phaseolus vulgaris. Plant Cell Environ 13: 257–266 (1990).
Showalter AM, Bell JN, Cramer CL, Bailey JA, Varner JE, Lamb CJ: Accumulation of hydroxyproline-rich glycoprotein mRNAs in response to fungal elicitor and infection. Proc Natl Acad Sci USA 82: 6651–6555 (1985).
Showalter AM, Rumeau D: Molecular biology of the plant cell wall hydroxyproline-rich glycoproteins. In: Adair WS, Mecham RP (eds) Organization and Assembly of Plant and Animal Extracellular Matrix, pp. 247–281. Academic Press, New York (1990).
Showalter AM, Varner JE: Plant hydroxyproline-rich glycoproteins. In: Marcus A (ed) The Biochemistry of Plants, Vol 15, pp. 485–520. Academic Press, New York (1989).
Showalter AM, Zhou J, Rumeau D, Worst SG, Varner JE: Tomato extensin and extensin-like cDNAs: structure and expression in response to wounding. Plant Mol Biol 16: 547–565 (1991).
Templeton MD, Dixon RA, Lamb CJ, Lawton MA: Hydroxyproline-rich glycoprotein transcripts exhibit different spatial patterns of accumulation in compatible and incompatible interaction between Phaseolus vulgaris and Colletotrichum lindemuthianum. Plant Physiol 94: 1265–1269 (1990).
Varner JE, Cooper JB: Hydroxyproline-rich glycoproteins extracted from the cell walls of aerated correct root slices. In: Ciferri O, Dure L (eds) Structure and Function of Plant Genomes, pp. 463–480. Plenum Press, New York (1983).
Ye ZH, Varner JE: Tissue-specific expression of cell wall proteins in developing soybean tissues. Plant Cell 3: 23–37 (1991).
Zhou J, Rumeau D, Showalter AM: Isolation and characterization of two wound-regulated tomato extensin genes. Plant Mol Biol (manuscript submitted).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Showalter, A.M., Butt, A.D. & Kim, S. Molecular details of tomato extensin and glycine-rich protein gene expression. Plant Mol Biol 19, 205–215 (1992). https://doi.org/10.1007/BF00027342
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00027342