Abstract
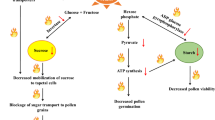
The molecular mechanisms that initiate and control the metabolic activities of seed germination are largely unknown. Sugars may play important roles in regulating such metabolic activities in addition to providing an essential carbon source for the growth of young seedlings and maintaining turgor pressure for the expansion of tissues during germination. To test this hypothesis, we investigated the physiological role of sugars in the regulation of α-amylase gene expression and carbohydrate metabolism in embryo and endosperm of germinating rice seeds. RNA gel blot analysis revealed that in the embryo and aleurone cells, expression of four α-amylase genes was differentially regulated by sugars via mechanisms beyond the well-known hormonal control mechanism. In the aleurone cells, expression of these α-amylase genes was regulated by gibberellins produced in the embryo and by osmotically active sugars. In the embryo, expression of two α-amylase genes and production of gibberellins were transient, and were probably induced by depletion of sugars in the embryo upon imbibition, and suppressed by sugars influx from the endosperm as germination proceeded. The differential expression of the four α-amylase genes in the embryo and aleurone cells was probably due to their markedly different sensitivities to changes in tissue sugar levels. Our study supports a model in which sugars regulate the expression of α-amylase genes in a tissue-specific manner: via a feedback control mechanism in the embryo and via an osmotic control mechanism in the aleurone cells. An interactive loop among sugars, gibberellins, and α-amylase genes in the germinating cereal grain is proposed.
Similar content being viewed by others
References
Briggs DE: Barley germination: Biochemical changes and hormonal control. In: Shewry PR (ed), Barley: Genetics, Biochemistry, Molecular Biology and Biotechnology, pp. 369–401. CAB International, Oxon, UK (1992).
Chan M-T, Chang H-H, Ho S-L, Tong W-F, Yu S-M: Agrobacterium-mediated production of transgenic rice plants expressing a chimeric α-amylase promoter/β-glucuronidase gene. Plant Mol Biol 22: 491–506 (1993).
Chan M-T, Chao Y-C, Yu S-M: Novel gene expression system for plant cells based on induction of α-amylase promoter by carbohydrate starvation. J Biol Chem 269: 17635–17641 (1994).
Chao Y-C, Wood HA, Chang CY, Lee H-J, Shen W-C, Lee H-T: Differential expression of Hz-1 baculovirus genes during productive and persistent viral infections. Virol 66: 1442–1448 (1992).
Chen M-H, Liu L-F, Chen Y-R, Wu H-K, Yu S-M: Expression of α-amylases, carbohydrate metabolism, and autophagy in cultured rice cells are coordinately regulated by sugar nutrient. Plant J 6: 625–636 (1994).
Criqui MC, Durr A, Parmentier Y, Marrach J, Fleck J, Jamet E: How are photosynthetic genes repressed in freshly-isolated mesophyll protoplasts of Nicotiana sylvestris? Plant Physiol Biochem 30: 597–601 (1992).
Feinberg AP, Vogelstein B: A technique for radiolabelling DNA restriction endonuclease fragments to high specific activity. Anal Biochem 132: 6–13 (1983).
Graham IA, Denby KJ, Leaver CJ: Carbon catabolite repression regulates glyoxylate cycle gene expression in cucumber. Plant Cell 6: 761–772 (1994).
Groat JI, Briggs DE: Gibberellins and α-amylase formation in germinating barley. Phytochemistry 8: 1615–1627 (1969).
Hattori T, Nakagawa S, Nakamura K: High-level expression of tuberous root storage protein genes of sweet potato in stems of plantlets grown in vitro on sucrose medium. Plant Mol Biol 14: 595–604 (1990).
Huang N, Chandler J, Thomas BR, Koizumi N, Rodriguez RL. Metabolic regulation of α-amylase gene expression in transgenic cell cultures of rice (Oryza sativa L.). Plant Mol Biol 23: 737–747 (1993).
Ingle J, Beevers L, Hageman RH: Metabolic changes associated with the germination of corn. I. Changes in weight and metabolites and their redistribution in the embryo axis, scutellum, and endosperm. Plant Physiol 39: 735–740 (1964).
Jacobsen JV, Chandler PM: Gibberellin and abscisic acid in germinating cereals. In: Davies PJ (ed) Plant Hormones and Their Role in Plant Growth and Development, pp. 164–193. Martinus Nijhoff, Dordrecht (1987).
Jang J-C, Sheen J: Sugar sensing in higher plants. Plant Cell 6: 1665–1679 (1994).
Jones RL, Armstrong JE: Evidence for osmotic regulation of hydrolytic enzyme production in germinating barley seeds. Plant Physiol 48: 137–142 (1971).
Karrer EE, Litts JC, Rodriguez RL: Differential expression of α-amylase genes in germinating rice and barley seeds. Plant Mol Biol 16: 797–805 (1991).
Karrer EE, Rodriguez RL: Metabolic regulation of rice α-amylase and sucrose synthase genes in planta. Plant J 2: 517–523 (1992).
Koch KE, Nolte KD, Duke ER, McCarty DR, Avigne WT: Sugar levels modulate differential expression of maize sucrose synthase genes. Plant Cell 4: 59–69 (1992).
Mayer AM, Shain Y: Control of seed germination. Ann Rev Plant Physiol 25: 167–193 (1974).
McElroy D, Rothenberg M, Wu R: Structural characterization of a rice actin gene. Plant Mol Biol 14: 163–171 (1990).
Müller-Röber BT, Kossmann J, Hannah LC, Willmitzer L, Sonnewald U: One of two different ADP-glucose pyrophosphorylase genes from potato responds strongly to elevated levels of sucrose. Mol Gen Genet 224: 136–146 (1990).
Murata T, Akazawa T, Fukuchi S: Enzymic mechanism of starch breakdown in germinating rice seeds. I. An analytical study. Plant Physiol 43: 1899–1905 (1968).
Nakamura K, Ohto M-A, Yoshida N, Nakamura K: Sucrose-induced accumulation of β-amylase occurs concomitant with the accumulation of starch and sporamin in leaf-petiole cuttings of sweet potato. Plant Physiol 96: 902–909 (1991).
Nishijima T, Koshioka M, Yamaji H: Nondwarf rice seedling bioassay for gibberellins. Plant Physiol 98: 962–965 (1992).
Okamoto K, Kitano H, Akazawa T: Biosynthesis and excretion of hydrolases in germinating cereal seeds. Plant Cell Physiol 21: 201–204 (1980).
Radley M: The effect of the endosperm on the formation of gibberellin by barley embryos. Planta 86: 218–223 (1969).
Ranjhan S, Karrer EE, Rodriguez RL: Localizing α-amylase gene expression in germinated rice grains. Plant Cell Physiol 33: 73–79 (1992).
Rocha-Sosa M, Sonnewald U, Frommer W, Stratmann M, Schell J, Willmitzer L: Both developmental and metabolic signals activate the promoter of a class I patatin gene. EMBO J 8: 23–29 (1989).
Salanoubat M, Belliard G: The steady-state level of potato sucrose synthase mRNA is dependent on wounding, anaerobiosis, and sucrose concentration. Gene 84: 181–185 (1989).
Sheen J: Metabolic repression of transcription in higher plants. Plant Cell 2: 1027–1038 (1990).
Sheen J: Feedback control of gene expression. Photosynthe Res 39: 427–438 (1994).
Sheu J-J, Jan S-P, Lee H-T, Yu S-M: Control of transcription and mRNA turnover as mechanisms of metabolic repression of α-amylase gene expression. Plant J 5: 655–664 (1994).
Sun Z, Henson CA: A quantitative assessment of the importance of barley seed α-amylase, β-amylase, debranching enzyme, and α-glucosidase in starch degradation. Arch Biochem Biophys 284: 298–305 (1991).
Thomas BR, Rodriguez RL: Metabolite signals regulate gene expression and source/sink relations in cereal seedlings. Plant Physiol 106: 1235–1239 (1994).
Verwoerd TC, Dekker BMM, Hoekema A: A small-scale procedure for the rapid isolation of plant RNAs. Nucl Acids Res 17: 2362 (1989).
Yu S-M, Kuo Y-H, Sheu G, Sheu Y-J, Liu L-F: Metabolic derepression of α-amylase gene expression in suspension-cultured cells of rice. J Biol Chem 266: 21131–21137 (1991).
Yu S-M, Tzou W-S, Lo W-S, Kuo Y-H, Lee H-T, Wu R: Regulation of α-amylase-encoding gene expression in germinating seeds and cultured cells of rice. Gene 122: 247–253 (1992).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Yu, SM., Lee, YC., Fang, SC. et al. Sugars act as signal molecules and osmotica to regulate the expression of α-amylase genes and metabolic activities in germinating cereal grains. Plant Mol Biol 30, 1277–1289 (1996). https://doi.org/10.1007/BF00019558
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00019558