Abstract
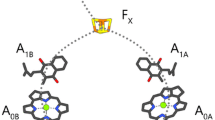
The kinetic and spectroscopic properties of the secondary electron acceptor A1 were determined by flash absorption spectroscopy at room and cryogenic temperatures in a Photosystem I (PS I) core devoid of the iron-sulfur clusters FX, FB and FA. It was shown earlier (Warren, P.V., Golbeck, J.H. and Warden, J.T. (1993) Biochemistry 32: 849–857) that the majority of the flash-induced absorbance increase at 820 nm, reflecting formation of P700+, decays with a t1/2 of 10 μs due to charge recombination between P700+ and A1 −. Following A1 − directly around 380 nm, where absorbance changes due to the formation of P700+ are negligible, two major decay components were resolved in this study with t1/2 of ≈ 10 μs and 110 μs at an amplitude ratio of ≈ 2.5:1. The difference spectra between 340 and 490 nm of the two kinetic phases are highly similar, showing absorbance increases from 340 to 400 nm characteristic of the one-electron reduction of the phylloquinone A1. When measured at 10 K, the flash-induced absorbance changes around 380 nm can be fitted with two decay phases of t1/2 ≈ 15 μs and 150 μs at an amplitude ratio ≈ 1:1. The difference spectra of both kinetic phases from 340 to 400 nm are similar to those determined at 298 K and are therefore attributed to charge recombination in the pair P700+A1 −. These results indicate that the backreaction between P700+ and A1 − is multiphasic when FX, FB and FA are removed, and only slightly temperature dependent in the range of 298 K to 10 K.
Similar content being viewed by others
Abbreviations
- Chl:
-
chlorophyll
- D:
-
pathlength for the measuring light through the sample
- DPIP:
-
2,6-dichlorophenolindophenol
- EPR:
-
electron paramagnetic resonance
- IR:
-
infrared
- PS I:
-
Photosystem I
- Tris:
-
Tris(hydroxymethyl)aminomethane
- UV:
-
ultraviolet
References
Bock CH, Van der Est AJ, Brettel K and Stehlik D (1989) Nanosecond electron transfer kinetics in Photosystem I as obtained from transient EPR at room temperature. FEBS Lett 247: 91–96
Brettel K (1988) Electron transfer from A1 − to an iron-sulfur center with t1/2=200 ns at room temperature in Photosystem I. Characterization by flash absorption spectroscopy. FEBS Lett 239: 93–98
Brettel K (1989) New assignment for the 250 μs kinetics of Photosystem I: P-700+ recombines with A1 − (not FX −). Biochim. Biophys Acta 976: 246–249
Brettel K and Sétif P (1987) Magnetic-field effects on primary reactions in Photosystem I. Biochim Biophys Acta 893: 109–114
Brettel K, Sétif P and Mathis P (1986) Flash-induced absorption changes in Photosystem I at low temperature: Evidence that the electron acceptor A1 is vitamin K1. FEBS Lett 203: 220–224
DenBlanken HF and Hoff AF (1983) High-resolution absorbance-difference spectra of the triplet state of the primary donor P-700 in Photosystem I subchloroplast particles measured with absorbance-detected magnetic resonance at 1.2 K. Evidence that P-700 is a dimeric chlorophyll complex. Biochim Biophys Acta 724: 52–61
Fenton FM, Pellin MF, Govindjee and Kaufmann KJ (1979) Primary photochemistry of the reaction center of Photosystem I. FEBS Lett 100: 1–4
Golbeck JH (1992) Structure and Function of Photosystem I. In: Burke I (ed) Annual Review of Plant Physiology and Plant Molecular Biology, Vol 43, pp 293–324. Annual Reviews Inc, Palo Alto, CA
Golbeck JH, Velthuys BR and Kok B (1978) Evidence that the intermediate electron acceptor, A2, in Photosystem I is a bound iron-sulfur protein. Biochim Biophys Acta 504: 226–230
Hastings G, Kleinherenbrink FAM, Lin S, McHugh TF and Blankenship RE (1994) Observation of the reduction and reoxidation of the primary electron acceptor in Photosystem I. Biochemistry 33: 3193–3200
Ke B (1972) The rise time of photoreduction, difference spectrum, and oxidation-reduction potential of P430. Arch Biochem Biophys 152: 70–77
Koike H and Katoh S (1982) Spectral features of the bound electron acceptor A2 of Photosystem I. Photochem Photobiol 35: 527–531
Krantz W and Myers J (1955) Nutrition and growth of several blue-green algae. Amer J. Bot 42: 282–287
Krauss N, Hinrichs W, Witt I, Fromme P, Pritzkow W, Dauter Z, Betzel C, Wilson KS, Witt HT and Saenger W (1993) 3-Dimensional structure of system-I of photosynthesis at 6 Ångstrom resolution. Nature 361: 326–330
Lüneberg F, Fromme P and Schlodder E (1994) Spectroscopic characterization of PS I core complexes from thermophilic Synechococcus sp.-identical reoxidation kinetics of A1 − before and after removal of the iron-sulfur-clusters FA and FB. FEBS Lett 338: 197–202
Mathis P and Sétif P (1988) Kinetic studies on the function of A1 in the Photosystem I reaction center. FEBS Lett 237: 65–68
Mathis P, Sauer K and Remy R (1978) Rapidly reversible flash-induced electron transfer in a P-700 chlorophyll-protein complex isolated with SDS. FEBS Lett 88: 275–278
Moenneloccoz P, Heathcote P, Maclachlan DJ, Berry MC, Davis IH and Evans MCW (1994) Path of electron transfer in Photosystem I: Direct evidence of forward electron transfer from A(1) to Fe-Sx. Biochemistry 33: 10037–10042
Parrett KG, Mehari T, Warren P and Golbeck JH (1989) Purification and properties of the intact P700 and FX-containing Photosystem I core protein. Biochim Biophys Acta 973: 324–332
Sauer K, Mathis P, Acker S and Van Best JA (1978) Electron acceptors associated with P-700 in Triton solubilized Photosystem I particles from spinach chloroplasts. Biochim Biophys Acta 503: 120–134
Sétif P and Bottin H (1989) Identification of electron-transfer reactions involving the acceptor A1 of Photosystem I at room temperature. Biochemistry 28: 2689–2697
Sétif P and Brettel K (1990) Photosystem I photochemistry under highly reducing conditions: Study of the P700 triplet state formation from the secondary radical pair (P700+-A1 +). Biochim Biophys Acta 1020: 232–238
Sétif P and Brettel K (1993) Forward electron transfer from phylloquinone-A1 to iron sulfur centers in spinach Photosystem-I. Biochemistry 32: 7846–7854
Sétif P and Mathis P (1986) Photosystem I photochemistry: A new kinetic phase at low temperature. Photosynth Res 9: 47–54
Sétif P, Hervo G and Mathis P (1981) Flash-induced absorption changes in Photosystem I. Radical pair or triplet state formation? Biochim Biophys Acta 638: 257–267
Sétif P, Bottin H and Mathis P (1985) Absorption studies of primary reactions in Photosystem I. Yield and rate of formation of the P700 triplet state. Biochim Biophys Acta 808: 112–122
Shuvalov VA, Nuijs AM, Van Gorkom HJ, Smit HWJ and Duysens LNM (1986) Picosecond absorbance changes upon selective excitation of the primary electron donor P-700 in Photosystem I. Biochim Biophys Acta 850: 319–323
Van Best JA and Mathis P (1978) Apparatus for the measurement of small absorption change kinetics at 820 nm in the nanosecond range after a ruby laser flash. Rev Sci Instrum 49: 1332–1335
Van der Est A, Bock CH, Golbeck JH, Brettel K, Sétif P and Stehlik D (1994) Electron transfer from the acceptor A1 to the iron-sulfur centers in Photosystem I as studied by transient EPR spectroscopy. Biochemistry 33: 11789–11797
Warren PV, Parrett KG, Warden JT and Golbeck JH (1990) Characterization of a Photosystem I core containing P700 and intermediate electron acceptor A1. Biochemistry 29: 6545–6550
Warren PV, Golbeck JH and Warden JT (1993) Charge recombination between P700+ and A1 − occurs directly to the ground state of P700 in a Photosystem I core devoid of FX, FB, and FA. Biochemistry 32: 849–857
Wasielewski MR, Fenton FM and Govindjee (1987) The rate of formation of P700+-A0 − in Photosystem I particles from spinach as measured by picosecond transient absorption spectroscopy. Photosynth Res 12: 181–190
Author information
Authors and Affiliations
Additional information
Published as Journal Series #10890 of the University of Nebraska Agricultural Research Division and supported by a grant from the National Science Foundation (MCB-9205756).
Rights and permissions
About this article
Cite this article
Brettel, K., Golbeck, J.H. Spectral and kinetic characterization of electron acceptor A1 in a Photosystem I core devoid of iron-sulfur centers FX, FB and FA . Photosynth Res 45, 183–193 (1995). https://doi.org/10.1007/BF00015559
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00015559