Abstract
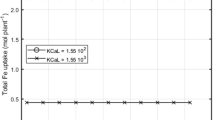
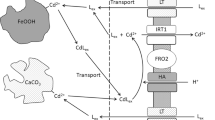
Both experimental extractions and theoretical calculations were undertaken to assess whether organic acid-mediated Fe dissolution could play a significant role in elevating the concentration of Fe-complexes in the rhizosphere, and further, whether this could satisfy the Fe demands of a plant utilizing ferric reduction to acquire Fe. Using a mathematical computer model, it was predicted that organic acids released from and diffusing away from the root would result in a solution organic acid concentration at the root surface of between 1 to 50 μM. Over 99% of the organic acids lost by the root were predicted to remain within 1 mm of the root surface. The experimental results indicated that citrate-mediated Fe dissolution of amorphous Fe(OH)3, was rapid in comparison with citrate dissolution of the Fe-oxides, Fe2O3 and Fe3O4. The rate of citrate and malate mediated Fe-dissolution was dependent on many factors such as pH, metal cations and phosphate saturation of the Fe(OH)3 surface. At pH values ≤6.8, citrate formed stable complexes with Fe and dissolution proceeded rapidly. Under optimal growth conditions for a plant utilizing a reductive-bound mechanism of Fe acquisition (dicots and non-grass monocots), it can be expected that citrate and malate may be able to satisfy a significant proportion of the plant's Fe demand through the formation of plant-available organic-Fe3+ complexes in the rhizosphere. In high pH soils (pH≥7.0), the plant must rely on other sources of Fe, as citrate-mediated Fe dissolution is slow and Fe-citrate complexes are unstable. Alternatively, the root acidification of the rhizosphere could allow the formation of stable Fe-organic complexes. ei]H Marschner
Similar content being viewed by others
References
Barber S A 1984 Soil Nutrient Bioavailability: A Mechanistic Approach. John Wiley and Sons, New York, USA. 398 p.
Bienfait H F, Bino R J, van derBliek A M, Duivenvoorden J F and Fontaine J M 1983 Characterization of ferric reducing activity in roots of Fe-deficient Phaseolus vulgaris. Physiol. Plant. 59, 196–202.
Brown I C, Jolley V D and Lytle C M 1991 Comparative evaluation of iron solubilizing substances (phytosiderophores) released by oat and corn: Iron-efficient and iron-inefficient plants. Plant and Soil 130, 157–163.
Brüggemann W, Moog P R, Nakagawa H, Janiesch P Kuiper P J C 1990 Plasma membrane-bound NADH:Fe3+-EDTA reductase and iron deficiency in tomato (Lycopersicon esculentum). Is there a turbo reductase?. Physiol. Plant. 79, 339–346.
Carson K C, Holliday S, Glenn A R and Dilworth M J 1992 Siderophore and organic acid production in root nodules. Arch. Microbiol. 157, 264–271.
Cline G R, Powell P E, Szaniszlo P J and Reid C P P 1982 Comparison of the abilities of hydroxamic, synthetic, and other natural organic acids to chelate iron and other ions in nutrient solution. Soil Sci. Soc. Am. J. 46, 1158–1164.
Darrah P R 1991a Measuring the diffusion coefficients of rhizosphere exudates in soil. II. The diffusion of sorbing compounds. J. Soil Sci. 42, 421–436.
Darrah P R 1991b Models of the rhizosphere I. Microbial population dynamics around a root releasing soluble and insoluble carbon. Plant and Soil 133, 187–199.
Delhaize E, Ryan P R and Randall P J 1993 Aluminum tolerance in wheat (Triticum aestivum L.). II. Aluminum-stimulated excretion of malic acid from root apices. Plant Physiol. 103, 695–702.
Dinkelaker B, Römheld V and Marschner H 1989 Citric acid excretion and precipitation of calcium citrate in the rhizosphere of white lupin (Lupinus albus L.). Plant Cell. Environ. 12, 285–292.
Elkhatib E A 1990 Simultaneous determination of low molecular weight organic acids in soil solution by ion chromatography. Z. Pflanzenernahr. Bodenkd. 153, 201–205.
Gerke J 1993 Solubilization of Fe(III) from humic iron complexes humic iron oxide mixtures and from poorly ordered iron-oxide by organic acids. Z. Pflanzenernahr. Bodenkd. 156, 253–257.
Gerke J, Roemer W and Jungk A 1994 Excretion of citric and malic acid by proteoid roots of Lupinus albus L.; Effect on soil solution concentrations of phosphate, iron, and aluminum in the proteoid rhizosphere in samples of oxisol and a luvisol. Z. Pflanzenernahr. Bodenkd. 157, 289–294.
Grusak M A, Welch R M and Kochian L V 1990 Physiological significance of a single-gene; mutant of Pisum sativum L. exhibiting excess iron accumulation. Plant Physiol. 93, 976–981.
Grusak M A, Kochian L V and Welch R M 1993 Spatial and temporal development of iron(III) reductase activity in root systems of Pisum sativum (Fabaceae) challenged with iron-deficiency stress. Am. J. Bot. 80, 300–3118.
Guerinot M L and Li Y 1994 Iron: Nutritious, noxious, and not readily available. Plant Physiol. 104, 815–820.
Hoffland E, Findenegg G R and Nelemans J A 1989 Solubilization of rock phosphate by rape. II. local root exudation of organic acids as a response to P-starvation. Plant and Soil 113, 161–165.
Jones D L and Darrah P R 1994 Role of root derived organic acids in the mobilization of nutrients from the rhizosphere. Plant and Soil 166, 247–257.
Jones D L and Darrah P R 1995 Influx and efflux of organic acids across the soil-root interface of Zea mays L. and its implications in rhizosphere C flow. Plant and Soil 173, 103–109.
Jones D L and Edwards A C 1993 Evaluation of polysulfone hollow fibres and ceramic suction samplers as devices for the in situ extraction of soil solution. Plant and Soil 150, 157–165.
Jones D L and Edwards A C 1993 Effect of moisture content and preparation technique on the composition of soil solution obtained by centrifugation. Commun. Soil Sci. Plant Anal. 24, 171–186.
Karathanasis A D 1991 Seasonal variation in solution composition and mineral stability of two Kentucky Alfisols. Soil Sci. Soc. Am. J. 55, 881–890.
Kochian L V 1991 Mechanisms of micronutrient uptake and translocation in plants. In Micronutrients in Agriculture, 2nd Edition. pp 229–296. Soil Science Society of America, Madison, WI, USA.
Landsburg E C 1986 Function of rhizodermal transfer cells in the Fe-stress response of Capsicum annuum L., Plant Physiol. 82, 511–517.
Lindsay W L 1979 Chemical Equilibria in Soils. Wiley-Interscience, New York, USA.
Marschner H 1986 Mineral Nutrition in Higher Plants. Academic Press, London, UK.
Moody M D 1986 Microorganisms and iron limitation. BioScience 36, 618–623.
Mori S, Nishizawa N, Hayashi H, Chino M, Yoshimura E and Ishihara J 1991 Why are young rice plants highly susceptible to iron deficiency? Plant and Soil 130, 143–156.
Murphy J and Riley J P 1962 A modified single solution method for the determination of phosphate in natural waters. Anal. Chim. Acta. 27, 31–36.
Nye P H and Tinker P B 1977 Solute movement in the soil-root system. Blackwells Scientific, Oxford, UK. 328 p.
Parker D R, Chaney R L and Norvell W A 1995 Chemical equilibria models: Applications to plant nutrition research. In Chemical Equilibria and Reaction Models. Ed R H Loeppert, Soil Science Society of America Special Publication No. 42, Madison, WI, USA.
Pohlman A A and McColl J G 1988 Soluble organics from forest litter and their role in metal dissolution. Soil Sci. Soc. Am. J. 52, 265–271.
USDA 1975 Soil Taxonomy. Soil Conservation Service, U S Department of Agriculture, Washington DC, USA. Agriculture Handbook No. 436.
VonWiren N, Mori S, Marschner H and Römheld V 1994 Iron inefficiency in maize mutant ysl (Zea mays L. cv Yellow-Stripe) is caused by a defect in uptake of iron phytosiderophores. Plant Physiol. 106, 71–77.
Welch R M 1995 Micronutrient nutrition of plants. Crit. Rev. Plant Sci. 14, 49–82.
Welkie G W and Miller G W 1993 Plant iron uptake physiology by nonsiderophore systems. In Iron Chelation in Plants and Microorganisms. pp 345–369. Academic Press, London, UK.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Jones, D.L., Darah, P.R. & Kochian, L.V. Critical evaluation of organic acid mediated iron dissolution in the rhizosphere and its potential role in root iron uptake. Plant Soil 180, 57–66 (1996). https://doi.org/10.1007/BF00015411
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00015411