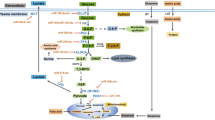
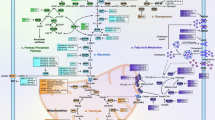
Abstract
Over 80 years ago, Otto Warburg discovered the phenomena of aerobic glycolysis, known now as the Warburg effect, whereby cancer cells relies on glycolysis, rather than the more efficient process of oxidative phosphorylation, for ATP production, even when oxygen is available. Recent progress in the field of cancer metabolism documents that alterations of many oncogenes, such as MYC, or tumor suppressor genes are involved in the metabolic switch of cancer cells to aerobic glycolysis. Myc was the first oncogenic transcription factor reported to regulate microRNAs (miRNAs), which are non-coding RNAs of ~22-nucleotides that act as post-transcriptional regulators of gene expression. Hundreds of miRNAs have been identified in the human genome thus far that are estimated to regulate about 30% of human genes involved in diverse processes including development, cell differentiation, cell proliferation, apoptosis, and metabolism. In this chapter, I will review the characteristics of cancer metabolism and its molecular basis, with an emphasis on the interplay of miRNAs and oncogenes/tumor suppressors in the field. It is hoped that understanding of molecular basis of cancer metabolism, including the involvement of miRNAs, will provide new and powerful strategies to target this deadly disease.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Baas AF, Kuipers J, van der Wel NN, et al. Complete polarization of single intestinal epithelial cells upon activation of LKB1 by STRAD. Cell. 2004;116:457–66.
Bensaad K, Tsuruta A, Selak MA, et al. TIGAR, a p53-inducible regulator of glycolysis and apoptosis. Cell. 2006;126:107–20.
Boudeau J, Baas AF, Deak M, et al. MO25alpha/beta interact with STRADalpha/beta enhancing their ability to bind, activate and localize LKB1 in the cytoplasm. EMBO J. 2003;22:5102–14.
Chan SY, Zhang YY, Hemann C, et al. MicroRNA-210 controls mitochondrial metabolism during hypoxia by repressing the iron-sulfur cluster assembly proteins ISCU1/2. Cell Metab. 2009;10:273–84.
Chang TC, Mendell JT. MicroRNAs in vertebrate physiology and human disease. Annu Rev Genomics Hum Genet. 2009;8:215–39.
Chang TC, Yu D, Lee YS, et al. Widespread microRNA repression by Myc contributes to tumorigenesis. Nat Genet. 2008;40:43–50.
Cho WC. MicroRNAs in cancer – from research to therapy. Biochim Biophys Acta. 2010;1805:209–17.
Cole MD. The myc oncogene: its role in transformation and differentiation. Annu Rev Genet. 1986;20:361–84.
Cory S, Harris AW, Langdon WY, et al. The myc oncogene and lymphoid neoplasia: from translocations to transgenic mice. Hamatol Bluttransfus. 1987;31:248–51.
Crosby ME, Kulshreshtha R, Ivan M, et al. MicroRNA Regulation of DNA repair gene expression in hypoxic stress. Cancer Res. 2009;69:1221–9.
Dang CV, Kim JW, Gao P, et al. The interplay between MYC and HIF in cancer. Nat Rev Cancer. 2008;8:51–6.
Easton JB, Houghton PJ. mTOR and cancer therapy. Oncogene. 2006;25:6436–46.
Fasanaro P, D’Alessandra Y, Di Stefano V, et al. MicroRNA-210 modulates endothelial cell response to hypoxia and inhibits the receptor tyrosine kinase ligand Ephrin-A3. J Biol Chem. 2008;283:15878–83.
Favaro E, Ramachandran A, McCormick R, et al. MicroRNA-210 regulates mitochondrial free radical response to hypoxia and Krebs cycle in cancer cells by targeting iron sulfur cluster protein ISCU. PLoS One. 2010;5:e10345.
Fukuda R, Zhang H, Jim JW, et al. HIF-1 regulates cytochrome oxidase subunit composition to optimize efficiency of respiration in hypoxic cells. Cell. 2007;129:111–22.
Gao P, Tchernyshyov I, Chang TC, et al. c-Myc suppression of miR-23a/b enhances mitochondrial glutaminase expression and glutamine metabolism. Nature. 2009;458:762–5.
Giannakakis A, Sandaltzopoulos R, Greshock J, et al. MiR-210 links hypoxia with cell cycle regulation and is deleted in human epithelial ovarian cancer. Cancer Biol Ther. 2008;7:255–64.
Godlewski J, Nowicki MO, Bronisz A, et al. MicroRNA-451 regulates LKB1/AMPK signaling and allows adaptation to metabolic stress in glioma cells. Mol Cell. 2010;37:620–32.
Hawley SA, Boudeau J, Reid JL, et al. Complexes between the LKB1 tumor suppressor, STRAD alpha/beta and MO25 alpha/beta are upstream kinases in the AMP-activated protein kinase cascade. J Biol. 2003;2:28.
Hermeking H. p53 enters the MicroRNA world. Cancer Cell. 2007;12:414–8.
Inoki K, Li Y, Zhu T, et al. TSC2 is phosphorylated and inhibited by Akt and suppresses mTOR signalling. Nat Cell Biol. 2002;4:648–57.
Jamerson MH, Johnson MD, Dickson RB. Of mice and Myc: c-Myc and mammary tumorigenesis. J Mammary Gland Biol Neoplasia. 2004;9:27–37.
Kaelin WG Jr.. ROS: really involved in oxygen sensing. Cell Metab. 2005;1:357–8.
Kim JW, Dang CV. Cancer’s molecular sweet tooth and the Warburg effect. Cancer Res. 2006;66:8927–30.
Kim JW, Mori S, Nevins JR. Myc-induced microRNAs integrate Myc-mediated cell proliferation and cell fate. Cancer Res. 2010;70:4820–8.
Kim JW, Tchernyshyov I, Semenza GL, et al. HIF-1-mediated expression of pyruvate dehydrogenase kinase: a metabolic switch required for cellular adaptation to hypoxia. Cell Metab. 2006;3:177–85.
Kim JW, Zeller KI, Wang Y, et al. Evaluation of Myc E-box phylogenetic footprints in glycolytic genes by chromatin immunoprecipitation assays. Mol Cell Biol. 2004;24:5923–36.
Kulshreshtha R, Ferracin M, Wojcik S, et al. A microRNA signature of hypoxia. Mol Cell Biol. 2007;27:1859–67.
Langdon WY, Harris AW, Cory S, et al. The c-myc oncogene perturbs B lymphocyte development in E-mu-myc transgenic mice. Cell. 1986;47:11–18.
Lau LF, Nathans D. Expression of a set of growth-related immediate early genes in BALB/c 3T3 cells: coordinate regulation with c-fos or c-myc. Proc Natl Acad Sci USA. 1987;84:1182–6.
Leder A, Pattengale PK, Kuo A, et al. Consequences of widespread deregulation of the c-myc gene in transgenic mice: multiple neoplasms and normal development. Cell. 1986;45:485–95.
Matoba S, Kang JG, Patino WD, et al. P53 regulates mitochondrial respiration. Science. 2006;312:1650–3.
O’Donnell KA, Wentzel EA, Zeller KI, et al. c-Myc-regulated microRNAs modulate E2F1 expression. Nature. 2005;435:839–43.
Olive V, Bennett MJ, Walker JC, et al. MiR-19 is a key oncogenic component of miR-17-92. Genes Dev. 2009;23:2839–49.
Oren M. Decision making by p53: life, death and cancer. Cell Death Differ. 2003;10:431–42.
Papandreou I, Cairns RA, Fontana L, et al. HIF-1 mediates adaptation to hypoxia by actively downregulating mitochondrial oxygen consumption. Cell Metab. 2006;3:187–97.
Park SY, Lee JH, Ha M, et al. MiR-29 miRNAs activate p53 by targeting p85 alpha and CDC42. Nat Struct Mol Biol. 2009;16:23–9.
Phang JM, Liu W, Zabirnyk O. Proline metabolism and microenvironmental stress. Annu Rev Nutr. 2010;30:441–63.
Rana TM. Illuminating the silence: understanding the structure and function of small RNAs. Nat Rev Mol Cell Biol. 2009;8:23–36.
Sachdeva M, Zhu S, Wu F, et al. P53 represses c-Myc through induction of the tumor suppressor miR-145. Proc Natl Acad Sci USA. 2009;106:3207–12.
Sanders MJ, Ali ZS, Hegarty BD, et al. Defining the mechanism of activation of AMP-activated protein kinase by the small molecule A-769662, a member of the thienopyridone family. J Biol Chem. 2007;282:32539–48.
Semenza GL. Targeting HIF-1 for cancer therapy. Nature Rev Cancer. 2003;3:721–32.
Semenza GL, Roth PH, Fang HM, et al. Transcriptional regulation of genes encoding glycolytic enzymes by hypoxia-inducible factor 1. J Biol Chem. 1994;269:23757–63.
Sheiness D, Bishop JM. DNA and RNA from uninfected vertebrate cells contain nucleotide sequences related to the putative transforming gene of avian myelocytomatosis virus. J Virol. 1979;31:514–21.
Shim H, Dolde C, Lewis BC, et al. C-Myc transactivation of LDH-A: implications for tumor metabolism and growth. Proc Natl Acad Sci USA. 1997;94:6658–63.
Shin S, Cha HJ, Lee EM, et al. MicroRNAs are significantly influenced by p53 and radiation in HCT116 human colon carcinoma. Int J Oncol. 2009;34:1645–52.
Suzuki HI, Yamagata K, Sugimoto K, et al. Modulation of microRNA processing by p53. Nature. 2009;460:529–33.
Vander Heiden MG, Cantley LC, Thompson CB. Understanding the Warburg effct: the metabolic requirements of cell proliferation. Science. 2009;324:1029–33.
Vogelstein B, Lane D, Levine AJ. Surfing the p53 network. Nature. 2000;408:307–10.
Vousden KH, Lane DP. p53 in health and disease. Nat Rev Mol Cell Biol. 2007;8:275–83.
Warburg O. On respiratory impairment in cancer cells. Science. 1956;124:269–70.
Wise DR, DeBerardinis RJ, Mancuso A, et al. Myc regulates a transcriptional program that stimulates mitochondrial glutaminolysis and leads to glutamine addiction. Proc Natl Acad Sci USA. 2008;105:18782–7.
Xiao C, Srinivasan L, Calado DP, et al. Lymphoproliferative disease and autoimmunity in mice with increased miR-17-92 expression in lymphocytes. Nat Immunol. 2008;9:405–14.
Yuneva M. Finding an “Achilles’ heel” of cancer: the role of glucose and glutamine metabolism in the survival of transformed cells. Cell Cycle. 2008;7:2083–9.
Zeller KI, Zhao X, Lee CW, et al. Global mapping of c-Myc binding sites and target gene networks in human B cells. Proc Natl Acad Sci USA. 2006;103:17834–9.
Zhang H, Bosch-Marce M, Shimoda LA, et al. Mitochondrial autophagy is an HIF-1-dependent adaptive metabolic response to hypoxia. J Biol Chem. 2008;283:10892–903.
Acknowledgments
I thank Dr. Chi Dang for his critical suggestions. Our original work is supported by NIH, Leukemia and Lymphoma Society, and Maryland Stem Cell Research Foundation grants.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2011 Springer Netherlands
About this chapter
Cite this chapter
Gao, P. (2011). MicroRNAs and Cancer Metabolism. In: Cho, W. (eds) MicroRNAs in Cancer Translational Research. Springer, Dordrecht. https://doi.org/10.1007/978-94-007-0298-1_20
Download citation
DOI: https://doi.org/10.1007/978-94-007-0298-1_20
Published:
Publisher Name: Springer, Dordrecht
Print ISBN: 978-94-007-0297-4
Online ISBN: 978-94-007-0298-1
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)