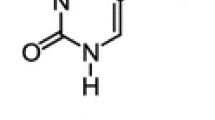
Abstract
The tetracyclines are a group of broad-spectrum antibiotics which, even 30 years after their discovery, have retained a prominent position in the treatment of infectious diseases.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Preview
Unable to display preview. Download preview PDF.
Similar content being viewed by others
References
Andre T, Ullberg S (1957) Radioactive tetracycline. J Am Chem Soc 79:494–495
Bakhtiar M, Selwyn S (1982) Comparative studies on the bactericidal activities of tetracyclines, chloramphenicol, and other “bacteriostatic” antibiotics. In: Periti P, Grassi GG (eds) Current chemotherapy and immunotherapy, proceedings of the 12th international congress of chemotherapy, vol. I. American Society for Microbiology, Washington, pp 76–77
Bakhtiar M, Selwyn S (1983) Antibacterial activity of a new thiatetracycline. J Antimicrob Chemother 11:291
Ball PR, Shales SW, Chopra I (1980) Plasmid-mediated tetracycline resistance in Escherichia coli involves increased efflux of the antibiotic. Biochem Biophys Res Commun 93:74–81
Barrett GC (1963) Synthesis of tetracycline analogs. J Pharm Sci 52:309–330
Beereboom JJ, Butler K (1962) Hydrolysis of 2-decarboxamido-2-cyano-6-deoxytetracycline derivatives. U.S. Patent 3,069, 467
Beereboom JJ, Ursprung JJ, Rennhard HH, Stephens CR (1960) Further 6-deoxytetracycline studies: effect of aromatic substituents on biological activity. J Am Chem Soc 82:1003–1004
Berlin YA, Kolosov MN, Vasina IV, Yartseva IV (1968) The structure of chromocyclomycin. Chem Commun 762–763
Bernardelli C, Bucher G, Lauria F, Logemann W, Tosolini G, Vita G (1967) Über die Einhorn-Reaktion mit Aminosäuren. Liebigs Ann Chem 706:243–249
Bernardi L, Castiglione R De, Colonna V, Masi P, Mazzoleni R (1974) Tetracycline derivatives. I - Esters of 5-oxytetracyclines: chemistry and biological activity. Farmaco (Sci) 29:902–909
Bernardi L, Castiglione R De, Scarponi U (1975 a) Simple methylation of amides. Chem Commun 320–321
Bernardi L, Castiglione R De, Colonna V, Masi P (1975 b) Tetracycline derivatives. Note II. A practical synthesis of minocycline. Farmaco (Sci) 30:736–741
Bernardi L, Castiglione R De, Masi P, Mazzoleni R, Scarponi U (1975 c) Tetracycline derivatives. Note III. 7- and 9-methyltetracyclines: synthesis and biological activity. Farmaco [Sci] 30:1025–1030
Bitha P, Hlavka JJ, Boothe JH (1970) 6-Fluorotetracyclines. J Med Chem 13:89–92
Blackwood RK, English AR (1970) Structure-activity relationships in the tetracycline series. Adv Appl Microbiol 13:237–266
Blackwood RK, Stephens CR (1964) Novel C-4 modified tetracycline derivatives. J Am Chem Soc 86:2736–2737
Blackwood RK, Stephens CR (1965) Some transformations of tetracycline at the 4-position. Can J Chem 43:1382–1388
Blackwood RK, Rennhard HH, Stephens CR (1960 a) Some transformations at the 12a-position of the tetracyclines. J Am Chem Soc 82:745–746
Blackwood RK, Rennhard HH, Stephens CR (1960 b) Some transformations at the 12a-position in the tetracycline series. J Am Chem Soc 82:5194–5197
Blackwood RK, Beereboom JJ, Rennhard HH, Schach von Wittenau M, Stephens CR (1961) 6-Methylenetetracyclines. A new class of tetracycline antibiotics. J Am Chem Soc 83:2773–2775
Blackwood RK, Beereboom JJ, Rennhard HH, Schach von Wittenau M, Stephens CR (1963) 6-Methylenetetracyclines. J Am Chem Soc 85:3943–3953
Boothe JH, Hlavka JJ (1978) Tetracyclines. Encycl Chem Tech 3:64–78
Boothe JH, Bonvicino GE, Waller CW, Petisi JP, Wilkinson RW, Broschard RB (1958) Chemistry of the tetracycline antibiotics. J Am Chem Soc 80:1654–1657
Boothe JH, Kende AS, Fields TL, Wilkinson RG (1959) Total synthesis of tetracyclines. I- (±)-Dedimethylamino-12a-deoxy-6-demethylanhydrochloro tetracycline. J Am Chem Soc 81:1006–1007
Boothe JH, Hlavka JJ, Petisi JP, Spencer JL (1960) 6-Deoxytetracyclines. I. Chemical modification by electrophilic substitution. J Am Chem Soc 82:1253–1254
Brown JR, Ireland DS (1978) Structural requirements for tetracycline activity. Pharmacol Chemother 15:161–202
Brunzell A (1962) N-Pyrrolidinomethyltetracycline. Stabilität in Lösungen und papierchromatographische Prüfung. Acta Chem Scand 16:245–246
Cammarata A, Yau SJ, Collett JH, Martin AN (1970) Correlation of the potencies of tetracyclines in vitro with a D-ring substituent Index. Mol Pharmacol 6:61–66
Chandra R, Dabral PK, Sharma MC, Mukerji S (1980) Water soluble tetracycline derivative: Part-II: 2-(D-glucosaminomethyl) tetracycline dihydrochloride. Indian J Pharm Sci 42:33–36
Cheney LC, Fayetteville, Risser WC, Gottstein WJ (1958) Mannich bases of tetracycline antibiotics. U.S. Patent 3,104, 240
Chopra I, Howe TGB, Linton AH, Linton KB, Richmond MH, Speller DCE (1981) The tetracyclines: prospects at the beginning of the 1980s. J Antimicrob Chemother 8:5–21
Chu DTW, Huckin SN (1980) Chemistry of hexamethyldisilazane. Silylation of ß-diketones and amination of ß-triketones. Can J Chem 58:138–142
Chu DTW, Huckin SN, Bernstein E, Garmaise DL, Egan RS, Stanaszek RS (1981) Chemistry of chelocardin. V. Condensation with amino reagents. Can J Chem 59:763–767
Church RFR, Schaub RE, Weiss MJ (1971) Synthesis of 7-dimethylamino-6-demethyl-6- deoxytetracycline (Minocycline) via 9-nitro-6-demethyl-6-deoxytetracycline. J Org Chem 36:723–725
Cid-Dresdner H (1965) The crystal structure of terramycin hydrochloride, C22H24N2O9.HCl. Z Kristallogr 121:170–189
Clive DLJ (1968) Chemistry of tetracyclines. Q Rev Chem Soc 22:435–456
Colaizzi JL, Klink PR (1969) pH-partition behavior of tetracyclines. J Pharm Sci 58:1184–1189
Collett JH, Collett C, Martin AN, Cammarata A (1970) The effects of some tetracyclines on synchronously growing cultures of Escherichia coli B/r. J Pharm Pharmacol 22:672–678
Conover LH, Butler K, Johnston JD, Korst JJ, Woodward RB (1962) The total synthesis of 6-demethyl-6-deoxytetracycline. J Am Chem Soc 84:3222–3224
de Carneri I, Coppi G, Lauria F, Logemann W (1961) Una nouva tetraciclina solubile: la tetraciclina-L-metilenlisina. Farmaco [Prat] 16:65–79
Dingeldein E, Ungethüm W (1979) Pharmacokinetics of EMD 33 330, a totally synthetic tetracycline. 11th International congress of chemotherapy and 19th interscience conference on antimicrobial agents chemotherapy, Boston, Oct 1979, Abstract 510
Dingeldein E, Wahlig H (1977) EMD 33 330, a new synthetic thiatetracycline. In vitro and in vivo evaluation. XVII. Interscience conference on antimicrobial agents and chemotherapy, New York, Oct 1977; Abstract 71
Doerschuk AP, Bitler BA, McCormick JRD (1955) Reversible isomerizations in the tetracycline family. J Am Chem Soc 77:4687
Donohue J, Dunitz JD, Trueblood KN, Webster MS (1963) The crystal structure of aureomycin (Chlortetracycline) hydrochloride. Configuration, bond distances and conformation. J Am Chem Soc 85:851–856
Dürckheimer W (1975) Tetracycline: Chemie, Biochemie und Struktur-Wirkungs-Beziehungen. Angew Chem 87:751–764
Esse RC, Lowery JA, Tamorria CR, Sieger GM (1964a) Tetracycloxides. I. A new class of tetracycline derivatives. J Am Chem Soc 86:3874–3875
Esse RC, Lowery JA, Tamorria CR, Sieger GM (1964 b) Tetracycloxides. II. Transformations at the C-4 position. J Am Chem Soc 86:3875–3877
Fabre J, Milek E, Kalfopoulos P (1971) La cinétique des tétracyclines chez l’homme. I. Absorption digestive et concentrations sériques. Schweiz Med Wochenschr 101:593–598
Fields TL, Kende AS, Boothe JH (I960) Total synthesis of tetracyclines. II. Stereospecific synthesis of (±)-dedimethylamino-6-demethyl-6,12a-dideoxy-7-chlorotetracycline. J AmChem Soc 82:1250–1251
Fields TL, Kende AS, Boothe JH (1961) Total synthesis of tetracyclines. V. The stereospecific elaboration of the tetracycline ring system. J Am Chem Soc 83:4612–4618
Fourtillan JB, Lefèbvre MA (1980) Corrélations structure-activité dans la famille des te-tracyclines. Nouv Presse Med 9:64–10
Free SM, Wilson JW (1964) A mathematical contribution to structure-activity studies. J Med Chem 1:395–399
Glatz B, Helmchen G, Muxfeldt H, Porcher H, Prewo R, Senn J, Stezowski JJ, Stojda RJ, White DR (1979) A total synthesis and structural aspects of racemic 8-oxygenated tetracyclines. J Am Chem Soc 101:2171–2181
Gottstein WJ, Minor WF, Cheney LC (1959) Carboxamido derivatives of the tetracyclines. J Am Chem Soc 81:1198–1201
Granatek ES, Kaplan MA, Buckwalter FH (1963) N-(3-phthalidyl)-tetracycline, a new carboxamido derivative of tetracycline. J Med Chem 6:202
Green A, Boothe JH (1960) Chemistry of the tetracycline antibiotics. III. 12a-Deoxytetracycline. J Am Chem Soc 82:3950–3953
Green A, Wilkinson RG, Boothe JH (1960) Chemistry of the tetracycline antibiotics. II. Bromination of dedimethylaminotetracyclines. J Am Chem Soc 82:3946–3950
Gulbis J, Everett W Jr (1975) A 13C Nuclear magnetic resonance analysis of the metal binding site in tetracycline. J Am Chem Soc 97:6248–6249
Gulbis J, Everett W Jr (1976) Metal binding characteristics of tetracycline derivatives in DMSO solution. Tetrahedron 32:913–917
Gurevich AI, Karapepetyan MG, Kolosov MN, Korobko VG, Onoprienko VV, Popravko SA, Shemyakin MM (1967) Synthesis of 12a-deoxy-5a,6-anhydrotetracycline. The first total synthesis of the naturally occurring tetracycline. Tetrahedron Lett 131–134
Hirokawa S, Okaya Y, Lovell FM, Pepinsky R (1959) Crystal structure of aureomycin hydrochloride. Z Krist 112:439–464
Hlavka JJ, Bitha P (1966) Photochemistry. IV. A photodeamination. Tetrahedron Lett 32:3843–3846
Hlavka JJ, Boothe JH (1973) The tetracyclines. Arzneimittelforsch 17:210–240
Hlavka J J, Buyske DA (1960) Radioactive 7-iodo-6-deoxy tetracycline in tumor tissue. Nature 186:1064–1065
Hlavka JJ, Krazinski HM (1963) The 6-deoxytetracyclines. VI. A photochemical transformation. J Org Chem 28:1422–1423
Hlavka JJ, Schneller A, Krazinski H, Boothe JH ( 1962 a) The 6-deoxy tetracyclines. III. Electrophilic and nucleophilic substitution. J Am Chem Soc 84:1426–1430
Hlavka JJ, Krazinski HM, Boothe JH ( 1962 b) The 6-deoxy tetracyclines. IV. A photochemical displacement of a diazonium group. J Org Chem 27:3674–3675
Hlavka JJ, Bitha P, Boothe JH (1968) 4-Hydroxypretetramids. J Am Chem Soc 90:1034–1037
Hochstein FA, Stephens CR, Conover LH, Regna PP, Pasternack R, Gordon PN, Pilgrim FJ, Brunings KJ, Woodward RB (1953) The structure of terramycin. J Am Chem Soc 75:5455–5475
Hochstein FA, Schach von Wittenau M, Tanner FW, Murai K (1960) 2-Acetyl-2-decarboxamidooxytetracycline. J Am Chem Soc 82:5934–5937
Holmlund CE, Andres WW, Shay AJ (1959) Chemical hydroxylation of 12a-deoxytetracycline. J Am Chem Soc 81:4748–4750
Hošt’álek Z, Blumauerová M, Vanek Z (1914) Genetic problems of the biosynthesis of tetracycline antibiotics. Adv Biochem Eng 3:13–67
Hüttenrauch R, Keiner I (1966) Epimerisierung von Mannich-Basen der Tetracyclin-Antibiotika. Naturwissenschaften 53:552
Hughes RE, Muxfeldt H, Dreele RB von (1971) Conformation of tetracycline ring systems. Structure of 5,12a-diacetyloxytetracycline. J Am Chem Soc 93:1037–1038
Hughes DW, Wilson WL, Butterfield AG, Pound NJ (1974) Stability of rolitetracycline in aqueous solution. J Pharm Pharmacol 26:79–80
Hughes LJ, Stezowski JJ, Hughes RE (1979) Chemical structural properties of tetracycline derivatives. 7. Evidence for the coexistence of the zwitterionic and nonionized forms of the free base in solution. J Am Chem Soc 101:7655–7657
Hussar DA, Niebergall PJ, Sugita ET, Doluisio JT (1968) Aspects of the epimerization of certain tetracycline derivatives. J Pharm Pharmacol 20:539–546
Jarowski CI (1963) Paper presented at the Sixth Pan American congress of pharmacy and biochemistry, Mexico City, December 1963
Jogun KH, Stezowski JJ (1976) Chemical-structural properties of tetracycline derivatives. 2. Coordination and conformation aspects of oxytetracycline metal ion complexation. J Am Chem Soc 98:6018–6026
Kelly RG (1964) Determination of anhydrotetracycline and 4-epianhydrotetracycline in a tetracycline mixture. J Pharm Sci 53:1551–1552
Kende AS, Fields TL, Boothe JH, Kushner S (1961) Total synthesis of tetracyclines. IV. Synthesis of an anhydrotetracycline derivative. J Am Chem Soc 83:439–449
Kirchlechner R (1981) Synthese von 6-Oxatetracyclin, einem weiteren Heteroanalogen Tetracyclin. Tetrahedron Lett 22/16:1497–1500
Kirchlechner R, Rogalski W (1980) Synthesis of 6-thiatetracycline, a highly active analogue of the antibiotic tetracycline. Tetrahedron Lett 21:247–250
Kirchlechner R, Seubert J (1982) Synthese von 6-Thiaminocyclin, einem Thiaanalogen des Antibiotikums Minocyclin. Arch Pharm 315:519–525
Kirchlechner R, Rogalski W, Dingeldein E, Wahlig H (1977) Thiatetracyclines, a new class of totalsynthetic tetracyclines. XVII. Interscience conference on antimicrobial agents and chemotherapy, New York, Oct 1977, Abstract 70
Kollat P, Stezowski JJ (1982) Nonionized 5a-epi-6-oxatetracycline free base. Acta Cryst B 38:2531–2533
Korst JJ (1972a) In 2-Stellung substituierte Tetracycline. Deutsche Offenlegungsschrift (Patent) 2065:149
Korst JJ (1972 b) Verfahren zur Herstellung von 2-decarboxamido-2-iminotetracyclinen, bzw. Ihren Säureadditionssalzen oder Metallsalzen. Deutsche Offenlegungsschrift (Patent) 2049:941
Korst JJ, Johnston JD, Butler K, Bianco EJ, Conover LH, Woodward RB (1968) The total synthesis of d,l-6 demethyl-6-deoxytetracycline. J Am Chem Soc 90:439–457
Koschel K. Hartmann G, Kersten W, Kersten H (1966) Die Wirkung des Chromomycins und einiger Anthracyclinantibiotica auf die DNA-abhängige Nucleinsäure-Synthese. Biochem Z 244:76–86
Last JA (1969) Studies on the binding of tetracycline to ribosomes. Biochim Biophys Acta 195:506–514
MacDonald H, Kelly RG, Allen ES, Noble JF, Kanegis LA (1973) Pharmacokinetics studies on minocycline in man. Clin Pharmacol Ther 14:852–861
Martell MJ, Boothe JH (1967) The 6-deoxytetracycline. VII. Alkylated aminotetracyclines possessing unique antibacterial activity. J Med Chem 10:44–46
Martell MJ Jr, Ross AS, Boothe JH ( 1967 a) The 6-deoxytetracycline. IX. Imidomethylation. J Med Chem 10:359–363
Martell MJ Jr, Ross AS, Boothe JH ( 1967 b) The 6-deoxytetracyclines. VIII. Acylaminomethylamides. J Med Chem 10:485–486
Martell MJ Jr, Ross AS, Boothe JH (1967 c) The dehydrotetracyclines. I. Epimerization at C-6. J Am Chem Soc 89:6780–6781
Martin W, Hartung H, Urbach H, Dürckheimer W (1973) Synthesen in der Tetracyclinreihe. I. Totalsynthese von d,l-7-Chlor-6-desoxytetracyclinen und d,l-7-Chlor-6-desmethyl-6-desoxytetracyclinen der natürlichen, der 5a-epi- und der 6-epi-Reihe. Tetrahedron Lett 3513–3516
McCapra F, Scott AI, Delmotte P, Delmotte-Plaquée J (1964) The constitution of monorden, an antibiotic with tranquilising action. Tetrahedron Lett 869–875
McCormick JRD, Jensen ER (1968) Biosynthesis of tetracyclines. J Am Chem Soc 90:7126–7127
McCormick JRD, Fox SM, Smith LL, Bitler BA, Reichenthal J, Origoni VE, Muller WH, Winterbottom R, Doerschuk AP (1956) On the nature of the reversible isomerizations occurring in the tetracycline family. J Am Chem Soc 78:3547–3548
McCormick JRD, Fox SM, Smith LL, Bitler BA, Reichenthal J, Origoni VE, Muller WH, Winterbottom R, Doerschuk AP (1957) Studies of the reversible epimerization occurring in the tetracycline family. The preparation, properties and proof of structure of some 4-epitetracyclines. J Am Chem Soc 79:2849–2858
McCormick JRD, Miller PA, Growich JA, Sjolander NO, Doerschuk AP (1958) Two new tetracycline-related compounds: 7-chloro-5a(11a)-dehydrotetracycline and 5a-epi-tetracycline. A new route to tetracycline. J Am Chem Soc 80:5572–5573
McCormick JRD, Jensen ER, Miller PA, Doerschuk AP (1960) The 6-deoxy tetracyclines. Further studies on the relationship between structure and antibacterial activity in the tetracycline-series. J Am Chem Soc 82:3381–3386
McCormick JRD, Winterbottom R, Bitha P (1965) Novel 7,11a-dihalotetracyclines and the process of preparing them. U.S. Patent 3,226, 435
McMurry L, Petrucci RE, Levy SB (1980) Active efflux of tetracycline encoded by four genetically different tetracycline resistance determinants in Escherichia coli. Proceedings of the national academy of sciences 77:3974–3977
McMurry L, Cullinane JC, Levy SB (1982) Transport of the lipophilic analog minocycline differs from that of tetracycline in susceptible and resistant Escherichia coli strains. Antimicrob Agents Chemother 5:791–799
Miller MW, Hochstein FA (1962) Isolation and characterization of two new tetracycline antibiotics. J Org Chem 27:2525–2528
Mitscher LA (1978) The chemistry of the tetracycline antibiotics. Med Res Ser 9
Mitscher LA, Bonacci AC, Sokoloski TD (1968) Circular dichroism and solution conformation of the tetracycline antibiotics. Antimicrob Agents Chemother 78–86
Mitscher LA, Bonacci AC, Slater-Eng B, Hacker AW, Sokoloski TD (1969) Interaction of various tetracyclines with metallic cations in aqueous solutions as measured by circular dichroism. Antimicrob Agents Chemother 111–115
Mitscher LA, Rosenbrook WM Jr, Andres WW, Egan RS, Schenck J, Juvarkar JV (1970) Structure of chelocardin, a novel tetracycline antibiotic. Antimicrob Agents Chemother 38–41
Mitscher LA, Slater-Eng B, Sokoloski TD (1972) Circular dichroism measurements of the tetracyclines. IV. 5-Hydroxylated derivatives. Antimicrob Agents Chemother 2:66–72
Money T, Scott AI (1968) Recent advances in the chemistry and biochemistry of tetracyclines. Prog Org Chem 7:1–34
Muxfeldt H (1962) Synthesen der Tetracyclin-Reihe. Angew Chem 74/13:443–452
Muxfeldt H, Kreutzer A (1961) Tetracycline. II. Synthese des Desdimethylamino-anhydroaureomycins. Chem Ber 94:881–893
Muxfeldt H, Rogalski W (1965) Tetracyclines. V. A total synthesis of (±)-6-deoxy-6-demethyltetracycline. J Am Chem Soc 87:933–934
Muxfeldt H, Hardtmann G, Kathawala F, Vedejs E, Mooberry JB (1968) Tetracyclines. VII. Total synthesis of dl-Terramycin. J Am Chem Soc 90:6534–6536
Muxfeldt H, Döpp H, Kaufmann JE, Schneider J, Hansen PE, Sasaki A, Geiser T (1973) Totalsynthese des Anhydro-aureomycins. Angew Chem Int Ed Engl 12:508–510
Muxfeldt H, Haas G, Hardtmann G, Kathawala F, Mooberry JB, Vedejs E (1979) Tetracyclines. 9. Total synthesis of dl-Terramycin. J Am Chem Soc 101:689–701
Neu HC (1978) Symposium on the tetracyclines: a major appraisal. Bulletin NY Acad Med 54:141–145
Nikaido H, Nakae T (1979) The outer membrane of gram-negative bacteria. Adv Microb Physiol 20:163–250
Notari RE (1973) Pharmacokinetics and molecular modification: implications in drug design and evaluation. J Pharm Sci 62:865–880
Oliver TJ, Prokop JF, Bower RR, Otto RH (1962) Chelocardin, a new broad-spectrum antibiotic. I. Discovery and biological properties. Antimicrob Agents Chemother 583–591
Pelcak E, Dornbush A (1948) Aureomycin - a new antibiotic. Ann NY Acad Sci 51:218
Peradejordi F, Martin AN, Cammarata A (1971) Quantum chemical approach to structure-activity relationships of tetracycline antibiotics. J Pharm Sci 60:576–582
Petisi J, Spencer JL, Hlavka JJ, Boothe JH (1962) 6-Deoxytetracyclines. III. Nitrations and subsequent reactions. J Med Pharm Chem 5:538–546
Prewo R, Stezowski JJ (1977) Chemical structural properties of tetracycline derivatives. 3. The integrity of the conformation of the nonionized free base. J Am Chem Soc 99:1117–1121
Prewo R, Stezowski JJ (1980) Chemical-structural properties of tetracycline derivatives. 9. 7-Chlorotetracycline derivatives with modified stereochemistry. J Am Chem Soc 102:7015–7020
Prewo R, Stezowski JJ, Kirchlechner R (1980) Chemical-structural properties of tetracycline derivatives. 10. The 6-thiatetracyclines. J Am Chem Soc 102:7021–7026
Purcell WP, Bass GE, Clayton JM (1973) Strategy of drug design, a molecular guide to biological activity. Wiley, New York
Purich SD, Colaizzi JL, Poust RI (1973) pH-partition behaviour of amino acid-like ß-lactum antibiotics. J Pharm Sci 62:545–549
Redin GS (1966) Antibacterial activity in mice of minocycline, a new tetracycline. Antimicrob Agents Chemother 371–376
Remmers EG, Sieger GM, Doerschuk AP (1963) Some observations on the kinetics of the C-4 epimerization of tetracycline. J Pharm Sci 52:752–756
Rennhard HH, Blackwood RK, Stephens CR (1961) Fluortetracyclines. I. Perchloryl fluoride studies in the tetracycline series. J Am Chem Soc 83:2775–2777
Rigler NE, Bag SP, Leyden DE, Sudmeier JL, Reilley CN (1965) Determination of a protonation scheme of tetracycline using nuclear magnetic resonance. Anal Chem 37:872–875
Rogalski W (1978) Structure-activity relationships of thiatetracyclines. XVIII. Interscience conference on antimicrobial agents and chemotherapy, Atlanta, Oct 1978
Russell AD, Ahonkhai I (1982) Antibacterial activity of a new thiatetracycline antibiotic, thiacycline, in comparison with tetracycline, doxycycline and minocycline. J Antimicrob Chemother 9:445–449
Schach von Wittenau M, Beereboom JJ, Blackwood RK, Stephens CR (1962) 6-Deoxytetracyclines. III. Stereochemistry at C-6. J Am Chem Soc 84:2645–2647
Schach von Wittenau M, Hochstein FA, Stephens CR (1963) Tautomerism of 5a-11a-dehydro-7-chlorotetracycline. Preparation of 5-alkoxy-7-chloranhydrotetracyclines. J Org Chem 28:2454–2456
Schach von Wittenau M, Blackwood RK, Conover LH, Glavert RH, Woodward RB (1965) The stereochemistry at C-5 in Oxytetracycline. J Am Chem Soc 87:134–135
Schach von Wittenau M, Blackwood RK (1966) Proton magnetic resonance; spectra of tetracyclines. J Org Chem 31:613–615
Schwarz JSP, Applegate HE (1967) Chemistry of tetracyclines. II. Allotetracycline and allooxytetracycline. J Org Chem 32:1241–1243
Schwarz JSP, Applegate HE, Bouchard JL, Weisenborn FL (1967) Chemistry of tetracyclines. I. Mercuric acetate oxidation of tetracycline. J Org Chem 32:1238–1241
Scott AI, Bedford CT (1962) Simulation of the biosynthesis of tetracyclines. A partial synthesis of tetracycline from anhydroaureomycin. J Am Chem Soc 84:2271–2274
Shu P (1966) Oxidative preparation of 9-hydroxytetracyclines. J Am Chem Soc 88:4529–4530
Siedel W, Söder A, Lindner F (1958) Die Aminomethylierung der Tetracycline. Zur Chemie des Reverin. Münch Med Wochenschr 17:661–663
Sinclair AC, Schenck JR, Post GG, Cardinal EV, Burokas S, Fricke HH (1962) Chelocardin, a new broad-spectrum antibiotic. II. Isolation and characterization. Antimicrob Agents Chemother 592–595
Spencer JL, Hlavka JJ, Petisi J, Krazinski HM, Boothe JH (1963) 6-Deoxytetracyclines. V. 7,9-Disubstituted products. J Med Chem 6:405–407
Steigbigel NH, Reed CW, Finland M (1968) Susceptibility of common pathogenic bacteria to seven tetracycline antibiotics in vitro. Am J Med Sci 255:179–195
Stephens CR (1962) U.S. Patent 3028 409 (assigned to Chas. Pfizer and Co., Inc.)
Stephens CR, Conover LH, Pasternack R, Hochstein FA, Moreland WT, Regna PP, Pilgrim FJ, Brunings KJ, Woodward RB (1954) The structure of aureomycin. J Am Chem Soc 76:3568–3575
Stephens CR, Bianco EJ, Pilgrim FJ (1955) A new reagent for dehydrating primary amides under mild conditions. J Am Chem Soc 77:1701–1702
Stephens CR, Conover LH, Gordon PN, Pennington FC, Wagner RL, Brunings KJ, Pilgrim FJ (1956) Epitetracycline - the chemical relationship between tetracycline and “Quatrimycin”. J Am Chem Soc 78:1515–1516
Stephens CR, Beereboom JJ, Rennhard HH, Gordon PN, Murai K, Blackwood RK, Schach von Wittenau M (1963) 6-Deoxytetracyclines. IV. Preparation, C-6 stereochemistry, and reactions. J Am Chem Soc 85:2643–2652
Stezowski JJ (1976) Chemical-structural properties of tetracycline derivatives. 1. Molecular structure and conformation of the free base derivatives. J Am Chem Soc 98:6012–6018
Stezowski JJ (1977) Chemical-structural properties of tetracycline antibiotics. 5. Ring A tautomerism involving the protonated amide substituent as observed in the crystal structure of a-6-deoxytetracycline hydrohaldes. J Am Chem Soc 99:1122–1129
Stezowski JJ (1982) Steric effects - Intramolecular information transfer in tetracycline antibiotics. In: Náray-Szabó (ed) Proceedings of the symposium on steric effects in biomolecules. Academia IAI Kiadó, Budapest, 1982, pp 3–13
Stezowski JJ, Prewo R (1980) Chemical-structural properties of tetracycline derivatives. Abstr Pap Am Chem Soc Meet Phys 179:87
Takeuchi Y, Buerger MJ (1960) The crystal structure of Terramycin hydrochloride. Proc Natl Acad Sci USA 46:1366–1370
Tamorria CR, Esse RC (1965) Alkoxyalkyltetracyclines. J Med Chem 8:870–872
Teare EL, Bakhtiar M, Selwyn S (1981) Comparative in vitro studies of a new thiatetracycline and three existing tetracyclines. Drugs Exp Clin Res VII (3):307–311
Terada H, Inagi T (1975) Proposed partition mechanism of tetracycline. Chem Pharm Bull 23:1960–1968
Toon S, Rowland M (1979) Quantitative structure pharmacokinetic activity relationships with some tetracyclines. J Pharm Pharmacol [Suppl] 31:43 P
Tritton TR (1977) Ribosome-tetracycline interactions. Biochemistry 16:4133–4138
Tubaro E, Banci F (1964) The pharmacology of chlormethylenecycline, a new chlortetracycline. Arzneimittelforsch 5:95–100
Tute MS (1975) Lipophilicity. Chem Ind (Lond) 100–105
Ungethüm W, Dingeldein E, Pabst J, Leopold G, Diekmann H (1980) Clinical pharmaco-kinetics of EMD 33 330, a new totally synthetic tetracycline. World conference on clinical pharmacology and therapuetics, London, 3rd-9th August 1980. Abstract No. 0438
Urbach H, Hartung H, Martin W, Dürckheimer W (1973) Synthesen in der Tetracyclinreihe. II. Totalsynthese von dl-4-Amino-7-chlor-2-N-methylcarbamyl-2-descarbamyl-4- desdimethylamino-6-desmethyl-6-desoxytetracycline. Tetrahedron Lett 1973:4907–4910
Valcavi U, Campanella G, Pacini N (1963) Pirazolderivati della tetracyclina e clorotetracyclina. Gazz Chim Ital 93:916–928
Valcavi U, Brandt A, Corsi GB, Minoja F, Pascucci G (1981) Chemical modifications in the tetracycline series. J Antibiotics 34:34–39
Vaněk Z, Cudlin J, Blumauerova M, Hošt’álek Z (1974) How many genes are required for the synthesis of chlortetracycline? Folia Microbiol (Prague) 16:225–240
van den Hende JH (1965) The crystal and molecular structures of 7-chloro- and 7-bromo-4-hydroxytetracycloxide. J Am Chem Soc 87:929–931
von Dreele RB, Hughes RE (1971) Crystal and molecular structure of 5,12a-diacetyloxy-tetracycline. J Am Chem Soc 93:7290–7296
Welling PG, Koch PA, Lau CC, Craig WA (1977) Bioavailability of tetracycline and doxycycline in fasted and nonfasted subjects. Antimicrob Agents Chemother 11:462–469
Wilkinson RG, Boothe JH (1961) Deutsches Patentamt (GP) Auslegeschrift 1, 1088, 481: Verfahren zur Herstellung neuer Verbindungen der Tetracylinreihe
Williamson DE, Everett W Jr (1975) A proton nuclear magnetic resonance study of the site of metal binding in tetracycline. J Am Chem Soc 97:2397–2405
Woodward RB (1963) The total synthesis of a tetracycline. Pure Appl Chem 6:561–573
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 1985 Springer-Verlag Berlin Heidelberg
About this chapter
Cite this chapter
Rogalski, W. (1985). Chemical Modification of the Tetracyclines. In: Hlavka, J.J., Boothe, J.H. (eds) The Tetracyclines. Handbook of Experimental Pharmacology, vol 78. Springer, Berlin, Heidelberg. https://doi.org/10.1007/978-3-642-70304-1_5
Download citation
DOI: https://doi.org/10.1007/978-3-642-70304-1_5
Publisher Name: Springer, Berlin, Heidelberg
Print ISBN: 978-3-642-70306-5
Online ISBN: 978-3-642-70304-1
eBook Packages: Springer Book Archive