Abstract
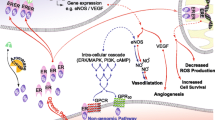
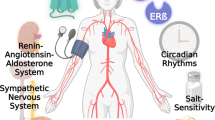
There is a sexual dimorphism in the development of atherosclerotic cardiovascular disease in humans (1). The incidence of cardiovascular disease is much lower in premenopausal women than in age matched men, but rises steadily in women after the menopause (2). Epidemiologic and observational clinical studies have shown that postmenopausal women on estrogen replacement therapy have less severe coronary artery disease and a lower risk of cardiovascular mortality than women without hormone treatment (3). Thus, ovarian hormones, principally estrogens, appear to inhibit the development of atherosclerotic cardiovascular disease in women (4). Large-scale clinical studies, including the Women’s Health Initiative (WHI) and the Heart Estrogen-Progestin Replacement Study (HERS), are currently evaluating the role of estrogen in the prevention of cardiovascular morbidity and mortality. Emerging evidence from these and other studies suggests that estrogen may be cardioprotective through a combination of mechanisms. For example, estrogen replacement therapy results in decreased plasma low-density lipoprotein (LDL) and increased high-density lipoprotein (HDL) concentrations (5-7). Other benefits of estrogen replacement therapy, including the improvement of systemic hemodynamic parameters, appear to be unrelated to changes in plasma lipoprotein profiles (8). It has been clearly shown that endothelial cells and vascular smooth muscle cells (VSMCs) possess estrogen receptors (ER) and are thus physiological targets for estrogen action (9,10).
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Preview
Unable to display preview. Download preview PDF.
Similar content being viewed by others
References
Glendy RE, Levine SA, White PD. Coronary disease in youth: comparison of 100 patients under 40 with 300 persons past 80. JAMA 109: 1775–1781, 1937.
Colditz GA, Willett WC, Stampfer MJ, Rosner B, Speizer FE, Hennekens CH. Menopause and the risk of coronary heart disease in women. N Engl J Med 316: 1105–1110, 1987.
Bush TL, Barrett-Connor E, Cowan LD, Criqui MH, Wallace RB, Suchindran CM, Tyroler HA, Rifkind BM: Cardiovascular mortality and non contraceptive use of estrogen in women: results from the Lipid Research Clinics Program Follow-up Study. Circulation 75: 1102–1109 1987.
Stampfer MJ, Colditz GA, Willett WC, Manson JE, Rosner B, Speizer FE, Hennekens CH. Postmenopausal estrogen therapy and cardiovascular disease. N Engl J Med 325: 756–762, 1991.
Knopp RH, Zhu X, Bonet B. Effects of estrogens on lipoprotein metabolism and cardiovascular disease in women. Atherosclerosis 110: S83–S91, 1994.
Grodstein F, Stampfer M. The epidemiology of coronary heart disease and estrogen replacement in postmenopausal women. Prog Cardiovasc Dis 38: 199–210, 1995.
Guetta V, Cannon RO III: Cardiovascular effects of estrogen and lipid-lowering therapies in postmenopausal women. Circulation 93: 1928–1937, 1996.
Williams JK, Kim YD, Adams MR, Chen MF, Myers AK, Ramwell PW. Effects of estrogen on cardiovascular responses of premenopausal monkeys. J Pharmacol Exp Ther 271: 671–676, 1994.
Orimo A, Inoue S, Ouchi Y, Orimo H. Vascular smooth muscle cells possess estrogen receptors and respond to estrogen. Ann NY Acad Sci 748: 592–594, 1995.
Farhat MY, Abi-Younes S, Ramwell PW. Non-genomic effects of estrogen and the vessel wall. Biochem Pharmacol 51: 571–576, 1996.
Foegh ML, Zhao Y, Farhat M, Ramwell PW. Oestradiol inhibition of vascular myointimal proliferation following immune, chemical and mechanical injury. Ciba Found Symp 191: 139–145, 1995.
Ross R. The pathogenesis of atherosclerosis: a perspective for the 1990s. (Review) Nature 362: 801–809, 1993.
Stamler J, Pick R, Katz LN. Prevention of coronary atherosclerosis by estrogen-androgen administration in the cholesterol-fed chick. Circ Res 1: 94–98, 1953.
Fischer-Dzpga K, Wissler RW, Vesselinovitch D. The effect of estradiol on the proliferation of rabbit aortic medial tissue culture cells induced by hyperlipemic serum. Exper Mol Pathol 39: 355–363, 1983.
Vargas R, Wroblewska B, Rego A, Hatch J, Ramwell PW. Oestradiol inhibits smooth muscle cell proliferation of pig coronary artery. Br J Pharmacol 109: 612–617, 1993.
Moskowitz MS, Moskowitz AA, Bradford WL, Wissler RW: Changes in serum lipids and coronary arteries of the rat in response to estrogens. Arch Pathol 61: 245–263, 1956.
Constantinides P, Gutmann-Auersperg N, Hospes D, Williams K. Estradiol and prednisone in rabbit atherosclerosis. Arch Pathol 73: 277–280, 1962.
Rhee CY, Spaet TH, Temerman MB, Lajam F, Shiang HH. Estrogen suppression of surgically induced vascular intimal hyperplasia in rabbits. J Lab Clin Med 90: 77–84, 1977.
Foegh ML, Khirabadi BS, Nakanishi T, Vargas R, Ramwell PW. Estradiol protects against experimental cardiac transplant atherosclerosis. Transplant 19: 90–95, 1987.
Foegh M, Asotra S, Howell M, Ramwell P: Estradiol inhibition of arterial neointimal hyperplasia after balloon injury. J Vase Surg 19: 722–726, 1994.
Sullivan TR Jr, Karas RH, Aronovitz M, Faller GT, Ziar JP, Smith JJ, ODonnell TF, Mendelsohn ME. Estrogen inhibits the response-to-injury in a mouse carotid artery model. J Clin Investp 96: 2482–2488, 1995.
Chen SJ, Li H, Durand J, Oparil S, Chen YF. Estrogen reduces myointimal proliferation after balloon injury of rat carotid artery. Circulation 93: 577–584, 1996.
Levine RL, Chen SJ, Durand J, Chen YF, Oparil S. Medroxyprogesterone attenuates estrogenmediated inhibition of neointima formation after balloon injury of the rat carotid artery. Circulation 94: 2221–2227, 1996.
Oparil S, Levine RL, Chen SJ, Durand J, Chen YF. Sexually dimorphic response of the balloon injured rat carotid artery to hormone treatment. Circulation 95: 1301–1307, 1997.
Iafrati MD, Karas RH, Aronovitz M, Kim S, Sullivan TR, Lubahn DB, O’Donnell TF, Korach KS, Mendelssohn ME. Estrogen inhibits the vascular injury response in estrogen receptor α-deficient mice. Nature Med 3: 545–548, 1997.
White CR, Darley-Usmar V, Oparil S. Gender and cardiovascular disease: recent insights. Trends Cardiovasc Med 7: 94–100, 1997.
White CR, Shelton J, Chen SJ, Darley-Usmar V, Allen L, Nabors C, Sanders PW, Chen YF, Oparil S. Estrogen restores endothelial cell function in an experiemental model of vascular injury. Circulation 96: 1624–1630, 1997.
Oparil S, Chen SJ, Chen YF, Durand JN, Allen L, Thompson JA. Estrogen reduces cellular proliferation following carotid artery balloon injury in ovariectomized rats. Circulation 96: 607, 1997. (Abstract)
Li G, Chen SJ, Chen YF, Durand J, Oparil S, Thompson JA. Adventitial fibroblast activation and migration contribute to neointima formation in balloon injured rat carotid arteries. In Press.
Lindner V, Fingerle J, Reidy MA. Mouse model of arterial injury. Circ Res 73: 792–796, 1993.
Kuiper GGJM, Enmark E, Pelto-Huikko M, Nilsson S, Gustafsson JA. Cloning of a novel receptor expressed in rat prostate and ovary. Proc Natl Acad Sci USA 93: 5925–5930, 1996.
Kuiper GGJM, Carlsson B, Grandien K, Enmark E, Haggblad J, Nilsson S, Gustafsson JA. Comparison of the ligand binding specificity and tissue distribution of estrogen receptors α and P. Endocrinology 138: 863–870, 1997.
Paech K, Webb P, Kuiper GGJM, Nilsson S, Gustafsson J-A, Kushner PJ, Scanlan TS: Differential ligand activation of estrogen receptors ERα and ERβ at API sites. Science 277: 1508–1510, 1997.
PEPI Trial Writing Group. Effects of estrogen or estrogen/progestin regimens on heart disease risk factors in postmenopausal women. The Postmenopausal Estrogen/Progestin Interventions (PEPI) Trial. JAMA 273: 199–208, 1995.
Miller VM, Vanhoutte PM. Progesterone and modulation of endothelium-dependent responses in canine coronary arteries. Am J Physiol 261: R1022–R1027, 1991.
Williams JK, Honore EK, Washburn SA, Clarkson TB. Effects of hormone replacement therapy on reactivity of atherosclerotic coronary arteries in cynomolgus monkeys. J Am Coll Cardiol 24: 1757–1761, 1994.
Gebara OCE, Mittleman MA, Sutherland P, Lipinska I, Matheney T, Xu P, Welty FK, Wilson PWF, Levy D, Muller JE, Toiler GH. Association between increased estrogen status and increased fibrinolytic potential in4he Framingham Offspring Study. Circulation 91: 1952–1958, 1995.
Miyagawa K, Rosch, Stanczyk F, Hermsmeyer K. Medroxyprogesterone interferes with ovarian steroid protection against coronary vasospasm. Nat Med 3: 324–327, 1997.
Williams JK, Adams MR. Estrogens, progestins and coronary artery reactivity. Nat Med; 3: 273–274, 1997.
Grodstein F, Stampfer MJ, Manson JE, Colditz GA, Willett WC, Rosner B, Speizer FE, Hennekens CH. Postmenopausal estrogen and progestin use and the risk of cardiovascular disease. N Engl J Med 335: 453–461, 1996.
Lee W-S, Harder J A, Yoshizumi M, Lee M-E, and Haber E. Progesterone inhibits arterial smooth muscle cell proliferation. Nat Med 3: 1005–1008, 1997.
Collins P, Rosano GMC, Sarrel PM, Ulrich L, Adamapoulos S, Beale CM, McNeill JG, Poole-Wilson PA. 17P-estradiol attenuates acetylcholine-induced coronary arterial constriction in women but not men with coronary heart disease. Circulation 92: 24–30, 1995.
Blumenthal R, Heldman AW, Brinker JA, Resar JR, Coombs VJ, Gloth ST, Gerstenblith G, Reis SE. Acute effects of conjugated estrogens on coronary blood flow response to acetylcholine in men. Am J Cardiol 80: 1021–1024, 1997.
Bhoopalam VB, Murali S, Counihan PJ, Conrad Smith AG, Feldman MD, Cohen HA, Pham SM, Zell KA, Reis SE. Estrogen acutely abolishes abnormal coronary vasoconstriction in male cardiac transplant recipients. J Am Coll Cardiol 27: 284A, 1996. (Abstract)
Reis SE, Bhoopalam V, Zell KA, Couhnihan PJ, Conrad Smith AJ, Pham S, Murali SM. Conjugated estrogens acutely abolish abnormal cold-induced coronary vasoconstriction in male cardiac allografts. Circulation 97: 23–25, 1998.
Washburn SA, Honore EK, Cline JM, Helman M, Wagner JD, Adelman SJ, Clarkson TB. Effects of 17α-dihydroequilenin sulfate on atherosclerotic male and female rhesus monkeys. Am J Obstet Gynecol 175: 341–351, 1996.
Sudhir K, Chou TM, Mullen WL, Hausmann D, Collins P, Yock PG, Chatterjee K. Mechanisms of estrogen-induced vasodilation: in vivo studies in canine coronary conductance and resistance arteries. J Am Coll Cardiol 26: 807–814, 1995.
Tang W, Chen YF, Oparil S. Estrogen, but not androgen, inhibits the proliferation of cultured vascular smooth muscle cells, derived from male and female spontaneously hypertensive rats. J Invest Med 44: 68A, 1996.
Venkov C, Rankin A, Vaughan DE. Identification of authentic estrogen receptors in cultured endothelial cells. Circulation 94: 727–733, 1996.
Kim-Schulze S. McGowan KA, Hubchak SC, Cid MC, Martin MB, Leinman HK, Greene GL, Schnaper HW. Expression of an estrogen receptor by human coronary artery and umbilical vein endothelial cells. Circulation 94: 1402–1407, 1996.
Fischer GM, Bashey RI, Rosenbaum H, Lyttle CR. A possible mechanism in arterial wall for mediation of sex difference in atherosclerosis. Exper Mol Pathol 43: 288–296, 1985.
Alexander JJ, Hoenig M, Graham D, Imbembo AL. The effect of estrogen on low-density lipoprotein binding kinetics in aortic endothelial cells. Surgery 106: 386–391, 1989.
Williams JK, Adams MR, Klopfenstein HS. Estrogen modulates responses of atherosclerotic coronary arteries. Circulation 81: 1680–1687, 1990.
Williams JK, Adams MR, Herrington DM, Clarkson TB. Short-term administration of estrogen and vascular responses of atherosclerotic coronary arteries. J Am Coll Cardiol 20: 452–457, 1992.
Reis SE, Gloth ST, Blumenthal RS, Resar JR, Zacur HA, Gerstenblith G, Brinker JA. Ethinyl estradiol acutely attenuates abnormal coronary vasomotor responses to acetylcholine in postmenopausal women. Circulation 89: 52–60, 1994.
Keaney JF Jr, Shwaery GT, Xu A, Nicolosi RJ, Loscalzo J, Foxall TL, Vita JA. 17β-estradiol preserves endothelial vasodilator function and limits low-density lipoprotein oxidation in hypercholesterolemic swine. Circulation 89: 2251–2259, 1994.
Gilligan DM, Quyyumi AA, Cannon RO, III. Effects of physiological levels of estrogen on coronary vasomotor function in postmenopausal women. Circulation 89: 2545–2551, 1994.
Rifici VA, Khachadurian AK. The inhibition of low-density lipoprotein oxidation by 17-β estradiol. Metabolism: Clinical & Experimental 41: 1110–1114, 1992
Weiner CP, Lizasoain I, Baylis SA, Knowles RG, Charles IG, Moncada S. Induction of calcium-dependent nitric oxide synthases by sex hormones. Proc Natl Acad Sci USA 91: 5212–5216, 1994.
Palmer RMJ, Ferrige AG, Moncada S. Nitric oxide release accounts for the biological activity of endothelium-derived relaxing factor. Nature 327: 524–526, 1987.
Chang WC, Nakao J, Orimo H, Murota SI. Stimulation of prostaglandin cyclooxygenase and prostacyclin synthetase activities by estradiol in rat aortic smooth muscle cells. Biochim Biophys Acta 620: 472–479, 1980.
Hayashi T, Fukuto JM, Ignarro LJ, Chaudhuri G. Basal release of nitric oxide from aortic rings is greater in female rabbits than in male rabbits: implications for atherosclerosis. Proc Natl Acad Sci USA 89: 11259–11263, 1992.
Mendelsohn ME, Karas RH. Estrogen and the blood vessel wall. Curr Opin Cardiol 9: 619–626, 1994.
White CR, Darley-Usmar VM, Oparil S. Gender and cardiovascular disease:recent insights. Trends Cardiovasc Med 7: 94–100, 1997.
Jiang C, Sarrel PM, Poole-Wilson PA, Collins P. Acute effect of 17β-estradiol on rabbit coronary artery contractile responses to endothelin-1. Am J Physiol 263: H271–H275, 1992.
Harder DR, Coulson PB. Estrogen receptors and effects of estrogen on membrane electrical properties of coronary vascular smooth muscle. J Cell Physiol 100: 375–382, 1979.
Zhang F, Ram JL, Standley PR, Sowers JR. 17β-estradiol attenuates voltage-dependent Ca2+ currents in A7r5 vascular smooth muscle cell line. Am J Physiol 266: C975–C980, 1994.
Cooke JP, Tsao PS. Cytoprotective effects of nitric oxide. Circulation 88: 2451–2452, 1993.
Morales DE, McGowan KA, Grant DS, Maheshwari S, Bhartiya D, Cid MC, Kleinman HK, Schnaper HW. Estrogen promotes angiogenesis activity in human umbilical vein endothelial cells in vitro and in a murine model. Circulation 91: 755–763, 1995.
Cid MC, Kleinman HK, Grant DS, Schnaper HW, Fauci AS, Hoffman GS. Estradiol enhances leukocyte binding to tumor necrosis factor (TNF)-stimulated endothelial cells via an increase in TNF-induced adhesion molecules E-selectin, intercellular adhesion molecule type 1 and vascular cell adhesion molecule type 1. J Clin Invest 93: 17–25, 1994.
Shweiki D, Itin A, Neufeld G, Gitay-Goren H, Keshet E. Patterns of expression of vascular endothelial growth factor (VEGF) and VEGF receptors in mice suggest a role in hormonally regulated angiogenesis. J Clin Invest 91: 2235–2243, 1993.
Cullinan-Bove K, Koos RD. Vascular endothelial growth factor/vascular permeability factor expression in the rat uterus:rapid stimulation by estrogen correlates with estrogen-induced increases in uterine capillary permeability and growth. Endocrinology 133: 829–837, 1993.
Lehtola L, Partanen J, Sistonen L, Korhonen J, Warri A, Harkonen P, Clarke R, Alitalo K. Analysis of tyrosine kinase mRNAs including four FGF receptor mRNAs expressed in MCF-7 breast-cancer cells. Int J Cancer 50: 598–603, 1992.
Asahara T, Bauters C, Pastore C, Kearney M, Rossow S, Bunting S, Ferrara N, Symes JF, Isner JM. Local delivery of vascular endothelial growth factor accelerates reendothelialization and attenuates intimal hyperplasia in balloon-injured rat carotid artery. Circulation 91: 2793–2801, 1995.
Krasinski K, Spyridopoulos I, Asahara T, van der Zee R, Isner JM, Losordo DW. Estradiol accelerates functional endothelial recovery after arterial injury. Circulation 95: 1768–1772, 1997.
White CR, Chen SJ, Faye-Peterson O, Shelton J, Allen L, Durand J, Chen YF, Oparil S. Effects of gender on the structural and functional re-endothelialization of the balloon injured rat carotid artery. In Press 1998.
Prescott MF, McBride CK, Court M. Development of intimal lesions after leukocyte migration into the vascular wall. Am J Pathol 135: 835–846, 1989.
Beesley JE, Honey AC, Martin JF. Ultrastructural assessment of lesion development in the collared rabbit carotid artery model. Cells and Materials 2: 201–208, 1992.
Shi Y, Pieniek M, Fard A, O’Brien JE, Mannion JD, Zalewski A. Adventitial remodeling following coronary arterial injury. Circulation 93: 340–348, 1996.
Shi Y, O’Brien JE, Fard A, Mannion JD, Wang D, Zalewski A. Adventitial myofibroblasts contribute to neointimal formation in injured porcine coronary arteries. Circulation 94: 1655–1664, 1996.
Clowes AW, Reidy MA, Clowes MM. Kinetics of cellular proliferation after arterial injury, I: smooth muscle cell growth in tha absence of endothelium. Lab Invest 49: 327–333, 1983.
Clowes AW, Reidy MA, Clowes MM. Mechanisms of stenosis after arterial injury. Lab Invest 49: 208–215, 1983.
Lindner V, Reidy MA. Proliferation of smooth muscle cells after vascular injury is inhibited by an antibody against basic fibroblast growth factor. Proc Natl Acad Sci USA 88: 3739–3743, 1991.
Olson NE, Chao S, Lindner V, Reidy MA. Intimal smooth muscle cell proliferation after balloon catheter injury: the role of basic fibroblast growth factor. Am J Pathol. 140: 1017–1023, 1992.
Schwartz SM, DeBlois D, O’Brien ERM. The intima: soil for atherosclerosis and restenosis. Circulation Research 77: 445–465, 1995.
Suzuki A, Mizuno K, Ino Y, Okada M, Kikkawa F, Mizutani S, Tomoda Y. Effects of 17β-estradiol and progesterone on growth-factor-induced proliferation and migration in human female aortic smooth muscle cells in vitro. Cardiovascular Research 31: 516–523, 1996.
Orimo A, Inoue S, Ikegami A, et al. Vascular smooth muscle cells as a target for estrogen. Biochem BiophysRes Commun 195: 730–736, 1993.
Majesky MW. Neointima formation after acute vascular injury. Role of counteradhesive extracellular matrix protein. Tex Heart Inst J 21: 78–85, 1994.
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 1998 Springer Science+Business Media New York
About this chapter
Cite this chapter
Chen, YF., Oparil, S. (1998). Effects of Sex Steroids in Vascular Injury. In: Levin, E.R., Nadler, J.L. (eds) Endocrinology of Cardiovascular Function. Endocrine Updates, vol 1. Springer, Boston, MA. https://doi.org/10.1007/978-1-4615-5569-8_3
Download citation
DOI: https://doi.org/10.1007/978-1-4615-5569-8_3
Publisher Name: Springer, Boston, MA
Print ISBN: 978-1-4613-7547-0
Online ISBN: 978-1-4615-5569-8
eBook Packages: Springer Book Archive