Abstract
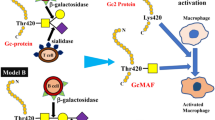
Macrophages are essential for host defense and play an important role in orchestrating immune response of the host against threat signals. Macrophages are also known to have a critical role in antitumor immunity, can infiltrate into tumor, and are found in most tumor sites. Meanwhile, Gc protein (also known as vitamin D-binding protein) is a serum protein with multifunctional properties and has been reported as a precursor for macrophage activation factor. Gc protein can be converted by an inducible ß-galactosidase of B cells and neuraminidase of T cells to a potent macrophage activating factor (GcMAF), a protein with N-acetylgalactosamine (GaINAc) as the remaining sugar moiety. Activated macrophages express tumoricidal activity by ingestion of tumor cells and release of reactive oxygen species (ROS) and reactive nitrogen species (RNS), or both. We reported that in situ modification of Gc protein with B-galactosidase and neuraminidase increased the release of superoxide in thioglycolate-elucidated mouse peritoneal macrophage. Yamamoto et al. reported the possibility of using GcMAF as an immunomodulator for cancer treatment, so it is important to provide an assay for GcMAF. Kanan et al. reported the quantitative analysis of GcMAF from human serum. However, the sugar moiety of GcMAF has never been qualitatively studied. Here we present the study of qualitative analysis of GcMAF from purified human serum as well as its influence on the macrophage activity.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Preview
Unable to display preview. Download preview PDF.
Similar content being viewed by others
References
J. MacMicking, Q. W. Xie, and C. Nathan, Nitric Oxide and macrophage functionAnn. Rev. Immunol. 15 323–350 (1997).
A. Mantovani, B. Bottazzi, F. Colotta, S. Sozzani, and L. Ruco, The origin and function of tumor associated macrophage, Immunol. Today 13 265–270 (1992).
R. Sejelid, and L. T. Busund, The biology of macrophage: I1. Inflammation and tumors, Eur. J. Haenratol. 52, 1–12 (1993).
P. White, and N. Cooke, The multifunctional properties and characteristics of vitamin D-binding protein, TEM. 11 320–327 (2000).
N. Yamamoto, and S. Homma, Vitamin D3 binding protein (group specific component) is a precursor for the macrophage-activating signal, Proc.Natl. Acad. Sci. USA 88 8539–8543 (1991).
N. Yamamoto, and R. Kumashiro, Conversion of vitamin D3 binding protein (group specific component) to a macrophage activating factor by the stepwise action of ß-galactosidase of B cells and sialidase of T cells, J. lmmunol. 151 2794–2802 (1993).
N. Yamamoto, S. Homma, J. G. Haddad, M. A. Kowalski, Vitamin D3 binding protein required for in vitro activation of macrophages after alkylglycerol treatment of mouse peritoneal cells, immunology. 74 420–424 (1991).
S. B. Mohamad, 14. Nagasawa, Y. Uto, & Hitoshi, Hori, Tumor cell alpha-N-acetylgalactosaminidase activity and its involvement in GcMAF-related macrophage activationComp. Biochem. Physiol. B in press (2001).
N. Yamamoto, Structural definition of a potent macrophage activating factor derived from vitamin D3-binding protein with adjuvant activity for antibody production, Mol. Immunol, 33, 1157–1164 (1996).
R. M. Kanan, D. B. Cook, H. K. Datta,. Lectin immunoassay for macrophage-activating factor (Gc-MAF) produced by deglycosylation of Gc-globulin: Evidence for noninducible generation of Gc-MAF, Clin. Chem. 46 412–414 (2000).
R. P. Link, K. L. Perlman, E. A. Pierce, H. K. Schoes, and H. F. DeLuca, Purification of human serum vitamin D-binding protein by 25-hydroxyvitamin D3-sepharose chromatography, Anal. Biochem 157 262–269 (1986).
R. B. Johnston, C. A. Godzik, and Z. A. Cohn, Increased superoxide anion production by immunologically activated and chemically elicited macrophagesJ. Exp. Med. 148 115–127 (1978).
S. Hammarstrom, and E. A. Kabat, Studies on specificity and binding properties of the blood group A reactive hemagglutinin from Helix pomatia, Biochemistry 10, 1684–1692 (1971).
N. Yamamoto, and V. R. Naraparaju, A defect in inducible ß-galactosidase of B lymphocytes in the osteopetrotic (mi/mi) mouse, Immunology. 88 604–610 (1996).
P. R. Odgren, S. N. Popoff, F. F. Safadi, C. A. MacKay, A. Mason-Savas, M. F. Seifert., and S. C. Marks Jr, The toothless osteopetrotic rat has a normal vitamin D-binding protein-macrophage activating factor (DBP-MAF) cascade and chondrodysplasia resistant to treatments with Colony Stimulating Factor-I (CSF-1) and/or DBP-MAF, Bone 25 175–181 (1999).
N. Swamy, S. Ghosh, G. B. Schneider, and R Ray, Baculovirus-expressed vitamin D-binding protein-macrophage activating factor (DBP-MAF) activates osteoclasts and binding of 25-hydroxyvitamin D3 does not influence this activity, J. Cell. Biochem. 81 535–546 (2001).
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2003 Springer Science+Business Media New York
About this chapter
Cite this chapter
Mohamad, S.B., Hori, H., Nagasawa, H., Usui, K., Uto, Y. (2003). Characterization of Human Gc Protein- Derived Macrophage Activation Factor (GcMAF) and Its Functional Role in Macrophage Tumoricidal Activity. In: Wilson, D.F., Evans, S.M., Biaglow, J., Pastuszko, A. (eds) Oxygen Transport To Tissue XXIII. Advances in Experimental Medicine and Biology, vol 510. Springer, Boston, MA. https://doi.org/10.1007/978-1-4615-0205-0_13
Download citation
DOI: https://doi.org/10.1007/978-1-4615-0205-0_13
Publisher Name: Springer, Boston, MA
Print ISBN: 978-1-4613-4964-8
Online ISBN: 978-1-4615-0205-0
eBook Packages: Springer Book Archive