Abstract
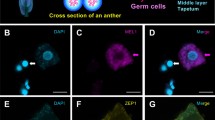
Small RNAs specifically expressed in reproductive tissues are key regulators of germline development in eukaryotes. Rice microRNA2118 (miR2118), which is enriched during reproduction in grasses, is a trigger to produce phased small interfering RNAs (phasiRNAs). These phasiRNAs demonstrate the temporal regulation with premeiotic phasiRNAs and meiotic phasiRNAs in anther development. Furthermore, the site-specific regulation via miR2118 and phasiRNAs is of importance in soma and germ development in anthers. Accordingly, histological imaging methods are essential tools for understanding spatiotemporal regulation during reproduction and elucidating the reproductive roles of miRNAs and phasiRNAs. We successfully developed a method to visualize the three-dimensional (3D) structure of entire rice anthers, which can also be used for distinguishing the internal structure of the anthers in other plants. Here, we describe the detailed methods of in situ hybridization for miR2118 localization and the visualization of the 3D structure of entire anthers of rice.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Iwakawa HO, Tomari Y (2015) The functions of MicroRNAs: mRNA decay and translational repression. Trends Cell Biol 25(11):651–665
Komiya R (2017) Biogenesis of diverse plant phasiRNAs involves an miRNA-trigger and dicer-processing. J Plant Res 130(1):17–23
Johnson C, Kasprzewska A, Tennessen K, Fernandes J, Nan GL, Walbot V, Sundaresan V, Vance V, Bowman LH (2009) Clusters and superclusters of phased small RNAs in the developing inflorescence of rice. Genome Res 19(8):1429–1440
Song X, Li P, Zhai J, Zhou M, Ma L, Liu B, Jeong DH, Nakano M, Cao S, Liu C et al (2012) Roles of DCL4 and DCL3b in rice phased small RNA biogenesis. Plant J 69(3):462–474
Komiya R, Ohyanagi H, Niihama M, Watanabe T, Nakano M, Kurata N, Nonomura K (2014) Rice germline-specific Argonaute MEL1 protein binds to phasiRNAs generated from more than 700 lincRNAs. Plant J 78(3):385–397
Song X, Wang D, Ma L, Chen Z, Li P, Cui X, Liu C, Cao S, Chu C, Tao Y et al (2012) Rice RNA-dependent RNA polymerase 6 acts in small RNA biogenesis and spikelet development. Plant J 71(3):378–389
Zhai J, Zhang H, Arikit S, Huang K, Nan GL, Walbot V, Meyers BC (2015) Spatiotemporally dynamic, cell-type-dependent premeiotic and meiotic phasiRNAs in maize anthers. Proc Natl Acad Sci U S A 112(10):3146–3151
Teng C, Zhang H, Hammond R, Huang K, Meyers BC, Walbot V (2020) Dicer-like 5 deficiency confers temperature-sensitive male sterility in maize. Nat Commun 11(1):2912
Nonomura K, Miyoshi K, Eiguchi M, Suzuki T, Miyao A, Hirochika H, Kurata N (2003) The MSP1 gene is necessary to restrict the number of cells entering into male and female sporogenesis and to initiate anther wall formation in rice. Plant Cell 15(8):1728–1739
Fu Z, Yu J, Cheng X, Zong X, Xu J, Chen M, Li Z, Zhang D, Liang W (2014) The Rice basic helix-loop-helix transcription factor TDR INTERACTING PROTEIN2 is a central switch in early anther development. Plant Cell 26(4):1512–1524
Ta KN, Sabot F, Adam H, Vigouroux Y, De Mita S, Ghesquiere A, Do NV, Gantet P, Jouannic S (2016) miR2118-triggered phased siRNAs are differentially expressed during the panicle development of wild and domesticated African rice species. Rice 9(1):10
Araki S, Le NT, Koizumi K, Villar-Briones A, Nonomura KI, Endo M, Inoue H, Saze H, Komiya R (2020) miR2118-dependent U-rich phasiRNA production in rice anther wall development. Nat Commun 11(1):3115
Komiya R (2021) Spatiotemporal regulation and roles of reproductive phasiRNAs in plants. Genes Genet Syst 96(5):209–215
Jiang P, Lian B, Liu C, Fu Z, Shen Y, Cheng Z, Qi Y (2020) 21-nt phasiRNAs direct target mRNA cleavage in rice male germ cells. Nat Commun 11(1):5191
Zhang YC, Lei MQ, Zhou YF, Yang YW, Lian JP, Yu Y, Feng YZ, Zhou KR, He RR, He H et al (2020) Reproductive phasiRNAs regulate reprogramming of gene expression and meiotic progression in rice. Nat Commun 11(1):6031
Malone CD, Hannon GJ (2009) Small RNAs as guardians of the genome. Cell 136(4):656–668
Sato K, Siomi MC (2018) Two distinct transcriptional controls triggered by nuclear Piwi-piRISCs in the drosophila piRNA pathway. Curr Opin Struct Biol 53:69–76
Ursache R, Andersen TG, Marhavý P, Geldner N (2018) A protocol for combining fluorescent proteins with histological stains for diverse cell wall components. Plant J 93(2):399–412
Acknowledgments
This work was supported by JST PRESTO Program (Grant number JPMJPR17Q3, Japan), JST FORESTO Program (Grant Number JPMJFR204U, Japan), KAKENHI Programs (Grant numbers JP17H05608 and JP15H01476), the Naito Foundation, and the Okinawa Institute of Science and Technology Graduate University, Japan, to R.K. We thank Ms. Saori Araki and Ms. Hinako Tamotsu for rice growth and sampling.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
1 Electronic Supplementary Material
The 3D imaging movie of the 1 mm rice anther taken by a Lightsheet microscope. Cyan shows the fluorescence of the somatic anther walls stained with SR2200 dye. Magenta shows autofluorescence of the anther. Laser excitation/emission: 405 nm/420-470 nm (SR2200), 561 nm/575 nm LP (autofluorescence). (MOV 9549 kb)
Rights and permissions
Copyright information
© 2022 The Author(s), under exclusive license to Springer Science+Business Media, LLC, part of Springer Nature
About this protocol
Cite this protocol
Koizumi, K., Komiya, R. (2022). 3D Imaging and In Situ Hybridization for Uncovering the Functions of MicroRNA in Rice Anther. In: Parrish, N.F., Iwasaki, Y.W. (eds) piRNA. Methods in Molecular Biology, vol 2509. Humana, New York, NY. https://doi.org/10.1007/978-1-0716-2380-0_6
Download citation
DOI: https://doi.org/10.1007/978-1-0716-2380-0_6
Published:
Publisher Name: Humana, New York, NY
Print ISBN: 978-1-0716-2379-4
Online ISBN: 978-1-0716-2380-0
eBook Packages: Springer Protocols