Abstract
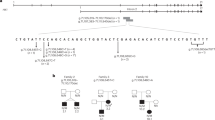
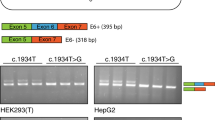
It is a widely held paradigm in molecular biology that a change in the third base of a codon is silent in terms of expression. In this investigation, results are presented that challenge that paradigm, at least in terms of one polymorphism in KCNJ11, which is one of five genes that have been implicated in the disorder Hyperinsulinism of Infancy. In two cohorts of Australian patients, an uneven distribution of KCNJ11 SNP’s was observed. A silent polymorphism at codon 190 was over-represented in the patients who responded well to medical treatment and under-represented in those that required radical surgical intervention. In an attempt to investigate this polymorphism, it was expressed in vitro and western blot analysis showed that there were virtually no bands from the homozygous variant samples, while strong bands were seen in normal controls. The human genome is highly redundant in terms of tRNA species for each amino acids but enigmatically under-represents a number of specific codons. The polymorphism in question occurs within one such codon. We propose that the presence of a base change at the third position of codon that is not represented by a corresponding anti-codon within the human nuclear tRNA leads to a decreased rate of expression of the protein.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Preview
Unable to display preview. Download preview PDF.
Similar content being viewed by others
References
Dunne MJ, Cosgrove KE, Shepherd RM, Aynsley-Green A, Lindley KJ. Hyperinsulinism in Infancy: from Basic Science to Clinical Disease. Physiol Rev, 2004. 84(1), 239–275.
Aynsley-Green A, Hussain K, Hall J, Saudubray JM, Nihoul-Fekete C, De Lonlay-Debeney P, Brunelle F, Otonkoski T, Thornton P, Lindley KJ. Practical management of hyperinsulinism in infancy. Arch Dis Child Fetal Neonatal Ed. Arch Dis Child, 2000. 82(2), F98–F107.
Davis EA, Cuesta-Munoz A, Raoul M, Buettger C, Sweet I, Moates M, Magnuson MA, Matschinsky FM. Mutants of glucokinase cause hypoglycaemia- and hyperglycaemia syndromes and their analysis illuminates fundamental quantitative concepts of glucose homeostasis. Diabetologia, 1999. 42(10), 1175–1186.
Stanley CA, Lieu YK, Hsu BY, Burlina AB, Greenberg CR, Hopwood NJ, Perlman K, Rich BH, Zammarchi E, Poncz M. Hyperinsulinism and hyperammonemia in infants with regulatory mutations of the glutamate dehydrogenase gene. N Engl J Med, 1998. 338(19), 1352–1357.
Molven A, Matre GE, Duran M, Wanders RJ, Rishaug U, Njolstad PR, Jellum E, Sovik O. Familial Hyperinsulinemic Hypoglycemia caused by a defect in the SCHAD enzyme of mitochondrial fatty acid oxidation. Diabetest, 2004. (531), 221–227.
Molven A, Rishaug U, Matre GE, Njolstad PR, Sovik O. Hunting for a hypoglycemia gene: Severe neonatal hypoglycemia in a consanguineous family. Am J Med Genet, 2002. 113(1) 40–46.
Jack MM, Greer RM, Thomsett MJ, Walker RM, Bell JR, Choong C, Cowley DM, Herington AC, Cotterill AM. The outcome in Australian children with hyperinsulinism of infancy: Early extensive surgery in severe cases lowers risk of diabetes. Clin Endocrinol (Oxf), 2003. 58(3), 355–364.
Shah JH, Maguire DJ, Brown D, Cotterill AM. The role of ATP sensitive channels in insulin secretion and the implications in Persistent Hyperinsulinemic Hypoglycaemia of Infancy (PHHI), In Press
Database of Single Nucleotide Polymorphisms (dbSNP). Bethesda (MD): National Center for Biotechnology Information, National Library of Medicine. dbSNP accession: rs5218. Available from: http://www.ncbi.nlm.nih.gov/SNP/http://www.ncbi.nlm.nih.gov/SNP/
Nielsen KB, Sorensen S, Cartegni L, Corydon TJ, Doktor TK, Schroeder LD, Reinert LS, Elpeleg O, Krainer AR, Gregersen N, Kjems J, Andresen BS. Seemingly neutral polymorphic variants may confer immunity to splicing-inactivating mutations: A synonymous SNP in exon 5 of MCAD protects from deleterious mutations in a flanking exonic splicing enhancer. Am J Hum Genet. 2007 80(3), 416–432.
Lowe TM, Eddy SR.tRNAscan-SE: A program for improved detection of transfer RNA genes in genomic sequence Nucl. Acids Res. 1997. 25, 955–964. (The genomic tRNA database. (http://lowelab.ucsc.edu/GtRNAdb/)http://lowelab.ucsc.edu/GtRNAdb/)
Nakamura, Y. Codon Usage Datebase. (http://www.kazusa.or.jp/codon/)http://www.kazusa.or.jp/codon/)12th June 2006.
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2008 Springer Science+Business Media, LLC
About this paper
Cite this paper
Shah, J., Maguire, D., Munce, T., Cotterill, A. (2008). Alanine in HI: A Silent Mutation Cries Out!. In: Kang, K.A., Harrison, D.K., Bruley, D.F. (eds) Oxygen Transport to Tissue XXIX. Advances In Experimental Medicine And Biology, vol 614. Springer, Boston, MA. https://doi.org/10.1007/978-0-387-74911-2_17
Download citation
DOI: https://doi.org/10.1007/978-0-387-74911-2_17
Publisher Name: Springer, Boston, MA
Print ISBN: 978-0-387-74910-5
Online ISBN: 978-0-387-74911-2
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)