Abstract
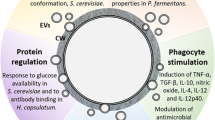
The comprehension of fungal biology is important for several reasons. Besides being used in biotechnological processes and in the food industry, fungi are also important animal and vegetal pathogens. Fungal diseases in humans have a great importance worldwide, and understanding fungal biology is crucial for treatment and prevention of these diseases, especially because of emerging antifungal resistance that poses great epidemiological risks. Communication through extracellular vesicles is a ubiquitous mechanism of molecule transfer between cells and is used to transport proteins, nucleic acids, lipids, and other biologically active molecules. Several pathogens can produce and transfer extracellular vesicles, and the importance of this pathway in fungal communication with hosts and between fungal cells has been described for several species in the last years, as shown for Saccharomyces cereviseae, Cryptococcus neoformans, Candida albicans, Paracoccidioides braziliensis, Sporothrix schenckii, Candida parapsilosis, Malassezia sympodialis, Histoplasma capsulatum, among others. In this chapter, we review the role of extracellular vesicles in fungal communication, interaction with hosts and with the environment, and also highlighting important molecules found in fungal EVs.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Albuquerque PC, Nakayasu ES, Rodrigues ML, et al (2008) Vesicular transport in Histoplasma capsulatum: an effective mechanism for trans-cell wall transfer of proteins and lipids in ascomycetes. Cell Microbiol 10:1695–1710. https://doi.org/10.1111/j.1462-5822.2008.01160.x
Anderson J, Mihalik R, Soll DR (1990) Ultrastructure and antigenicity of the unique cell wall pimple of the Candida opaque phenotype. J Bacteriol 172:224–235
Andrade CM, Tinoco MLP, Rieth AF et al (2016) Host-induced gene silencing in the necrotrophic fungal pathogen Sclerotinia sclerotiorum. Plant Pathol 65:626–632. https://doi.org/10.1111/ppa.12447
Arroyo JD, Chevillet JR, Kroh EM et al (2011) Argonaute2 complexes carry a population of circulating microRNAs independent of vesicles in human plasma. Proc Natl Acad Sci U S A 108:5003–5008. https://doi.org/10.1073/pnas.1019055108
Bielska E, Sisquella MA, Aldeieg M et al (2018) Pathogen-derived extracellular vesicles mediate virulence in the fatal human pathogen Cryptococcus gattii. Nat Commun 9. https://doi.org/10.1038/s41467-018-03991-6
Binder U, Lass-Flörl C (2011) Epidemiology of invasive fungal infections in the Mediterranean area. Mediterr J Hematol Infect Dis 3. https://doi.org/10.4084/mjhid.2011.016
Bongomin F, Gago S, Oladele R, Denning D (2017) Global and multi-national prevalence of fungal diseases—estimate precision. J Fungi 3:57. https://doi.org/10.3390/jof3040057
Brown L, Wolf JM, Prados-Rosales R, Casadevall A (2015) Through the wall: extracellular vesicles in Gram-positive bacteria, mycobacteria and fungi. Nat Rev Microbiol 13:620–630. https://doi.org/10.1038/nrmicro3480
Cai Q, Qiao L, Wang M et al (2018) Plants send small RNAs in extracellular vesicles to fungal pathogen to silence virulence genes. Science 360:1126–1129. https://doi.org/10.1126/science.aar4142
Chaloner T, van Kan JAL, Grant-Downton RT (2016) RNA ‘Information Warfare’ in pathogenic and mutualistic interactions. Trends Plant Sci 21:738–748. https://doi.org/10.1016/j.tplants.2016.05.008
Cheng W, Song X-S, Li H-P et al (2015) Host-induced gene silencing of an essential chitin synthase gene confers durable resistance to Fusarium head blight and seedling blight in wheat. Plant Biotechnol J 13:1335–1345. https://doi.org/10.1111/pbi.12352
Cipriano MJ, Hajduk SL (2018) Drivers of persistent infection: pathogen-induced extracellular vesicles. Essays Biochem 62:135–147. https://doi.org/10.1042/EBC20170083
Da Silva RP, Heiss C, Black I et al (2015) Extracellular vesicles from Paracoccidioides pathogenic species transport polysaccharide and expose ligands for DC-SIGN receptors. Sci Rep 5:1–12. https://doi.org/10.1038/srep14213
Da Silva TA, Roque-Barreira MC, Casadevall A, Almeida F (2016) Extracellular vesicles from Paracoccidioides brasiliensis induced M1 polarization in vitro. Sci Rep 6:1–10. https://doi.org/10.1038/srep35867
Eisenman HC, Mues M, Weber SE et al (2007) Cryptococcus neoformans laccase catalyses melanin synthesis from both d- and l-DOPA. Microbiology 153:3954–3962. https://doi.org/10.1099/mic.0.2007/011049-0
Farmakiotis D, Kontoyiannis DP (2017) Epidemiology of antifungal resistance in human pathogenic yeasts: current viewpoint and practical recommendations for management. Int J Antimicrob Agents 50:318–324. https://doi.org/10.1016/j.ijantimicag.2017.05.019
Gehrmann U, Qazi KR, Johansson C et al (2011) Nanovesicles from malassezia sympodialis and host exosomes induce cytokine responses—novel mechanisms for host-microbe interactions in atopic eczema. PLoS ONE 6:1–10. https://doi.org/10.1371/journal.pone.0021480
Ghag SB, Shekhawat UKS, Ganapathi TR (2014) Host-induced post-transcriptional hairpin RNA-mediated gene silencing of vital fungal genes confers efficient resistance against Fusarium wilt in banana. Plant Biotechnol J 12:541–553. https://doi.org/10.1111/pbi.12158
Ghannoum MA, Jurevic RJ, Mukherjee PK et al (2010) Characterization of the oral fungal microbiome (mycobiome) in healthy individuals. PLoS Pathog 6. https://doi.org/10.1371/journal.ppat.1000713
Harding C, Heuser J, Stahl P (1984) Endocytosis and intracellular processing of transferrin and colloidal gold-transferrin in rat reticulocytes: demonstration of a pathway for receptor shedding. Eur J Cell Biol 35:256–263
Huang SH, Wu CH, Chang YC et al (2012) Cryptococcus neoformans-derived microvesicles enhance the pathogenesis of fungal brain infection. PLoS One 7. https://doi.org/10.1371/journal.pone.0048570
Huffnagle GB, Noverr MC (2013) The emerging world of the fungal microbiome. Trends Microbiol 21:334–341. https://doi.org/10.1016/j.tim.2013.04.002
Iguchi H, Kosaka N, Ochiya T (2010) Secretory microRNAs as a versatile communication tool. Commun Integr Biol 3:478–481. https://doi.org/10.4161/cib.3.5.12693
Joffe LS, Nimrichter L, Rodrigues ML, Del Poeta M (2016) Potential roles of fungal extracellular vesicles during infection. mSphere 1. https://doi.org/10.1128/msphere.00099-16
Johansson HJ, Vallhov H, Holm T et al (2018) Extracellular nanovesicles released from the commensal yeast Malassezia sympodialis are enriched in allergens and interact with cells in human skin. Sci Rep 8:9182. https://doi.org/10.1038/s41598-018-27451-9
Kabani M, Melki R (2015) Sup35p in its soluble and prion states is packaged inside extracellular vesicles. MBio 6. https://doi.org/10.1128/mbio.01017-15
Kerr JF, Wyllie AH, Currie AR (1972) Apoptosis: a basic biological phenomenon with wide-ranging implications in tissue kinetics. Br J Cancer 26:239–257
Kmetzsch L, Joffe LS, Staats CC et al (2011) Role for golgi reassembly and stacking protein (GRASP) in polysaccharide secretion and fungal virulence. Mol Microbiol 81:206–218. https://doi.org/10.1111/j.1365-2958.2011.07686.x
Kutralam-Muniasamy G, Flores-Cotera LB, Perez-Guevara F (2015) Potential of yeast secretory vesicles in biodelivery systems. Drug Discov Today 20:659–666. https://doi.org/10.1016/j.drudis.2015.03.014
Leone F, Bellani L, Muccifora S et al (2018) Analysis of extracellular vesicles produced in the biofilm by the dimorphic yeast Pichia fermentans. J Cell Physiol 233:2759–2767. https://doi.org/10.1002/jcp.25885
Liu S, Hossinger A, Hofmann JP et al (2016) Horizontal transmission of cytosolic Sup35 prions by extracellular vesicles. MBio 7. https://doi.org/10.1128/mbio.00915-16
Matos Baltazar L, Nakayasu ES, Sobreira TJ et al (2016) Antibody binding alters the characteristics and contents of extracellular vesicles released by Histoplasma capsulatum. mSphere 1:1–17. https://doi.org/10.1128/msphere.00085-15 [pii]\rmSphere00085-15 [pii]
Nimrichter L, De Souza MM, Del Poeta M et al (2016) Extracellular vesicle-associated transitory cell wall components and their impact on the interaction of fungi with host cells. Front Microbiol 7:1–11. https://doi.org/10.3389/fmicb.2016.01034
Nimrichter L, Rodrigues ML (2011) Fungal glucosylceramides: from structural components to biologically active targets of new antimicrobials. Front Microbiol 2:212. https://doi.org/10.3389/fmicb.2011.00212
Nowara D, Gay A, Lacomme C et al (2010) HIGS: host-induced gene silencing in the obligate biotrophic fungal pathogen Blumeria graminis. Plant Cell 22:3130–3141. https://doi.org/10.1105/tpc.110.077040
Oliveira DL, Freire-de-Lima CG, Nosanchuk JD et al (2010a) Extracellular vesicles from Cryptococcus neoformans modulate macrophage functions. Infect Immun 78:1601–1609. https://doi.org/10.1128/IAI.01171-09
Oliveira DL, Nakayasu ES, Joffe LS et al (2010b) Characterization of yeast extracellular vesicles: evidence for the participation of different pathways of cellular traffic in vesicle biogenesis. PLoS One 5. https://doi.org/10.1371/journal.pone.0011113
Oliveira DL, Rizzo J, Joffe LS et al (2013) Where do they come from and where do they go: candidates for regulating extracellular vesicle formation in fungi. Int J Mol Sci 14:9581–9603. https://doi.org/10.3390/ijms14059581
Panepinto J, Komperda K, Frases S et al (2009) Sec6-dependent sorting of fungal extracellular exosomes and laccase of Cryptococcus neoformans. Mol Microbiol 71:1165–1176. https://doi.org/10.1111/j.1365-2958.2008.06588.x
Panwar V, McCallum B, Bakkeren G (2013) Endogenous silencing of Puccinia triticina pathogenicity genes through in planta-expressed sequences leads to the suppression of rust diseases on wheat. Plant J 73:521–532. https://doi.org/10.1111/tpj.12047
Peres da Silva R, Puccia R, Rodrigues ML et al (2015) Extracellular vesicle-mediated export of fungal RNA. Sci Rep 5:7763. https://doi.org/10.1038/srep07763
Pliego C, Nowara D, Bonciani G et al (2013) Host-induced gene silencing in barley powdery mildew reveals a class of ribonuclease-like effectors. Mol Plant Microbe Interact 26:633–642. https://doi.org/10.1094/MPMI-01-13-0005-R
Raposo G, Stoorvogel W (2013) Extracellular vesicles: exosomes, microvesicles, and friends. J Cell Biol 200:373–383. https://doi.org/10.1083/jcb.201211138
Rayner S, Bruhn S, Vallhov H et al (2017) Identification of small RNAs in extracellular vesicles from the commensal yeast Malassezia sympodialis. Sci Rep 7:1–9. https://doi.org/10.1038/srep39742
Rella A, Farnoud AM, Del Poeta M (2016) Plasma membrane lipids and their role in fungal virulence. Prog Lipid Res 61:63–72. https://doi.org/10.1016/j.plipres.2015.11.003
Rizzo J, Albuquerque PC, Wolf JM et al (2017a) Analysis of multiple components involved in the interaction between Cryptococcus neoformans and Acanthamoeba castellanii. Fungal Biol 121:602–614. https://doi.org/10.1016/j.funbio.2017.04.002
Rizzo J, Nimrichter L, Rodrigues ML (2017b) What is new? Recent knowledge on fungal extracellular vesicles. Curr Fungal Infect Rep 11:141–147. https://doi.org/10.1007/s12281-017-0293-6
Rodrigues ML, Franzen AJ, Nimrichter L, Miranda K (2013) Vesicular mechanisms of traffic of fungal molecules to the extracellular space. Curr Opin Microbiol 16:414–420. https://doi.org/10.1016/j.mib.2013.04.002
Rodrigues ML, Nakayasu ES, Almeida IC, Nimrichter L (2014) The impact of proteomics on the understanding of functions and biogenesis of fungal extracellular vesicles. J Proteomics 97:177–186. https://doi.org/10.1016/j.jprot.2013.04.001
Rodrigues ML, Nakayasu ES, Oliveira DL et al (2008a) Extracellular vesicles produced by Cryptococcus neoformans contain protein components associated with virulence. Eukaryot Cell 7:58–67. https://doi.org/10.1128/EC.00370-07
Rodrigues ML, Nimrichter L, Cordero RJB, Casadevall A (2011) Fungal polysaccharides: biological activity beyond the usual structural properties. Front Microbiol 2:171. https://doi.org/10.3389/fmicb.2011.00171
Rodrigues ML, Nimrichter L, Oliveira DL et al (2008b) Vesicular trans-cell wall transport in fungi: a mechanism for the delivery of virulence-associated macromolecules? Lipid Insights 2:27–40
Rodrigues ML, Nimrichter L, Oliveira DL et al (2007) Vesicular polysaccharide export in Cryptococcus neoformans is a eukaryotic solution to the problem of fungal trans-cell wall transport. Eukaryot Cell 6:48–59. https://doi.org/10.1128/EC.00318-06
Rodrigues ML, Travassos LR, Miranda KR et al (2000) Human antibodies against a purified glucosylceramide from Cryptococcus neoformans inhibit cell budding and fungal growth. Infect Immun 68:7049–7060
Shi L, Albuquerque PC, Lazar-Molnar E et al (2008) A monoclonal antibody to Histoplasma capsulatum alters the intracellular fate of the fungus in murine macrophages. Eukaryot Cell 7:1109–1117. https://doi.org/10.1128/EC.00036-08
Silva BMA, Prados-Rosales R, Espadas-Moreno J et al (2014) Characterization of Alternaria infectoria extracellular vesicles. Med Mycol 52:202–210. https://doi.org/10.1093/mmy/myt003
Tinoco M, Dias BB, Dall’Astta RC et al (2010) In vivo trans-specific gene silencing in fungal cells by in planta expression of a double-stranded RNA. BMC Biol 8:27. https://doi.org/10.1186/1741-7007-8-27
Valadi H, Ekström K, Bossios A et al (2007) Exosome-mediated transfer of mRNAs and microRNAs is a novel mechanism of genetic exchange between cells. Nat Cell Biol 9:654–659. https://doi.org/10.1038/ncb1596
Vallejo MC, Nakayasu ES, Longo LVG et al (2012a) Lipidomic analysis of extracellular vesicles from the pathogenic phase of Paracoccidioides brasiliensis. PLoS ONE 7:1–10. https://doi.org/10.1371/journal.pone.0039463
Vallejo MC, Nakayasu ES, Matsuo AL et al (2012b) Vesicle and vesicle-free extracellular proteome of paracoccidioides brasiliensis: comparative analysis with other pathogenic fungi. J Proteome Res 11:1676–1685. https://doi.org/10.1021/pr200872s
Vallejo MC, Puccia R (2011) The pathogenic fungus Paracoccidioides brasiliensis exports extracellular vesicles containing highly immunogenic alfa-Galactosyl epitopes. Eukaryot Cell
van der Grein SG, Nolte-’t Hoen ENM (2014) “Small Talk” in the innate immune system via RNA-containing extracellular vesicles. Front Immunol 5:542. https://doi.org/10.3389/fimmu.2014.00542
van der Pol L, Stork M, van der Ley P (2015) Outer membrane vesicles as platform vaccine technology. Biotechnol J 10:1689–1706. https://doi.org/10.1002/biot.201400395
Vargas G, Rocha JDB, Oliveira DL et al (2015) Compositional and immunobiological analyses of extracellular vesicles released by Candida albicans. Cell Microbiol 17:389–407. https://doi.org/10.1111/cmi.12374
Wang M, Weiberg A, Lin F-M et al (2016) Bidirectional cross-kingdom RNAi and fungal uptake of external RNAs confer plant protection. Nat Plants 2:16151. https://doi.org/10.1038/nplants.2016.151
Wang X, Thompson CD, Weidenmaier C, Lee JC (2018) Release of Staphylococcus aureus extracellular vesicles and their application as a vaccine platform. Nat Commun 9:1379. https://doi.org/10.1038/s41467-018-03847-z
Weiberg A, Wang M, Lin F-M et al (2013) Fungal small RNAs suppress plant immunity by hijacking host RNA interference pathways. Science 342:118–123. https://doi.org/10.1126/science.1239705
Winters CM, Chiang H-L (2016) Yeast as a model system to study trafficking of small vesicles carrying signal-less proteins in and out of the cell. Curr Protein Pept Sci 17:808–820
Wolf JM, Casadevall A (2014) Challenges posed by extracellular vesicles from eukaryotic microbes. Curr Opin Microbiol 22:73–78. https://doi.org/10.1016/j.mib.2014.09.012
Wolf JM, Espadas-Moreno J, Luque-Garcia JL, Casadevall A (2014) Interaction of Cryptococcus neoformans extracellular vesicles with the cell wall. Eukaryot Cell 13:1484–1493. https://doi.org/10.1128/EC.00111-14
Wolf JM, Rivera J, Casadevall A (2012) Serum albumin disrupts Cryptococcus neoformans and Bacillus anthracis extracellular vesicles. Cell Microbiol 14:762–773. https://doi.org/10.1111/j.1462-5822.2012.01757.x
Yin C, Jurgenson JE, Hulbert SH (2011) Development of a host-induced RNAi system in the wheat stripe rust fungus Puccinia striiformis f. sp. tritici. Mol Plant-Microbe Interact 24:554–561. https://doi.org/10.1094/MPMI-10-10-0229
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2018 Springer Nature Switzerland AG
About this chapter
Cite this chapter
de Toledo Martins, S., Szwarc, P., Goldenberg, S., Alves, L.R. (2018). Extracellular Vesicles in Fungi: Composition and Functions. In: Rodrigues, M. (eds) Fungal Physiology and Immunopathogenesis . Current Topics in Microbiology and Immunology, vol 422. Springer, Cham. https://doi.org/10.1007/82_2018_141
Download citation
DOI: https://doi.org/10.1007/82_2018_141
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-030-30236-8
Online ISBN: 978-3-030-30237-5
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)